* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download EOC Review - Dorman Freshman Campus
Bond valence method wikipedia , lookup
Nuclear binding energy wikipedia , lookup
Electrolysis of water wikipedia , lookup
Resonance (chemistry) wikipedia , lookup
Artificial photosynthesis wikipedia , lookup
Lewis acid catalysis wikipedia , lookup
Chemical thermodynamics wikipedia , lookup
Bioorthogonal chemistry wikipedia , lookup
Atomic orbital wikipedia , lookup
Periodic table wikipedia , lookup
Chemical element wikipedia , lookup
Transition state theory wikipedia , lookup
Chemical reaction wikipedia , lookup
Isotopic labeling wikipedia , lookup
Low-energy electron diffraction wikipedia , lookup
X-ray fluorescence wikipedia , lookup
Photoredox catalysis wikipedia , lookup
Molecular orbital diagram wikipedia , lookup
Physical organic chemistry wikipedia , lookup
Stoichiometry wikipedia , lookup
X-ray photoelectron spectroscopy wikipedia , lookup
History of molecular theory wikipedia , lookup
Rutherford backscattering spectrometry wikipedia , lookup
IUPAC nomenclature of inorganic chemistry 2005 wikipedia , lookup
Electronegativity wikipedia , lookup
Electron scattering wikipedia , lookup
Elementary particle wikipedia , lookup
Electrochemistry wikipedia , lookup
Oxidative phosphorylation wikipedia , lookup
History of chemistry wikipedia , lookup
Electrical resistivity and conductivity wikipedia , lookup
Hypervalent molecule wikipedia , lookup
Chemistry: A Volatile History wikipedia , lookup
Extended periodic table wikipedia , lookup
Electron configuration wikipedia , lookup
Metallic bonding wikipedia , lookup
Photosynthetic reaction centre wikipedia , lookup
Chemical bond wikipedia , lookup
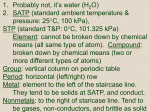
Final Review Physical Science 1. 1. Protons – positive 2. Neutrons - neutral 3. Electrons - negative 2. 1. Protons – nucleus 2. Neutrons – nucleus 3. Electrons – electron cloud 3. 1. Protons 2. Neutrons Charge – positive 4. Protons and neutrons. Not based on electrons because the mass of the electron is too small to count. 5. Stable isotope – same number of protons but different number of neutrons. Does not break down Unstable isotope – radioactive isotope used for nuclear weapons, nuclear power plants, and biomedicine. Highly radioactive,Unpredictable in breaking down. 6. Atomic number Yes, because the number of protons never change 7. Oxidation number – number of electrons an atom can gain, lose, or share (based on group #) O: 2Ba: 2+ Li: 1+ N: 3C: 4+ 8. They have 8 valence electrons and are stable . They do not need to react with another group to become stable. 9. Atomic number 10 - 12. • Metals – left of stair step line • Nonmetals – right of stair step line • Metalloids – on stair step line 13. # of valence electrons Element Atomic Mass Atomic # # of protons # of # of electron neutron s s H 1.01 1 1 1 0 He 4.00 2 2 2 2 C 12.01 6 6 6 6 Cl 35.45 17 17 17 19 O 16.00 8 8 8 8 20. Group 1 has one valence electron and has to give it away. Group 17 has 7 valence electrons and needs one more to be stable. 21. Halogens - Group 17: 1Alkali Metals - Group 1: 1+ Alkaline earth – Group 2: 2+ Nobel Gases – Group 18: 0 22. Physical properties: Color, shape, size, odor, melting point, boiling point, state of matter, viscosity, density, solubility Chemical properties: Flammability, sensitivity to light, oxidation, tarnishing, corrosion of metals 23. Physical change: Change in a substance’s size, shape, or state of matter Chemical change: A change of one substance into a different substance Do you still have the same substance or is it a new substance? 24. Iron Rusts: Chemical Ice Melting: Physical Milk Sours: Chemical Food is digested: Chemical Evaporation: Physical Sugar dissolves in water: Physical 25. 1. Physical 2. Density = mass / volume (d=m/v) 3. D = 30g / 12cm3 d = 2.5 g/cm3 26. • Organic compounds always contain carbon and hydrogen atoms. 27. Atom: smallest part of an element that can be identified as that element. Molecule: neutral particle that results from a covalent bond. Can also be polar or nonpolar. 28. Element: Substance that cannot be chemically broken down into a simpler substance. Compound: Two or more elements chemically combined. Differences: Element is by itself where a compound is two or more elements. 29. Compounds are chemically combined while mixtures are physically combined. 