* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Slide 1
Cross-species transmission wikipedia , lookup
Onchocerciasis wikipedia , lookup
Human cytomegalovirus wikipedia , lookup
Influenza A virus wikipedia , lookup
Sexually transmitted infection wikipedia , lookup
Traveler's diarrhea wikipedia , lookup
Gastroenteritis wikipedia , lookup
African trypanosomiasis wikipedia , lookup
Hospital-acquired infection wikipedia , lookup
Eradication of infectious diseases wikipedia , lookup
Oesophagostomum wikipedia , lookup
Typhoid fever wikipedia , lookup
Hepatitis C wikipedia , lookup
Schistosomiasis wikipedia , lookup
Trichinosis wikipedia , lookup
Yellow fever wikipedia , lookup
Herpes simplex virus wikipedia , lookup
Antiviral drug wikipedia , lookup
Middle East respiratory syndrome wikipedia , lookup
Henipavirus wikipedia , lookup
Hepatitis B wikipedia , lookup
West Nile fever wikipedia , lookup
Coccidioidomycosis wikipedia , lookup
Yellow fever in Buenos Aires wikipedia , lookup
Ebola virus disease wikipedia , lookup
Orthohantavirus wikipedia , lookup
Leptospirosis wikipedia , lookup
Rocky Mountain spotted fever wikipedia , lookup
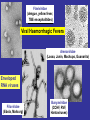
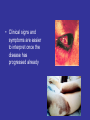
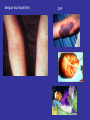
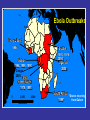
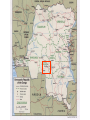
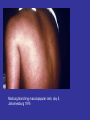
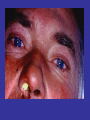
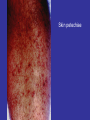
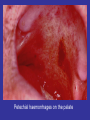
Viral Haemorrhagic Fevers Craig Corcoran NHLS Virology, Groote Schuur Hospital VHF- what is it all about? • VHF’s attract the attention of medical professionals and the general public for a variety of reasons • They are high on the public mind as they are thought of as highly infectious, killing their victims in a dramatic way • Mysteries remain as to the source of some of them “Viral Haemorrhagic Fever” An acute febrile illness characterized by malaise, myalgia, and prostration dominated by generalized abnormalities of vascular permeability, and regulation. Bleeding manifestations often occur, particularly in severe cases; they are usually diffuse and reflect widespread vascular damage rather than lifethreatening volume loss. Flaviviridae (dengue, yellow fever, TBE encephalitides) Viral Haemorrhagic Fevers Arenaviridae (Lassa, Junin, Machupo, Guanarito) Enveloped RNA viruses Filoviridae (Ebola, Marburg) Bunyaviridae (CCHF, RVF, Hantaviruses) These viruses share a number of features: • They are all RNA viruses and are enveloped (i.e covered in a fatty (lipid) coating • Their survival is dependent on an animal or insect host called the natural reservoir • They are geographically restricted to areas where their host species live • Humans are not the natural reservoir for any of these viruses. Humans are infected when they come into contact with infected hosts, and with some viruses, can transmit the virus to one another • Human outbreaks occur sporadically and irregularly. These outbreaks cannot be easily predicted • With few exceptions, there is no cure or established drug treatment for VHFs • VHF and other infectious diseases travel quickly nowadays • Early clinical signs and symptoms may be very discrete and cannot easily be distinguished from those of other illnesses • Clinical signs and symptoms are easier to interpret once the disease has progressed already VHF-clinical picture • • • • • • • • • • Short incubation period Non-specific onset of illness Headache, myalgia, arthralgia Pharyngitis, conjunctival injection/bleed GIT discomfort/disturbances Impaired consciousness Haemorrhages Proteinuria Jaundice Rash, exanthema VHF-differential diagnosis • VHF vs. VHF: – clinical picture-unreliable, epidemiology-approximate, laboratoryproof • VHF vs. bacterial infections – Typhoid, leptospirosis, tick-bite