* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Lectures 1-3: Review of forces and elementary statistical
Artificial gene synthesis wikipedia , lookup
G protein–coupled receptor wikipedia , lookup
Protein moonlighting wikipedia , lookup
Interactome wikipedia , lookup
Point accepted mutation wikipedia , lookup
Genetic code wikipedia , lookup
Chemical biology wikipedia , lookup
Expanded genetic code wikipedia , lookup
Biosynthesis wikipedia , lookup
Bioinformatics wikipedia , lookup
Homology modeling wikipedia , lookup
Metalloprotein wikipedia , lookup
Western blot wikipedia , lookup
Biochemistry wikipedia , lookup
History of biotechnology wikipedia , lookup
Protein–protein interaction wikipedia , lookup
Protein adsorption wikipedia , lookup
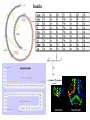
Insulin Insulin: the protein of the 20th century • One of the first proteins crystallized in 1926 (Structural Genomics) • First protein fully sequenced in 1955 (Bioinformatics) • First protein chemically synthesized in 1958 • First human protein manufactured via recombinant in 1979 (Biotechnology) Studying the three-dimensional structure of insulin As described above, human insulin consists of 51 amino acids, divided into two chains, commonly labeled A and B, with 21 and 30 amino acids respectively. The chains are linked by three disulfide bridges, two forming inter-chain cystine at A7-B7 and A20-B19, and one forming an intra-chain cystine at A6-A11. A piece of anti-parallel b-sheet is formed upon dimerization: residues B23 to B28 of one monomer lie anti-parallel to the same stretch in the other monomer. There are two very small a-helices in the A chain, and a three turn a-helix running from residues B9 to B19 is found in every insulin structure known so far.