* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Matter Unit
Electronegativity wikipedia , lookup
Hypervalent molecule wikipedia , lookup
Molecular orbital diagram wikipedia , lookup
Resonance (chemistry) wikipedia , lookup
Periodic table wikipedia , lookup
Elementary particle wikipedia , lookup
Photoelectric effect wikipedia , lookup
Metastable inner-shell molecular state wikipedia , lookup
Photoredox catalysis wikipedia , lookup
X-ray photoelectron spectroscopy wikipedia , lookup
X-ray fluorescence wikipedia , lookup
Chemistry: A Volatile History wikipedia , lookup
Metallic bonding wikipedia , lookup
Electron transport chain wikipedia , lookup
Low-energy electron diffraction wikipedia , lookup
Marcus theory wikipedia , lookup
History of chemistry wikipedia , lookup
IUPAC nomenclature of inorganic chemistry 2005 wikipedia , lookup
Condensed matter physics wikipedia , lookup
Extended periodic table wikipedia , lookup
Chemical bond wikipedia , lookup
Atomic orbital wikipedia , lookup
Atomic nucleus wikipedia , lookup
Rutherford backscattering spectrometry wikipedia , lookup
Light-dependent reactions wikipedia , lookup
Photosynthetic reaction centre wikipedia , lookup
Hydrogen atom wikipedia , lookup
History of molecular theory wikipedia , lookup
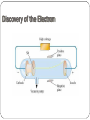
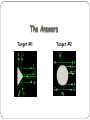
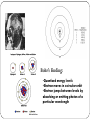
Matter Unit Development of the Atomic Theory https://video.weber.k12.ut.us/vportal/VideoPlayer.jsp?ccsi d=6B8E52B30643AEB849FBD9552FD102E9:1 460 B.C. – Democritus Democritus believed that all matter was composed of atoms that could be divided no further. 350 B.C. - Aristotle Modified an earlier theory that matter was made of four “elements”: earth, fire, water, air. This theory was wrong. However, his theory persisted for 2000 years. 1800 – John Dalton Dalton's Atomic Theory: Matter is composed of small particles called atoms. All atoms of an element are identical, but are different from those of any other element. During chemical reactions, atoms are neither created nor destroyed, but are simply rearranged. Atoms always combine in whole number multiples of each other. For example, 1:1, 1:2, 2:3 or 1:3. 1897- J.J. Thomson Thomson’s 'Raisin in the Pudding' model of the atom He discovered the electron and proposed a model for the structure of the atom. Positive Sphere Negative Charges Discovery of the Electron 1910 – Ernest Rutherford Rutherford’s Gold Foil Experiment Try it Yourself! In the following pictures, there is a target hidden by a cloud. To figure out the shape of the target, we shot some beams into the cloud and recorded where the beams came out. Can you figure out the shape of the target? The Answers Target #1 Target #2 Rutherford’s Findings “Like howitzer shells bouncing off of tissue paper!” Conclusions: The nucleus is small The nucleus is dense The nucleus is positively charged 1912 – Neils Bohr Bohr came up with two rules which agreed with his experiment: RULE 1: Electrons can orbit only at certain allowed distances from the nucleus. RULE 2: Atoms radiate energy when an electron jumps from a higher-energy orbit to a lower-energy orbit. Also, an atom absorbs energy when an electron gets boosted from a low- energy orbit to a high-energy orbit. Bohr’s Finding: •Quantized energy levels •Electron moves in a circular orbit •Electron jumps between levels by absorbing or emitting photon of a particular wavelength Practice Bohr Diagrams James Chadwick 1932 Discovered the Neutron. Quantum Mechanical Model Electrons are located in specific energy levels. There is no exact path around the nucleus. The model estimates the probability of finding an electron in a certain position. The Electron Cloud The electron cloud represents positions where there is probability of finding an electron. The cloud is less dense where there is less probability of finding an electron. Modern Atomic Theory Element have a characteristic average mass which is unique to that element. Atoms cannot be subdivided, created, or destroyed in ordinary chemical reactions. However, these changes CAN occur in nuclear reactions! All matter is composed of atoms Atoms of any one element differ in properties from atoms of another element