* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download 4.1mb
Survey
Document related concepts
Innate immune system wikipedia , lookup
Gluten immunochemistry wikipedia , lookup
Duffy antigen system wikipedia , lookup
Complement system wikipedia , lookup
Adaptive immune system wikipedia , lookup
Immunocontraception wikipedia , lookup
Cancer immunotherapy wikipedia , lookup
Autoimmune encephalitis wikipedia , lookup
Anti-nuclear antibody wikipedia , lookup
Polyclonal B cell response wikipedia , lookup
Molecular mimicry wikipedia , lookup
Transcript
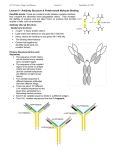
October 5, 2004 IMMUNITY ADAPTIVE CELL MEDIATED INNATE HUMORAL IMMUNITY ADAPTIVE CELL MEDIATED INNATE HUMORAL ANTIBODIES Structure Sequence diversity Nature of reaction with Ag Different functional properties If bacteria are injected into blood specific substances are found that exhibit different biologic properties Agglutinin - specifically clump only the bacteria used for immunization Opsonin - facilitate engulfment Antitoxin - neutralize toxin associated with the immunizing bacteria Cytolysis - lyse bacteria Precipitins - form flocculate precipitates when added to supernatant from the bacteria If bacteria are injected into blood specific substances are found that exhibit different biologic properties Agglutinin - specifically clump only the bacteria used for immunization Opsonin - facilitate engulfment Antitoxin - neutralize toxin associated with the immunizing bacteria Cytolysis - lyse bacteria Precipitins - form flocculate precipitates when added to supernatant from the bacteria Antibody (Ab) A molecule produced by animal in response to foreign substance (Ag) which has the property of binding the eliciting agent Immunoglobulin (Ig) Antibody (Ab) A molecule produced by animal in response to foreign substance (Ag) which has the property of binding the eliciting agent Immunoglobulin (Ig) Antibodies are found both as cell-associated receptors (BCR) and as circulating effector molecules Antigen (Ag) Any molecule capable of being bound by the combining site of an Ab or the T-cell receptor Immunogen A substance capable of eliciting an immune response; all immunogens are antigens but not all antigens are immunogens. We will now consider the experiments that were done to determine antibody atructure Immune serum from rabbits before and after the antibodies were removed by precipitation with Ag (ovalbumin) The antibodies migrated as a g-globulin Showed they w gamma globulin When purified antibodies (produced by dissolving immunoprecipiates) were analyzed in an ultracentrifuge they ran with a 7S sedimentation co-efficient indicating a m.w. of approx. 150,000 gamma globulin 7S = 150,000 molecular weight gamma globulin 7S = 150,000 molecular weight Molecular analysis of precipitates showed the valence = 2 Two molecules of Ag were precipitated by each Ab gamma globulin 7S = 150,000 molecular weight valence = 2 gamma globulin 7S = 150,000 molecular weight valence = 2 papain --> 2Fab + 1Fc Fab binds Ag but no precipitate Fc forms crystals MW of Fab and Fc = 50,000 Ratio: 2 Fab and 1 Fc Digestion of antibodies with the enzyme papain yielded two fragments, the Fab (fragment antigen binding), and the Fc (fragment crystallizable) gamma globulin 7S = 150,000 molecular weight valence = 2 papain --> 2Fab + 1Fc pepsin--> 1 (Fab')2 of 100,000 binds 2 molecules of Ag can precipitate (Fab')2 break SS-->2Fab gamma globulin 7S = 150,000 molecular weight valence = 2 papain --> 2Fab + 1Fc pepsin--> 1 (Fab')2 of 100,000 Ab--> cleave disulfides--> 2H(50000) + 2L(25000) Antiserum Anti-L reacts only with Fab Anti-H reacts with Fab and Fc Anti-Fab reacts with H and L If break disulfide bonds and separate on the basis of size you get two products gamma globulin gamma globulin7S = 150,000 molecular weight valence = 2 weight 7S = 150,000 molecular papain --> 2Fab + 1Fc valence = 2 pepsin--> 1 (Fab')2 of 100,000 papain --> 2FabAb--> + 1Fc cleave disulfides--> 2H(50000) + 2L(25000) pepsin--> 1 (Fab') 2 of 100,000 Antiserum Anti-L reacts only with Fab Ab--> cleave disulfides--> Anti-H reacts with Fab and Fc 2H(50000) + 2L(25000) Anti-Fab reacts with H and L Antiserum Anti-L reacts only with Fab Anti-H reacts with Fab and Fc Anti-Fab reacts with H and L gamma globulin 7S = 150,000 molecular weight valence = 2 papain --> 2Fab + 1Fc pepsin--> 1 (Fab')2 of 100,000 Ab--> cleave disulfides--> 2H(50000) + 2L(25000) Antiserum Anti-L reacts only with Fab Anti-H reacts with Fab and Fc Anti-Fab reacts with H and L gamma globulin 7S = 150,000 molecular weight valence = 2 papain --> 2Fab + 1Fc pepsin--> 1 (Fab')2 of 100,000 Ab--> cleave disulfides--> 2H(50000) + 2L(25000) Antiserum Anti-L reacts only with Fab Anti-H reacts with Fab and Fc Anti-Fab reacts with H and L This is the structure of the fundamental building block of an Ab molecule. Some antibodies (such as IgG) are made of one of these building blocks. Others (IgA and IgM) are made of multiple copies of this basic building block. One problem with further