* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download (+1) + - Edublogs
Flux (metallurgy) wikipedia , lookup
Multi-state modeling of biomolecules wikipedia , lookup
Marcus theory wikipedia , lookup
Chemistry: A Volatile History wikipedia , lookup
Transition state theory wikipedia , lookup
Metallic bonding wikipedia , lookup
Inorganic chemistry wikipedia , lookup
Electron configuration wikipedia , lookup
IUPAC nomenclature of inorganic chemistry 2005 wikipedia , lookup
Bioorthogonal chemistry wikipedia , lookup
Gaseous detection device wikipedia , lookup
Mössbauer spectroscopy wikipedia , lookup
Click chemistry wikipedia , lookup
Stoichiometry wikipedia , lookup
Lewis acid catalysis wikipedia , lookup
Antioxidant wikipedia , lookup
Atomic theory wikipedia , lookup
Geochemistry wikipedia , lookup
Total organic carbon wikipedia , lookup
Hydrogen-bond catalysis wikipedia , lookup
Oxidative phosphorylation wikipedia , lookup
Artificial photosynthesis wikipedia , lookup
Photosynthetic reaction centre wikipedia , lookup
Chemical reaction wikipedia , lookup
Electrolysis of water wikipedia , lookup
Microbial metabolism wikipedia , lookup
Water splitting wikipedia , lookup
Strychnine total synthesis wikipedia , lookup
Photoredox catalysis wikipedia , lookup
Extended periodic table wikipedia , lookup
Oxidation state wikipedia , lookup
Electrochemistry wikipedia , lookup
Metalloprotein wikipedia , lookup
Evolution of metal ions in biological systems wikipedia , lookup
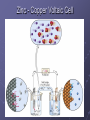
Oxidation-Reduction Reactions Oxidation Numbers Oxidation Numbers and Nomenclature Identifying Oxidation-Reduction Reactions Writing Equations for Oxidation-Reduction Reactions Oxidation-Reduction Titrations Oxidation by Oxygen Oxidation Numbers Definition: An oxidation number is a number that reflects the electrons gained, lost, or shared when an element reacts. Remember that when you lose electrons you become more positive and when you gain electrons you become more negative. Rules for Assigning Oxidation Numbers 1. The oxidation # of any uncombined element is zero. 2. The oxidation # of a monoatomic ion equals its’ charge. 3. The more electrongative element in a binary compound is assigned the number equal to the charge it would have if it were an ion. 4. The oxidation number of fluorine is always -1. 5. Oxygen has an oxidation number of -2. 6. Hydrogen has an oxidation number of +1. 7. In compounds, Group 1 and 2 elements and aluminum have oxidation numbers of +1, +2, and +3 respectively. 8. The sum of the oxidation numbers of atoms in a compound is zero. 9. The sum of the oxidation # in a polyattomic ion is equal to the charge of the ion. Oxidation number or state When dealing with simple ions, this is easy to determine. It is simply the charge on the ion. Examples Group IA (1) Group IIA (2) Group VIIA (17) +1 +2 -1 Oxygen -2 usually Hydrogen +1 if bonded to nonmetal Hydrogen -1 if bonded to metal Oxidation number For elements in their elemental state, the oxidation number is also pretty straightforward. Since all of the atoms are the same, the electrons are shared equally so the oxidation number is zero. Examples The atoms in N2, Na, P4, H2 and O2 all have oxidation numbers of zero. Oxidation numbers With polar covalent bonds electrons are shared but not equally. For electrons that are shared in these compounds, we assign the shared electrons to the most electronegative element. We are just acting as though the electronegativity difference was large enough for the transfer of electrons to occur. Example Assign the oxidation states for all elements in water. The electronegativities are: H = 2.2, O = 3.5 The electrons from both hydrogen are assigned to the oxygen. Oxidation numbers: O = -2 H = +1 Oxidation numbers Many elements have more than one possible oxidation