* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Urea
Clinical neurochemistry wikipedia , lookup
Gaseous signaling molecules wikipedia , lookup
Point mutation wikipedia , lookup
Peptide synthesis wikipedia , lookup
Fatty acid synthesis wikipedia , lookup
Metalloprotein wikipedia , lookup
Wilson's disease wikipedia , lookup
Nitrogen cycle wikipedia , lookup
Protein structure prediction wikipedia , lookup
Citric acid cycle wikipedia , lookup
Genetic code wikipedia , lookup
Glyceroneogenesis wikipedia , lookup
Proteolysis wikipedia , lookup
Fatty acid metabolism wikipedia , lookup
Biosynthesis wikipedia , lookup
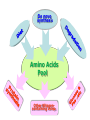
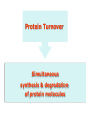
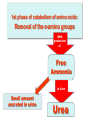
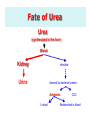
Amino Acids Metabolism: Disposal of Nitrogen No Storage of Amino Acids in the body • So, amino acids must be obtained from 1. Diet 2. De novo synthesis (of non-essential aa) 3. Degradation of protein (normal turnover) De novo synthesis Amino Acids Pool Other Nitogencontaining comp. Protein Turnover Simultaneous synthesis & degradation of protein molecules Protein turnover Most proteins in the body are constantly being synthesized & then degraded, permitting the removal of abnormal or unneeded proteins Protein Degradation By Two Major Enzyme Systems 1- Ubiquitin-proteasome mechanism • Energy-dependent • Mainly for endogenous proteins (proteins synthesized within the cell) 2- Lysosomes • Non-energy-dependent • Primarily for extracellular proteins as: - plasma proteins that are taken into cells by endocytosis - cell surface membrane proteins: for receptor-mediated endocytosis Amino Acids Catabolism Amino Acids Catabolism - Overview • Unlike glucose and fatty acids, amino acids are not stored by the body • Amino acids in excess of biosynthetic needs are degraded. • Degradation of amino acids involves: First Stage Removal of α-amino group Ammonia (NH3) Second Stage Remaining carbon skeleton Energy metabolism 1st phase of catabolism of amino acids: Removal of the α-amino groups With production of Free Ammonia In Liver Small amount excreted in urine Urea Amino Acids Catabolism - Overview • Ammonia is produced by all tissues from the catabolism of amino acids • Ammonia is mainly disposed is via formation of urea in liver • Blood level of ammoina must be kept very low, otherwise, hyperammonemia & CNS toxicity will occur • To solve this problem, ammonia is transported from peripheral tissues to liver via formation of: Glutamine (most tissues) Alanine (muscle) 2nd phase of A. A. catabolism Carbon skeletons of the α-ketoacids are converted to common intermediates of energy producing, metabolic pathways • ATP, CO2 & H2O (by Citric acid cycle) • Glucose (by gluconeogenesis) • Fatty Acids (from acetyl CoA) • Ketone Bodies (from acetyl CoA) Amino Acids Metabolism Removal of Nitrogen from Amino Acids Removing the a-amino group • Essential for producing energy from any amino acid • An obligatory step for the catabolism of all amino acids Deamination Pathways Amino group (nitrogen) is removed from an amino acid by either 1- Transamination : by transaminases 2- Oxidative Deamination: by glutamate dehydrogenase 1- Transamination ALL Amino Acids (except lysine & threonine) a-ketoglutarate accepts the amino group from amino acids to become glutamate by: Transaminases (aminotransferases) Transaminase Glutamate: Glutamate dehydrogenase Ammonia Energy, glucose, FAs or KB 2- Oxidative deamination by Glutamate Dehydrogenase Glutamate (from transamination steps) by enzyme Glutamate Dehydrogenase Ammonia a-ketoglutarate Urea Cycle Urea used for transamination of further amino acids Diagnostic Value of Plasma Aminotransferases • Aminotransferases are normally intracellular enzymes • Plasma contains low levels of aminotransferases representing release of cellular contents during normal cell turnover • Elevated plasma levels of aminotransferases indicate damage to cells rich in these enzymes (as physical trauma or disease to tissue) • Plasma AST & ALT are of particular diagnostic value Diagnostic Value of Plasma Aminotransferases 1- liver disease: Plasma ALT & AST are elevated in nearly all liver diseases but, particularly high in conditions that cause cell necrosis as: viral hepatitis toxic injury prolonged circulatory collapse ALT is more specific for liver disease than AST AST is more sensitive (as liver contains a large amount of AST) 2- Nonhepatic disease: as: Myocardial infarction Skeletal muscle disorders These disorders can be distinguished clinically from liver disease Metabolism of Ammonia • Ammonia is produced by all tissues during metabolism of a variety of compounds • Ammonia is disposed of primarily by formation of urea in the liver • The level of ammonia in blood must