analysis was the heterogeneity of antibodies: note the narrow peak of the homogenous protein albumin compared to the broad peak of the Ig It was a major breakthrough when it was realized that multiple myeloma is a tumor of plasma cells generally synthesizing a single species of Ig and that the myeloma proteins are homogeneous Abs produced by the plasma cells Bence-Jones proteins are monoclonal (homogeneous) L chains in patients with multiple myeloma How are antibodies capable of binding many different antigens? Light chains are of two types: anti- ++++ - anti- - ++++ Light chains are of two types: anti- ++++ - anti- - ++++ Take two kappa (or lambda) lights chains from two different patients with multiple myeloma and produce proteolytic fragments Light chains are of two types: anti- ++++ - anti- - ++++ Take two kappa (or lambda) lights chains from two different patients with multiple myeloma, produce proteolytic fragments. Analyze the products by 2-D peptide map (chromatography in one dimension, electrophoresis in the other). You find that about half of the cleavage products are identical between the two chains while the other half differ. The shaded spots indicate peptides shared by the two or chains. Note the difference between and . The conclusion is that the lights chains have a portion that is variable and a portion that is constant. Amino acid sequence analysis shows that the variable portions is at one end of the light (and heavy) chains VL CL ( or) VH CH (many different options) VL CL ( or) VH CH (many different options) More extensive analysis of the sequences of variable regions shows that they in fact contain relatively conserved regions and other regions which are hypervariable There are 20 different amino acids. If all are found with equal frequency variability is 20/.05 = 400. If only one amino acid is found the variability is 1/1 =1/ More extensive analysis of the sequences of variable regions shows that they in fact contain relatively conserved regions and other regions which are hypervariable. There are 20 different amino acids. If all are found with equal frequency variability is 20/.05 = 400. If only one amino acid is found the variability is 1/1 =1/ Hypervariable regions are also called complementarity determining regions (CDRs). The prediction is that the CDRs contact antigen. X-crystallography shows that the Ab molecule folds into compact domains. The CDRs are loops extending from the variable regions so that they are easily accessible for interaction with Ag. The other amino acids in the variable region are the “framework” amino acids and provide a scaffold to maintain the CDRs in the proper orientation. Hypervariable regions are also called complementarity determining regions (CDRs). The prediction is that the CDRs contact antigen. Light chains have two domains: one variable and one constant Heavy chains have four or five domains, one of which is variable with the remains constant All heavy chains have at least one carbohydrate moiety attached The forces are weak and operate at short distances Therefore the closer the approach (the better the fit) the stronger the interaction A complex of an Ab and a protein Ag shows: the reaction occurs between two interacting surfaces the better the fit, the stronger the interaction most but not all of the interaction with Ag occurs through the CDRs on a protein Ag the amino acids comprising the epitopes are adjacent in 3D space but not necessarily in linear sequence Another characteristic of the antibody molecule is a flexible hinge When a divalent hapten reacts with Abs, trimers, tetramers and other large complexes form. Because of the flexibility of the hinge region Digestion with papain removes the Fc There are several different possible structure of the constant regions of the heavy chains Classes Heavy Chain Subclasses (human) IgM g IgG IgG1, IgG2, IgG3, IgG4 IgA IgA1, IgA2 IgE IgD Classes + subclasses = isotypes Light chains have two isotypes: and Each isotype is encoded by a separate gene IgM H chain has four constant region domains and no hinge In addition to H and L also contains J chain (H2L2)5J or (H2L2)6 Usually the first Ig made in response to antigen Increased valence leads to increased avidity IgG IgG IgG Most abundant serum immunoglobulin IgA J SC Monomer Dimeric IgA Secretory IgA Present in secretions where it provides protection at epithelial surfaces Frequently found as a polymer with J chain In secretions it also has a fourth chain, secretory component, a product of the epithelial cells IgE Present at low concentrations Responsible for allergy Binds to receptors on mast cells IgD Principle role seems to be as a membrane Ig All isotypes can exist as both secreted and membrane bound antibodies All isotypes can exist as both secreted and membrane bound antibodies. The two forms differ in their carboxy terminal sequence. Secreted antibodies have a hydrophilic terminus. Membrane antibodies have a hydrophobic sequence which inserts into the plasma membrane and a short cytoplasmic tail.