number. Often, it is possible to determine the oxidation number of those elements in a compound simply by looking at what you do know. Follow the previous rules and then assign an oxidation number that insures that the overall compound has no net charge. Example Find the oxidation state for all elements in: HNO3 Hydrogen we know - it must be +1 Oxygen should be -2 in this case. What about nitrogen? Example See what you do know and find the difference. We know that H is assigned a value of +1 HNO3 Oxygen is -2 and we have 3 of them = -6 OK, so what's left over?? (+1) + (-6) + charge on nitrogen = 0 Nitrogen must have a value of +5 Determining Oxidation Numbers SO2 NO3- PCl5 SO42- H2O NH4+ H2SO3 MnO4- B Li Be +1 +2 H +3 He C N +4 +5 +4 -2 +3 +2 -4 +1 -3 O F -1 -2 -1 S Cl Ne +1 Al Na Mg +1 +3 +2 K Ca Sc +1 +2 Rb Sr +1 +2 +2 Y Mo Tc Ru Rh Pd Nb Zr +6 +7 +8 +6 +4 +4 Ag Cd +3 +3 +2 +3 +4 +5 +4 Hf Ta +4 +5 +4 -4 P +5 +6 +4 +7 +5 +3 +2 +3 +1 -3 -2 -1 Cr Mn Fe Co As Se Br Cu Ge Ni Zn Ga +6 +7 +6 5+ 6+ +5 3+ Fr Ra Lr +1 V +4 +5 +4 +3 +3 +3 +3 +4 +3 +2 +2 +2 +2 +2 Cs Ba Lu +1 Ti Si +4 +3 +6 +4 +3 +2 +2 +2 +1 In +4 -4 3+ 3- +3 +4 +2 +5 +3 -3 +2 W Re Os Ir Pt Au Hg Tl Pb Bi +6 +4 +4 +3 +4 +2 +3 +1 +4 +2 +5 +3 +3 +1 +2 +1 4+ 2- +1 -1 Sn Sb Te +3 +2 +8 +6 +2 +3 +4 +3 +7 +6 +4 +1 +2 I Ar Kr +4 +2 Xe +6 +7 +5 +6 +4 +1 +4 -2 -1 +2 Po +2 At Rn Common oxidation numbers -1 Oxidation numbers and the periodic table Some observed trends in compounds. Metals have positive oxidation numbers. Transition metals typically have more than one oxidation number. Nonmetals and semimetals have both positive and negative oxidation numbers. No element exists in a compound with an oxidation number greater than +8. Oxidation number and nomenclature Stock system For metals with several possible oxidation numbers, use Roman numeral in the name. FeSO4 Fe2(SO4)3 Cu2O CuO PbCl2 PbCl4 iron(II) sulfate iron (III) sulfate copper(I) oxide copper(II) oxide lead(II) chloride lead(IV) chloride Identifying oxidation-reduction reactions. Oxidation-Reduction - REDOX A chemical reaction where there is a net change in the oxidation number of one or more species. Both an oxidation and a reduction must occur during the reaction. Mg (s) + Cl2 (g) MgCl2 (s) Here the oxidation number of Mg has changed from zero to +2. Cl has changed from zero to -1. REDOX reactions Oxidation An increase in oxidation number. Reduction A decrease in oxidation number. If the oxidation number of any element changes in the course of a reaction, the reaction is oxidation-reduction. Example. 2 Fe(NO3)3 (aq) + Zn(s) Zn(NO3)2 (aq) 2 Fe(NO3)2 (aq) + Redox Reaction Fe(s) + O2(g) Fe2O3(s) Iron is oxidized (0 to +3) and O2 is reduced (0 to -2) Half Reactions Fe Fe3+ + 3e- (oxidation) O2 + 4e- 2 O2- (reduction) Half reactions Example. Half-reactions can be of the ‘net ionic’ form. Balance the follow Fe3+ + Zn (s) Fe2+ + Zn2+ 2 ( Fe3+ + e- Zn(s) Fe2+) (reduction) Zn2+ + 2e2Fe3+ + Zn (s) (oxidation) 2Fe2+ + Zn2+ Example +3 0 2Fe(NO3)3 (aq) + Zn(s) +2 +2 2Fe(NO3)2 (aq) Zn(NO3)2 (aq) Fe3+ is reduced to Fe2+ Zn is oxidized to Zn2+ NO3- is a spectator ion. Zinc - Copper Voltaic Cell Zn (s) Zn 2 (aq) 2 (aq) Cu Cu(s) A Zinc-Copper Voltaic Cell Zn electrode Cu electrode Anode (-) Cathode (+) Oxidation Reduction Half Reaction: Half Reaction: Zn Zn2+ + 2 e- Cu2+ + 2 e- Cu Voltaic Cell cont… In a voltaic cell the redox reaction is spontaneous and produces an electric current. This current can be measured using a voltmeter. Standard Electrode Potentials • A standard electrode potential, Eo, is based on the tendency for reduction to occur at the electrode. • The cell voltage, called the standard cell potential (Eocell), is the difference between the standard potential of the cathode and that of the anode. Eocell = Eo (cathode) – Eo (anode) Standard Electrode Potentials Balancing REDOX equations Many REDOX equations can be balanced by inspection. H2S (g) + H2O2 (aq) S (s) + 2 H2O (l) However, others are more difficult. 