* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Organic
Metalloprotein wikipedia , lookup
Fatty acid synthesis wikipedia , lookup
Two-hybrid screening wikipedia , lookup
Basal metabolic rate wikipedia , lookup
Gene expression wikipedia , lookup
Citric acid cycle wikipedia , lookup
Artificial gene synthesis wikipedia , lookup
Vectors in gene therapy wikipedia , lookup
Peptide synthesis wikipedia , lookup
Deoxyribozyme wikipedia , lookup
Protein structure prediction wikipedia , lookup
Point mutation wikipedia , lookup
Phosphorylation wikipedia , lookup
Blood sugar level wikipedia , lookup
Amino acid synthesis wikipedia , lookup
Fatty acid metabolism wikipedia , lookup
Genetic code wikipedia , lookup
Proteolysis wikipedia , lookup
Nucleic acid analogue wikipedia , lookup
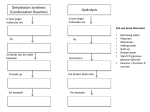
BIOCHEMISTRY “All life is chemistry.” Jan Baptist van Helmont (1648) • Introduction – Defn = chemistry of living organisms – 98% is C, H, O (99% is CHNOPS) – Organic chemistry deals mainly with ________. – Why is carbon so versatile? Draw its Lewis Dot structure. How many atoms of each element a human baby has. KEY for Model Building • • • • Black = ? White = ? Blue = ? Red = ? • What do the white tubes represent? Try making methane = CH4 It’s 3-D shape is what you built. Is it polar or Nonpolar? Try building carbon dioxide=CO2 Is it polar or nonpolar? Functional group, -COOH (aka, carboxylic acid) Why is it called an “acid?” Organic Polymers that Life Requires The BIG 4 Biomolecules 1. 2. 3. 4. Carbohydrates Lipids/Fats Proteins Nucleic Acids (DNA & RNA) All made up of C,H, O, N 1. Carbohydrates • Include mono- & polysaccharides • Examples: glucose, fructose, sucrose, deoxyribose, amylose • Named “carbo” “hydrates” b/c … each carbon is hydrated w/ H- and –OH. • Formula = Cn(H2O)n where n = whole number so carbs have 1C:2H:1O ratio. A. Monosaccharides • Building blocks of polysaccharides • Means “single sugars” • Examples include glucose, fructose, galactose (all C6H12O6)>>isomers=same chemical make-up, different arrangement Glucose Why is glucose so important? It is the energy molecule made by photosynthesis! B. Disaccharides • “double sugars” • 2 monosacs joined together via dehydration synthesis reaction + H0 2 • examples: glucose + glucose = maltose + water (see above) glucose + fructose = sucrose (table sugar) + H20 galactose + glucose = lactose (milk sugar) + H20 C. Polysaccharides • “many sugar” polymer made up of 200+ monosacs (“bricks”) to make a macromolecule “house” • Usually long chains of glucoses joined together via dehydration synthesis Examples of Polysaccharides • I’ll provide 1 structural & 1 energy store for both plants & animals • Made out of the same “brick”=glucose • How those glucoses are arranged is what makes them different (structurally and functionally) • Can you see how in the following 4 examples? #1= -What monomer is repeated in this chain? -What do you notice about how they are positioned in the long chain? -What kind of reaction occurred to join all of them together? Cellulose=structural polysac made up of αglucoses joined 14 alternating orientation found in plant cell walls (tough& fibrous). Can we humans digest it? • Humans cannot digest cellulose (but it is a source of good fiber) • Neither can cows nor termites but their microbial partners in their guts can #2=STARCH (aka, Amylose) made up of chains of glucoses w/ 14 rightside up linkages How are these glucoses joined differently than those in cellulose? Can we animals digest starch? YES! With Amylase =enzyme found in saliva that digests amylose by breaking bonds betw. glucoses Starch granules in plant cells Photo by David Webb How do animals store carbohydrates? #3=Glycogen (animals’ complex storage carb) Glycogen(use it all & you’ll hit the wall) #4=Chitin (animal structural polysac). What does it resemble? Chitin (continued) • Monomer=glucosamine (amine group at the #2 carbon in the glucose ring) • Found in the exoskeleton cuticle of insects and crustaceans Chitin--found in the exoskeleton of the cicada Chitin is sometimes purified & used in surgical wire Indicator Tests for Carbs 1. Reducing (simple) sugars: Benedict’s Solution made up of a Cupric ion (Cu 2+) which is blue reacts with reducing sugars that donate electrons resulting in Cuprous Oxide (Cu 1+) which is reddish in color. 