fever, shigellosis, purulent pharyngitis, sepsis (streptococcal, staphylococcal, meningococcal), plague • VHF vs. parasitic diseases – Malaria, african trypanosomiasis, amoebiasis • VHF vs. viral diseases – Viral hepatitis, herpes simplex Dengue fever • Main hosts- non human primates • Human-to-human transmission through Aedes spp. • 2.5 billion individuals at risk • 40-80 million infected each year with thousands of deaths Dengue-clinical features • Fever, headache, back pain , chills, musculoskeletal pain, rash, leucopaenia, thrombocytopaenia • Usually lasts 4-10 days • Dengue haemorrhagic fever/Dengue shock syndrome – Acute vascular hyperpermeability plus abnormal haemostasis – Rapid deterioration after 2-5 days – Scattered petechiae, ecchymoses, easy bruising/bleeding, hepatomegaly, epigastric pain • Pathogenesis: enhancing antibodies- maternal in infants, second infection with a different serotype • Supportive treatment, vaccine in development dengue tourniquet test DHF Yellow Fever • Historic illness stretching back 400 years • yellow: jaundice affecting certain patients • Mosquitos (Aedes and haemogogus) are the true reservoir and vector • Estimated 200 000 cases/year, 30 000 deaths • Symptoms vary from mild to severe with haemorrhagic manifestations Africa and South America only • ‘acute’ phase- fever, headache, muscle pain, GIT disturbance • 15% enter a ‘toxic’ phase and rapidly develop jaundice with bleeding manifestations and renal failure. 50% die within 1014 days • Supportive treatment • Prevention: vaccine- 17D live attenuated, safe and highly effective Filoviruses: Ebola HF • 1976- Simultaneous large outbreaks in Yambuku (Zaire, now DRC) and Nzara/Maridi (Sudan) • Originally thought to be one outbreak • Virology now recognises 2 distinct viruses • EBO-Z: 318 cases; 88% fatal • EBO-S: 284 cases; 53% fatal Ebola Outbreaks 1979, 2004 Ivory Coast 1994 Sudan 1976, 1979, 2004 Gabon 1994, 1996, 1996 Congo 2003 DRC Uganda 2000 (formerly Zaire) 1976, 1995 0 2,000 kilometers 4,000 South Africa 1996* *Doctor returning from Gabon Filoviruses: Marburg HF • 1967: Marburg, Frankfurt & Belgrade – African green monkeys from Uganda • 25 primary • 6 secondary – 1 sexual transmission from husband to wife 85 days after onset of illness, virus cultured from semen • 7 deaths Marburg outbreaks Routes of transmission: filoviruses • Contact with body fluids of an ill patient – HCW and relatives • Infected carcasses (handling/cutting of dead primates) • Needle transfer • Preparation of body for burial • Sexual transmission • Laboratory accident • Aerosol infectivity potential demonstrated experimentally in monkeys (Ebola) Reservoir of infection • Not identified in terrestrial animals or in insects • Non-human primates suffer but are not the reservoir • Association with caves and mines make bats suspects for Marburg • Fruit bats- ? reservoir for Ebola and Marburg (antibodies and RNA found by researchers in Gabon) Filoviruses: clinical presentation • 1-2 week incubation • Abrupt onset fever, headache, myalgia • Non-pruritic papular erythematous eruption becoming large coalescing macules and papules • Palatal petechiae and haemorrhages • GI symptoms, chest pain, delirium • Sever cases- haemorrhages from venipuncture sites, mucous membranes and venipuncture sites • 53-88% case-fatality • ~ 45% hemorrhage • Supportive treatment • Vaccines in development Marburg blanching maculopapular rash, day 5, Johannesburg 1975 Marburg 2005: 335 cases, 283 deaths Arenaviridae • Arenaviruses associated with human disease Virus Origin of Name Year Distribution Lassa Town, Nigeria 1969 West Africa Junin Town, Argentina 1957 South America Machupo River, Bolivia 1962 South America Guanarito Area, Venezuela 1989 South America Sabia Town, Brazil 1990 South America LCMV Clinical disease 1933 Worldwide Lassa: general facts • Viral