2KMnO4 (aq) + H2O2 (l) + 3H2SO4 (aq) 2MnSO4 (aq) + K2SO4 (aq) + 3O2 (g) + 4H2O (l) Balancing REDOX equations Half-Reaction method. With this approach, the reaction is broken into two parts. Oxidation half-reaction. The portion of the reaction where electrons are lost. A An+ + ne- Reduction half-reaction. The portion of the reaction where electrons are gained. Balancing REDOX equations The goal is then to make sure that the same number of electrons are being produced and consumed. (m) ( A (n) (me- + B nB + mA An+ + ne- ) Bm- ) mAn+ + nBm+ When properly balanced, the electrons Half reactions Another Example Determine the balanced equation for the reaction of Fe2+ with Cr2O72- in an acidic solution. Fe2+ + Cr2O72- H+ Fe3+ + Cr3+ The two half-reactions would be: Fe2+ Fe3+ Half reactions First, balance each half-reaction for all elements except hydrogen and oxygen. Fe2+ Fe3+ Cr2O72- 2Cr3+ Next, balance each half-reaction with respect to oxygen by adding an appropriate number of H2O. Fe2+ Fe3+ Half reactions Remember that this reaction occurs in an acid solution so we can add H+ as needed. Fe2+ Fe3+ 14H+ + Cr2O72- 2Cr3+ + 7H2O Now we need to know how many electrons are produced or consumed and place them in our half-reactions. Half-reactions Fe2+ Fe3+ + e- 6e- + 14 H+ + Cr2O72- 2Cr3+ + 7H2O We need the same number of electrons produced and consumed so: 6Fe2+ 6Fe3+ + 6e6e- + 14H+ + Cr2O72- 2Cr3+ + 7H2O As our final step, we need to combine Half-reactions 6Fe2+ + 14H+ + Cr2O726Fe3+ + 2Cr3+ + 7H2O In this reaction, Fe2+ is oxidized and the dichromate ion is reduced. This reaction is used for the determination of iron by titration. Disproportionation reactions In some reactions, the same species is both oxidized and reduced. Examples 2H2O2 (l) 2H2O (l) + O2 (g) 3Br2 (aq) +6OH- (aq) +3H2O(l) BrO3-(aq) +5Br-(aq) For this to occur, the species must be Oxidation-reduction titrations REDOX reactions can also serve as the basis for titrations. For example, we can determine the amount of iron in an ore by titration. Initially, we must dissolve the sample. This results in both iron(II) and iron(III) being produced in solution. Sample preparation One option is to use a reductor. You slowly wash your sample through the column with water. Jones Reductor Zn(Hg) Zn2+ + Hg(l) + 2e An amalgam is used to prevent Zn + 2H+reductorZn2+ + H2(g) For iron, we get Titrants Now that all of our iron is in a single oxidation state, we’re ready to do a titration. We need an oxidizing agent to convert all the iron from Fe2+ to Fe3+. Primary standard A material that is available in pure form. Common titrants Oxidizing titrants Dichromate - Cr2O72 The potassium salt is a primary standard material. Very stable solutions. If air is kept out, it can last for years. It is a very strong oxidizing agent. Need an indicator such as diphenylamine sulfonic acid. Common titrants Oxidizing titrants Permanganate - MnO4 The potassium salt is the most commonly used. It is not a primary standard. Solutions must be standardized - typically use Na2C2O4 ( a primary standard material.) Reagent slowly degrades and MnO2 must be removed No indicator is needed - excess reagent produces a pink solution. Common titrants The standardization of MnO42- with oxalate involves the following reaction. 2MnO42-(aq) + 5C2O42-(aq) + 8H+(aq) 2Mn2+(aq) + 10CO2 (g) + 8H2O (l) Since MnO42- in an intense Oxidation by oxygen Oxygen is not the strongest of oxidizing agents but it is about 19% of our atmosphere. It is able to react with all other elements except: noble gases halogens noble metals like gold Combustion Examples CH4(g) 2H2O(g) + 2O2(g) CO2(g) + S(s) + O2(g) SO2(g) N2(g) + O2(g) 2NO(g) + O2 (g) 2 NO(g) 2 NO2(g)