2. Starch Test • Lugol’s Iodine solution is yelloworangish in color but changes to bluish-black in the presence of the complex carb, starch. 2. Lipids • • Include fats, oils, waxes, & steroids Chemically different group classified together b/c they are insoluable in water (NONPOLAR). A. Triglycerides = 3 fatty acids + glycerol Saturated vs. Unsaturated Fats • So named b/c each carbon is “saturated with hydrogens” • Found mainly in animals vs. plants • Contain only single covalent bonds in long hydrocarbon chain vs. double bonds present in unsat. that lead to “kinks” in the long chain. • Solid vs. liquid at room temp. Importance of Fats • Great source of long-term energy storage (each gram of fat produces 2X as much energy as carbs/protein) • Insulation (esp. needed in first 3 years of brain development of humans to insulate your brain nerve cells) • Lubrication (keeps organs from tearing each other apart) B. Phospholipids=make up the bilayer of the cell membrane OUTSIDE Inside of Cell Membrane INSIDES of a CELL C. Steroids->all made from cholesterol = a set of 4 hydrocarbon rings 1.) Sex Steroids are hormones that use cholesterol as backbone Figure 2: Estradiol 2.) Fat-soluble vitamins like Vitamin D Formed from cholesterol. Important in maintaining strong bones and boosting immune system. Lipid Indicator test: Sudan stain. Sudan IV is not soluble in water; it is, however, soluble in lipids. Red Sudan IV is added to a solution along with ethanol to dissolve any possible lipids. If lipids are present the Sudan IV will stain them reddish-orange, giving a positive test, usu. Forming a layer on top. Sleep-Fat Study • ttp://www.npr.org/blogs/health/2012/10/17/ 163018568/poor-sleep-may-lead-to-toomuch-stored-fat-and-disease 3. PROTEINS • Means “first place” • Monomer = amino acids: central carbon w/ 1. Amino group 2. Carboxylic acid grp. 3. A Hydrogen 4. R group What is an R group? • R = Replacement Group • Could be anything but only 20 that life uses (see p. 367 for a list of them). • Build yours and attach (see index cards). • Gives each amino acid its uniqueness & specificity (dictates how the amino acid interacts w/ others) • Either nonpolar, polar, or charged AA1 + AA2 Dipeptide + H20 Left=remove –OH from carboxyl / Right =remove H from amine. Connect 2 aa with peptide bond + H2O Polypeptide = many amino acids joined together to create long amino acids chain. Smallest human one = gastric peptide (10aa) Largest human protein = TITAN (>27,000 aa) • For each peptide bond formed, a water molecule is lost thru dehydration synthesis. • How that polypeptide folds and takes on a 3-D shape is determined by its R groups and how they interact w/ each other. Roles that Proteins Play 1.) Speed up chemical reactions (catalysts) = ENZYMES Ex: amylase: starts breaking down starch Pepsin: starts breaking down proteins ATP Synthase: helps make ATP 2.) Structural / supportive Ex: collagen in tendons Does this look like anything we’ve seen before? 3.) Transport Ex: hemoglobin in red blood cells moves O2 throughout the body 4.) Communication Hormones = messenger molecules that are secreted one place but have their effect elsewhere (Ex: insulin : secreted from pancreas & tells body cells to remove glucose from bloodstream) Honors + Neurotransmitters (serotonin) insulin 5.) Energy storage—egg whites contain albumin 6.) Defense—antibodies that help immune system destroy invaders 7.) Movement—actin & myosin in muscle fibers 4. Nucleic Acids • Named so because they are found mainly in the nucleus of cells. • Include DNA & RNA. • Monomer = nucleotide A. Structure– the nucleotide is made up of 3 parts 1.) 5-carbon sugar = Deoxyribose or Ribose 2.) Nitrogen-Carbon Ringed Base • DNA has 4 = A, C, G, or T • RNA has 4 = A, C, G, or U 3.) Phosphate Group NUCLEOTIDE: all 3 parts together • See p. 48, Fig 2-16 • See class model -Red pentagon = sugar -Purple = phosphate -Middle colored “stairs” = adenine, thymine, cytosine, guanine B. Function of NAs • The “code of life” • Specifically they code for proteins • Each NA’s role: DNA—stores the info (w/in chromosomes) for all of life’s processes (growth, metabolism, reproduction, etc.) RNA—messenger that carries the info out Nucleic Acid Polymer Structure A. DNA = double helix wound w/in large chromosomes ` B. RNA = single stranded, much shorter nucleotide sequences, comes in many different forms to perform different jobs in the decoding process