hemorrhagic fever caused by the Arenavirus Lassa • Transmitted from rodents to humans • Discovered in Nigeria, 1969 • Endemic in portions of West Africa • Seasonal clustering: Late rainy and early dry season • Affects all age groups and both sexes Lassa virus “arenosus” (Latin “sandy”) • Endemic in areas of West Africa, including Nigeria, Liberia, Sierra Leone, and Guinea • Estimated 300,000-500,000 infections/year, with 5000 deaths • Rodent-to-human transmission (the “multimammate rat”, Mastomys species-complex) • Secondary human-to-human transmission with the potential for nosocomial outbreaks with high case-fatality Rodent reservoir Mastomys species complex Lassa: Transmission • Rodent-to-human: – Inhalation of aerosolized virus – Ingestion of food or materials contaminated by infected rodent excreta – Catching and preparing Mastomys as a food source Lassa: Transmission • Human-to-human: – Direct contact with blood, tissues, secretions or excretions of infected humans – Needlestick or cut – Inhalation of aerosolized virus – Sex – Breast feeding Lassa: Clinical Aspects • 80% asymptomatic • Incubation period of 5-21 days • Gradual onset of fever, headache, malaise and other non-specific signs and symptoms • Pharyngitis, myalgias, retro-sternal pain, cough and gastrointestinal symptoms typically seen • A minority present with classic symptoms of bleeding, neck/facial swelling and shock • Case fatality of hospitalized cases: 15-20% • Particularly severe in pregnant women and their offspring • Deafness a common sequela Lassa: Treatment • Supportive measures • Ribavirin – Guanosine nucleoside analog: blocks viral replication by inhibiting IMP dehydrogenase – Licensed for treatment of RSV and HCV – Potential adverse effects: • Dose dependent reversible anemia • Pancreatitis • Teratogen in rodents Crimean-Congo Haemorrhagic Fever CCHF-some background • 1944- Crimean peninsula- Crimean haemorrhagic fever (about 200 cases) • 1956- Belgian Congo- 1 child- Congo Fever • Virus isolated in suckling mice in 1967 • 1-10 cases diagnosed annually in South Africa • Case fatality rate 20-25%, 30-50% without proper medical attention • Mid 1980’s- nosocomial outbreak at TBH- 8 cases, 2 deaths • 27 cases October 1996- Oudtshoorn ostrich abattoir workers Distribution of CCHF virus Distribution of the bont-legged ticks in South Africa •reservoir and vector Hyalomma marginatum rufipes Hyalomma marginatum turanicum Hyalomma truncatum Hyalommas are two host ticks - Lavae and nymphs feed on the first host - Adults feed on the second host -Cattle -Sheep -Goats -Ostriches So when are humans at risk? • Bitten by tick/s or crushed tick/s with bare hands • Direct contact with fresh blood or other tissues of livestock or game animals (ear tagging, castration ect.) • Direct contact with blood, secretions or excretions of a confirmed or suspected CCHF patient including needlestick injuries • Resided in or visited a rural environment where contact with livestock or ticks was possible but a specific incident constituting exposure cannot be identified • NB- incubation period usually 2-7 days hence exposure usually < 7days What are the clinical features? • • • • • • • Sudden onset Fever ≥ 38ºC on at least one occasion Severe headache Myalgia Nausea and/or vomiting Pharyngitis, conjunctivitis Bleeding tendency: petechial rash, ecchymoses, epistaxis, haematemesis, haematuria or melaena Skin petechiae Petechial haemorrhages on the palate Large ecchymoses CCHF- laboratory findings • Leukopaenia or leukocytosis – WCC< 3 x 109/l or ≥ 9 x 109/l • Thrombocytopaenia – Platelet < 150 x 109/l – Usually < 100 x 109/l • Abnormal INR and APTT • Transaminitis – AST ≥ 100iu/l – ALT ≥ 100iu/l CCHF-differential diagnosis • Malaria, tick bite fever, disseminated HSV, viral hepatitis, typhoid, rift valley fever, anthrax, brucellosis, Q fever… • History