be kept very low • Slightly elevated concentrations (hyperammonemia) are toxic to CNS So, There must be a mechanism by which Ammonia is moved from peripheral tissues to the liver for disposal as urea While at the same time Ammonia must be maintained at low levels in blood Disposal of Ammonia 1- Urea in the liver • is quantitatively the most important disposal route for ammonia • Urea is formed in the liver from ammonia (urea cycle) • Urea travels in the blood from the liver to the kidneys where it is filtered to appear in urine Disposal of Ammonia cont. 2- Glutamine in most peripheral tissues especially brain, sk.ms. & liver • In most peripheral tissues, glutamate binds with ammonia by action of glutamine synthase • in the brain, it is the major mechanism of removal of ammonia from the brain • This structure provides a nontoxic storage & transport form of ammonia • Glutamine is transported to blood to other organs esp. liver & kidneys • In the liver & Kidney, glutamine is converted to ammonia & glutamate by the enzyme glutaminase. Disposal of Ammonia 3- cont. Alanine in skeletal muscles • Ammonia + Pyruvate form alanine in skeletal muscles • Alanine is transported in blood to liver • In liver, alanine is converted to pyruvate & ammonia • Pyruvate can be converted to glucose (by gluconeogenesis) • Glucose can enter the blood to be used by skeletal muscles (GLUCOSE - ALANINE PATHWAY) Disposal of Ammonia Glutamine in Most Tissues Esp. brain & Kidneys Urea in Liver Alanine in Skeletal Muscles cont. Urea Cycle • Urea is produced in the liver • From the liver, it is transported in the blood to the kidneys for excretion in urine Urea is composed of: Two nitrogen atoms • First nitrogen atom is from free ammonia • Second nitrogen atom is from aspartate Carbon & oxygen atoms are from CO2 Reactions of the Urea Cycle • First two reactions occur in the mitochondria • Remaining reactions occur in the cytosol Ammonia + Aspartate + CO2 + 3 ATP UREA + Fumarate + 2 ADP + AMP + 2 Pi + PPi + 3 H20 • Synthesis of urea is irreversible • 4 high-energy phosphates are consumed for synthesis of one molecule of urea Overview of Urea Cycle Fate of Urea Urea (synthesized in the liver) Blood Kidney intestine Urine cleaved by bacterial urease Ammonia In stool CO2 Reabsorbed in blood Hyperammonemia = Increase of ammonia level of blood • Blood Ammonia • Normal level of blood ammonia is 5-50 mmol/L • Hyperammonemia A medical emergency as ammonia has a direct neurotoxic effect on CNS • Ammonia intoxication: • • It is defined as toxicity of the brain due to increase in ammonia level in the systemic blood. This increased ammonia will be directed to α ketoglutarate to form glutamic acid then glutamine leading to interference with citric acid cycle so decrease ATP production in the brain cells. Clinical manifestations: Tremors, slurring of speech, somnolence, vomiting, cerebral edema & blurring of vision At high concentrations, ammonia can cause coma & death Types of Hyperammonemia 1- Acquired Hyperammonemia 1- Liver diseases: are common causes in adults 1- Acute causes: viral hepatitis, ischemia, hepatotoxins 2- Chronic causes: liver cirrhosis due to alcoholism, hepatitis, biliary obstruction…etc may result in the formation of collateral circulation around the liver So, portal blood is shunted directly into systemic circulation & detoxication of ammonia to urea is markedly impaired 2- Gatrointestinal Bleeding By action of bacteria of GIT on blood urea with production of big amounts of ammonia that is absorbed to blood. Types of Hyperammonemia cont. 1- Hereditary Hyperammonemia Genetic deficiencies can occur for each of the five enzymes of the urea cycle (overall prevalence 1:300,000 live births) Ornithine transcarbamoylase deficiency • X-linked • Most common deficiency among all 5 enzymes • Males are predominantly affected • Females carriers are clinically affected All other urea cycle disorders are autosomal recessive • • In each case, failure to synthesize urea leads to hyperammonemia during the first weeks following birth All inherited disorders of the urea cycle enzymes result in mental retardation Treatment of Hyperammonemia • Limiting protein in diet • Administration of compounds that bind covalently to amino acids To produce nitrogen-containing molecules that are excreted in the urine for example: Phenylbutyrate given orally converted to phenylacetate that condenses with glutamine to form phenylacetylglutamine which is excreted in urine Hyperammonemia in Renal Failure Rena Failure blood urea levels are elevated Transfer of urea to intestine is increased Much amounts of Ammonia is formed by bacterial urease Absorbed to blood Hyperammonemia • To reduce hyperammonemia: Oral neomycin reduces the amount of intestinal bacteria responsible for ammonia production