1. Hetergeneous – unevenly mixed ex: suspension, salad, chocolate chip cookie, granite 2. Homogeneous – evenly mixed ex: soda, vinegar, saltwater 30. Ionic: Metal and nonmetal Covalent: 2 or more nonmetals Difference: Ionic – gain and loses electrons Covalent – share electrons 31. H and O: H1+ O H2O 2- Na and Cl: Na 1+ Na and O: Na 1+ Be and O: Be 2+ Cl 1- O2O2- NaCl Na2O BeO 32. • Boiling Point: Covalent - low • Melting Point: Ionic - high (does not easily break down) Covalent – low (comes apart easily) • Conductivity: Ionic – conducts electricity; covalent does not 33. • Stirring • Temperature • Pressure • Surface Area 34. • Solids – tightly packed particles vibrating in place • Liquids – particles are close, but can move around each other and are not in one place • Gases – particles bounce off of each other and are as far apart as possible 35. • S to L: particles start slipping and sliding past each other. The bond holding the particles together is broken • L to G: particles move fast throughout container and are colliding with one another 36. • A. Solid • B. Melting Pt • C. Liquid • D. Boiling Pt • E. Gas • F. Condensation Pt • G. Freezing Pt 37. • Fe + O2 Fe2O3 No, more iron and oxygen on product side CH4 + O2 CO2 + H2O 38. • Exothermic: heat released; reaction becomes hot Ex: burning sugar • Endothermic: heat absorbed; reaction becomes cold Ex: ice pack 39 • Mass of a reaction is not created nor destroyed but conserved. 40 • You have to have equal amounts of each element on the reactant and product side of the equation • This is why the equation must be BALANCED Formulas Reactions with metals pH Acids All have Corrodes metals H as first element in formula 0-7 Bases All have They can OH in end but of formula generally do not Above 7 42 • How quick or slow it takes for a reaction to occur 43. Ways to increase reaction rate • Increase temp • Stir • Adding a catalysts • Increasing surface area (break down particle size) • Increasing the concentration 28 •Elements and compounds are both pure substances •Elements are made of one type of atom •Compounds are made of two or more types of atoms Pg 90-93 •Ionic bond – metal + nonmetal and electrons are transferred •Covalent bond – nonmetal + nonmetal and electrons are shared Pg 90-93 • Criss-Cross Method • H and O …H+1 O-2 • H2O • Remember – don’t write the charges, the 1’s, or if the numbers are the same -2 and +2 Pg 90-93 •Covalent bonds are weak: 1. low melting point 2. low boiling point 3. will not conduct electricity 110-111. •Temperature – faster if heated •Surface area – faster if more surface area •Stirring – faster if you stir it Pg 110-111 High concentration = more conductivity, lower freezing point Pg 111 •Polar – water is a polar substance, so if it will dissolve in water, it is polar…like vinegar •Nonpolar – anything that will not dissolve in water is nonpolar…like oil Pg 94 Compounds with a hydrogen and a carbon •Pg 94 (See board) •Draw the following hydrocarbons bonded •C2H6 •C8H18 Pg 96-99 •Polymer is a long chain of something •Protein – polymer of amino acids •Starch – polymer of sugar •Example – •Train – polymer of cars Pg 96-99 Polymers provide the nutrients we need for daily function…we take in nutrients and our body breaks them down and rearranges them into things we can use. Pg 96-99 Proteins Carbohydrates – starches and sugars give us energy Lipids – fats that Pg 173 Fe + See Board O2 Fe2O3 Pg 175. •Exothermic – heat is given off – temperature goes up •Endothermic – heat is taken in – temperature goes down Pg 173 •Matter cannot be created nor destroyed •The amount of mass you started with before the reaction is the same after the reaction. •Ex: burning a log Pg 173 An equation must be balanced to exhibit the above law – in an unbalanced equation, there would be mass being created or destroyed •Acids have a sour taste •Bases have a bitter taste and a slippery texture. Pg 190-193 •pH is very common in a grocery store… •Lemons – pH 2 •Drain cleaner – pH 14 •Apple juice – pH 3 •Soap – pH 10 Pg 169-171 Reaction rate is how quickly a reaction will happen Pg 169-171 You can speed up a reaction by raising the temperature, stirring it, adding catalysts or enzymes, increasing surface area, or increasing the concentration Pg 168-169 •Change in color •Change in temperature •A precipitate forms (a solid glob forms at the bottom) Acid rain is formed from the pollution and other gases emitted from the environment which is surrounded by dust and dirt in clouds….the dust and dirt fall as rain bringing with it the pollution which is usually acidic Pg 272-273 d d No motion Constant speed t t v Constant speed v acceleration t t Pg 428-431 Static electricity is made when two things rub together…electrons jump from one object to another Pg 462-465 •Electricity can produce a magnetic field •Magnets can make electricity flow faster Pg 442-443 •Voltage is how much electricity CAN flow •Amperage (current) is how much actually DOES flow Pg 292 - 293 PE = high KE = low PE = high KE = low PE = low KE = high Pg 296-305 Energy • Energy is the ability to do work • Work is using energy to do a task • Power is how fast you do work • Efficiency is how well you used the energy to do the work Mechanical Advantage tells you how well a machine works MA = resistance force effort force Ex. What is the MA of a lever that does 500N with 50N of effort?