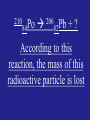* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Document
Molecular orbital wikipedia , lookup
Photoelectric effect wikipedia , lookup
Click chemistry wikipedia , lookup
Nuclear transmutation wikipedia , lookup
Electrolysis of water wikipedia , lookup
Isotopic labeling wikipedia , lookup
Transition state theory wikipedia , lookup
Acid–base reaction wikipedia , lookup
Physical organic chemistry wikipedia , lookup
Elementary particle wikipedia , lookup
Low-energy electron diffraction wikipedia , lookup
Chemical thermodynamics wikipedia , lookup
X-ray fluorescence wikipedia , lookup
Electronegativity wikipedia , lookup
Lewis acid catalysis wikipedia , lookup
X-ray photoelectron spectroscopy wikipedia , lookup
Chemical reaction wikipedia , lookup
Metastable inner-shell molecular state wikipedia , lookup
Photoredox catalysis wikipedia , lookup
IUPAC nomenclature of inorganic chemistry 2005 wikipedia , lookup
Chemistry: A Volatile History wikipedia , lookup
Resonance (chemistry) wikipedia , lookup
Electrochemistry wikipedia , lookup
Matter wave wikipedia , lookup
Wave–particle duality wikipedia , lookup
History of chemistry wikipedia , lookup
Bioorthogonal chemistry wikipedia , lookup
Atomic nucleus wikipedia , lookup
Hypervalent molecule wikipedia , lookup
Extended periodic table wikipedia , lookup
Molecular orbital diagram wikipedia , lookup
Metallic bonding wikipedia , lookup
Atomic orbital wikipedia , lookup
Metalloprotein wikipedia , lookup
Photosynthetic reaction centre wikipedia , lookup
Rutherford backscattering spectrometry wikipedia , lookup
Stoichiometry wikipedia , lookup
Chemical bond wikipedia , lookup
History of molecular theory wikipedia , lookup
Chemical Rx Final Structure of the Atom Stoichiometry Ionic Bonding Nuclear Chemistry Electrons in Atoms 200 200 200 200 200 200 400 400 400 400 400 400 600 600 600 600 600 600 800 800 800 800 800 800 1000 1000 1000 1000 1000 1000 1200 1200 1200 1200 1200 1200 Ions that do not participate in a chemical Rx Daily Double This controls the extent of the reaction The gain of electrons create this The splitting of atomic nuclei A wave’s height This is used to determine if a specific Rx will proceed This will tell you the difference between any type of atom Relationship between any two substances in a balanced chemical Rx The formula of Perclorate ion Half the original amount This form of energy exhibits wavelike behavior as it travels through space A solid produced during a chemical Rx is a solution The smallest particle of an element that retains the properties of the element The most product that can be made from a given reaction This type of ion is formed when valence electrons are removed to make it more stable A particle with no mass, but a negative charge Daily Double When oxygen is added to this reaction, these products always form This was used to discover the electron Aluminum Nitrate + Sulfuric Acid The mole ratio of Sulfuric acid to Nitric acid is When atoms valence electrons can move “freely” between other atoms that are bonded This type of particle makes up visible light This rule states that single electrons w/ the same spin must occupy each equal energy orbital before pairing up Double Replacement Reactions in Aqueous Solutions can produce these 3 products (Hint: Net ionic) This man’s experiment proved the existence of the atomic nucleus In the unbalanced reaction of P4 + O2 P4O10, if 25.0 g of P4 react w/ 50.0 g of O2, what is the limiting Reagent Elements in this group can have multiple charges 210 Po 84 206 Pb + ? 82 According to this reaction, the mass of this radioactive particle is lost This principal states that a maximum of 2 electrons can occupy a single atomic orbital and cannot have the same 4 quantum numbers The balanced products in net ionic when Aluminum Nitrate is mixed with Sulfuric Acid The average of these determines the mass of any atom In the unbalanced reaction of P4 + O2 P4O10, if 25.0 g of P4 react w/ 50.0 g of O2, how much of the excess reagent (g) remains The energy involved in forming an ionic compound (solid) During Beta decay, the atomic number is altered by this Knowing the position and velocity of an electron simultaneously is not possible b/c of this man’s theory This mans’ theory says that matter is composed of extremely small particles called atoms, all atoms of the same element are identical, and atoms cannot be created, divided, or destroyed Probability of finding an electron Spectator Ions Dalton Activity Atomic # Series Limiting Reagent Anion Molar Ratio Perchlorate Theoretical Yield Precipitate Atom CO2 + H2O Cathode Ray tube S, L, G Rutherford’s Gold Foil P4 Isotopes 17.6 g O2 Nothing 3:6 Cation Fission Amplitude Half-life Electromagnetic Radiation Beta Atomic Orbital Sea of Photons Electrons Transition 4 2He Increase Lattice E by 1 Hund’s Rule Pauli Exclusion Heisenberg Stoichiometry During the combustion of Acetone (C3H6O), 4.5 L of water is produced. How much Oxygen in ml is required to produce this much water Density of O = 1.308 g/L Rx: C3H6O + 4O2 3CO2 + 3H2O 4.5 L x 1000 ml/L = 4500 ml 4500 ml x 1.0 g/ml = 4500 g H2O 4500 g H2O x 1 mol/18 g H2O = 250 mol H2O 250 mol H2O x 4 mol O2/3 mol H2O = 333.33 mol O2 333.33 mol O2 x 32 g/1 mol = 11,000 g O2 11,000 g O2 x 1/1.308 g/L x 1000ml/1L = 8.4E6 ml O2