of exposure, incubation period following exposure, signs and symptoms, laboratory findings CCHF : viral/antibody kinetics IgM IgG viremia 0 5 10 RT-PCR 16 Viral isolation ELISA IgM IgG IgM duration: 2-3 months up to 6 months… IFA CCHF : laboratory diagnosis NICD, Johannesburg, BSL-4 (3) Viral detection: (blood specimen) – RT-PCR (nested) – Cell culture (Vero E6 cells) – Innoculation of newborn mice Antibody detection : (serum sample) - IFA - ELISA - NT Specific management Isolation and barrier nursing Supportive: monitoring of vital functions blood, fluid replacement treatment of DIC Specific: Ribavirin ?? Immune plasma PREVENTION OF CCHF • Ticks most active during Dec, Jan, Feb, Marchavoid hiking/camping • DEET repellents for skin • Permethrin repellents for clothing – (0.5% permethrin should be applied to clothing ONLY) • Check for and remove ticks at least twice daily. • If a tick attaches, do not injure or rupture the tick. Remove ticks by grasping mouthparts at the skin surface using forceps and apply steady traction. PREVENTION OF CCHF • Persons working with livestock- wear gloves and other protective clothing to prevent skin contact with infected tissue or blood • Quarantine and treatment with an ascaricide prior to slaughter (ostriches) Infection Control Handling laboratory specimens from patients with suspected or confirmed VHF-non viral diagnostic specimens • Common sense- know the risks • Blood and other specimens are highly infectious • Risk of transmission through skin/mucous membrane contact and needle stick injuries. ?? Respiratory transmission but avoid aerosolisation of specimens • Limit laboratory testing to what is strictly necessary and where possible run specimens at a time when there is minimal disruption to routine work • Useful to for two techs to work together- one to process the specimen, other to operate the instrument • Protective clothing: disposable gown, 2 pairs of gloves, mask and eye protection • Centrifuge with closed buckets and decontaminate after use • Open buckets, specimens and load instrument racks in a BSL-2 cabinet • Discard residual sample and sampling containers into 2% glutaraldehyde or sodium hypochlorite • Decontaminate instruments according to manufacturers instructions • Clean BSL-2 cabinet with glutaraldehyde or sodium hypochlorite • Discard protective clothing, gloves, specimens, ect. Into a biohazard labelled autoclave bag. Double bag and send for autoclaving • Haematology: – prepare slides in a BSL-2 cabinet, once fixed regard as non-infectious – Regard air dried slides as infectious, decontaminate microscope after use • Microbiology: – protective clothing, process specimens in BSL-2 cabinet, discard residual specimen into 2% glutaraldehyde or sodium hypochlorite – Process positive blood cultures in a BSL-2 cabinet – Referral of specimens- appropriate packaging, inform receiving laboratory • Virology: – Routine specimens- as above – VHF diagnosis- requires BSL 3-4 laboratory BSL-2 cabinet ? • provide personnel, environmental and product protection • Approx 30% air exhausted, 70% recirculated A. front opening B. sash C. exhaust HEPA filter D. rear plenum E. supply HEPA filter BSL-4 laboratory? • dangerous and exotic agents that pose a high individual risk of aerosol-transmitted laboratory infections and life-threatening disease • special engineering and design features to prevent microorganisms from being disseminated into the environment. • Activities are confined to Class III biological safety cabinets, or Class II biological safety cabinets used with one-piece positive pressure personnel suits ventilated by a life support system. Laboratory safety: BSL4 In contrast to patient-care, high-level protection required for: • Laboratory manipulation • Mechanical generation of aerosols • Concentrated infectious material • Viral culture THANK YOU…