* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Chapter-2-Human-Chemistry
Molecular orbital diagram wikipedia , lookup
Metallic bonding wikipedia , lookup
Resonance (chemistry) wikipedia , lookup
Hydrogen-bond catalysis wikipedia , lookup
Acid–base reaction wikipedia , lookup
Nuclear fusion wikipedia , lookup
Electrolysis of water wikipedia , lookup
Click chemistry wikipedia , lookup
Atomic nucleus wikipedia , lookup
X-ray photoelectron spectroscopy wikipedia , lookup
Bioorthogonal chemistry wikipedia , lookup
Electrochemistry wikipedia , lookup
Photosynthetic reaction centre wikipedia , lookup
Molecular dynamics wikipedia , lookup
Fatty acid metabolism wikipedia , lookup
Lewis acid catalysis wikipedia , lookup
Physical organic chemistry wikipedia , lookup
Hypervalent molecule wikipedia , lookup
Marcus theory wikipedia , lookup
Rutherford backscattering spectrometry wikipedia , lookup
Chemical reaction wikipedia , lookup
Chemical bond wikipedia , lookup
Basal metabolic rate wikipedia , lookup
Transition state theory wikipedia , lookup
Atomic theory wikipedia , lookup
Carbohydrate wikipedia , lookup
Enzyme catalysis wikipedia , lookup
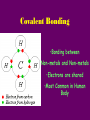
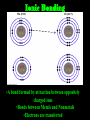
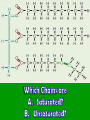
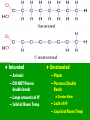
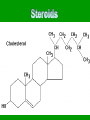
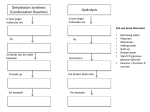
Energy cannot be created nor destroyed but transferred Human Kinetic Energy Muscular Contractions Peristalsis Heart Beat & Blood Movement Mastication Human Potential Energy Muscules (Rest) Heart Beat (Millisecond) Energy Forms 1. Chemical – Energy stored or released in chemical bonds 1. Food 2. Monosaccharides (candy bars) 2. Electrical – Transfer due to movement of charged particles 1. Conductivity of body (75% Water) 2. Nerve Impulses 3. Mechanical – Directly involved in moving matter 1. Muscle contraction (running, throwing) 2. Walking 3. Eating 4. Radiant – Energy that travels in waves – – X-rays, UV rays, etc. Body Heat The Charge of an Atom Element Configuration • Atomic Number - The number of protons in the nucleus of an atom • Atomic Mass Number - Total number of protons and neutrons # of Neutrons = Atomic # Symbol 6 C Atomic Mass # 12.011 Atomic Mass # - Atomic # Covalent Bonding •Bonding between Non-metals and Non-metals •Electrons are shared •Most Common in Human Body Ionic Bonding •A bond formed by attraction between oppositely charged ions •Bonds between Metals and Nonmetals •Electrons are transferred The sum of all the body’s chemical reactions Chemical breakdown of particles Decomposition reaction ~ Exergonic (Energy Released) AB A + B Chemical combining of particles Synthesis reaction ~ Endergonic (Energy Required) A + B AB Chemical Reactions Water’s Importance • Chemical Reactions – Takes place in intracellular reactions, protein production reactions • High Heat Capacity – Homeostasis maintenance • Heat of Evaporation – Temps must be very high for water to evaporate – Cooling mechanism for the skin • Lubricant – Linings of mucous membranes (Respiratory), joints (Skeletal), and Digestion (Digestive) • Solvent – Polar Covalent bonds are so strong that it easily dissociates compounds such as NaCl Importance of polarity and Ionic Bonding? • Polarity – A molecule with distinct positively and negatively charged sides Polar Covalent Forces of attraction (chemical bonds) influence whether water will dissolve certain compounds Myth: “You can NEVER drink enough Water.” Why wont people listen to Mr. Jeffrey??????? Salts in the Body KCl NaCl KBr KI • Formed from acids and bases reacting with one another • Dissociates itself into cations and anions when dissolved in water • Important for tissue strength, blood, and lymph HCl + KOH KCl + H2O KCl + Water K+ + Cl- Electrolytes • A compound that dissociates into positive and negative ions • Drinks such as Gatorade replenish body with ions to balance body’s homeostasis and diminish anaerobic respiration and Lactic Acid development Reactions occur when making or breaking chemical bonds 1. All reactions require an activation energy – Energy needed to disrupt and rearrange the stable electron configuration 2.Concentration and Temperature are main factors in influencing chemical reactions • Higher Concentration = Higher chance of particles colliding and causing reactions • Higher Temperatures = Higher Kinetic Energy of molecules = Higher Chances of Particle Collisions Acidity & Alkalinity • Acids & Bases – Acid dissociates itself into H+ ions and one or more anions pH 0-6.9 – Bases dissociates itself into OH- ions and one or more cations 7-14 • Buffers – Homeostasis !!!!!!!!! – Different mechanisms of body have different pH • Carbonic Acid in Blood • Sodium Bicarbonate in Pancreas Reversible Reaction Energy “Currency” – Body’s energy carrier Supplier of energy for many of body’s reactions ATP ADP + P + Energy Breakdown of glucose during cellular respiration Carbohydrates Lipids Proteins Carbohydrates Polysaccharides Monosaccharides Disaccharides Formed from Synthesis reaction of two monosaccharides Building Blocks 3 or more chains of monosaccharides Glycogen Glucose Lactose Candy, Simple Sugars Fructose Sucrose Found in animals Cellulose Ribose Found in Plants Fruit DNA & RNA Glucose + Fructose = Table Sugar Glucose + Galactose = Milk Sugar Carbohydrate Structures Lipids 1. Triglycerides – – – 2. Made up of glycerol & Fatty Acids (Figure: Next Slide) Stored in adipose tissue of animals More Energy provided than that of Proteins and Carbohydrates Phospholipids – – Compose cell membranes Hydrophilic & Hydrophobic Heads • Saturated – Animals – DO NOT Possess double bonds – Large amounts of H+ – Solid at Room Temp • Unsaturated – Plants – Possesses Double Bonds • Creates Kinks – Lack of H+ – Liquid at Room Temp 1. 2. Steroids – Cholesterol – Precursor to Aldosterone which regulates sodium counts in blood and sex hormones – Ananbolics – Increase of Testosterone Creation – Any time human body is supplied with a substance it makes naturally, the process will slow down Other Lipids – Fatty Acids – Catabolized to produce ATP – Vitamin E – Essential for wound healing, antioxidant for nervous system – Vitamin K – Blood Clotting – Carotenes – Photopigments synthesis for retinal maintenance Lipids Proteins • Functions – Structure • Nails, Hair – Keratin • Actin & Myosin – Muscles Tissue • Collagen - Skin – Hormone Regulator • Insulin in blood glucose levels – Immune System • Protects against foreign invaders – Oxygen Transport • Hemoglobin – Enzyme • Amylase (Mouth), Lactase, Lipase • Structure (cont’d) – Always contain C, H, N, O – Amino Acids – building blocks of proteins (20 total) – Peptide bonds are covalent bonds that connect amino acid chains in dehydration synthesis reaction Active Site: Lock and Key Mechanism ~ One enzyme for one active site Induced fit: Active site changes once substrate enters active site End in suffix -ase