* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Organic Chemistry II Introduction
Survey
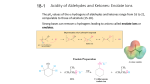
Document related concepts
Chemical equilibrium wikipedia , lookup
Woodward–Hoffmann rules wikipedia , lookup
Electrochemistry wikipedia , lookup
Equilibrium chemistry wikipedia , lookup
Acid dissociation constant wikipedia , lookup
Physical organic chemistry wikipedia , lookup
Rate equation wikipedia , lookup
George S. Hammond wikipedia , lookup
Reaction progress kinetic analysis wikipedia , lookup
Transition state theory wikipedia , lookup
Enzyme catalysis wikipedia , lookup
Ene reaction wikipedia , lookup
Photoredox catalysis wikipedia , lookup
Transcript
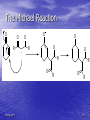
Organic Chemistry II Alpha Substitution Reactions Dr. Ralph C. Gatrone Department of Chemistry and Physics Virginia State University Spring, 2011 1 The Position • The carbon next to the carbonyl group is designated as being in the position gamma H H alpha H H beta Spring, 2011 O 2 Objectives • Keto-enol tautomerism – Enols – substitution – Enol acidity – the enolate – Alkylation of the enolate • Carbonyl Condensation Reactions – Aldol – Claisen • Michael Reaction • Stork Enamine • Robinson Annulation Spring, 2011 3 Enolization • conditions permit the enolization of ketones • Substitution can occur at the alpha position through either an enol or enolate ion Spring, 2011 4 Equilibrium of Tautomerism • Enol formation is equilibrium process O OH K = 10-6 O OH K = 10-8 • Keto form is predominant species • Enol is present in sufficient concentration to be involved in any reaction Spring, 2011 5 Tautomers • Tautomers are structural isomers • Tautomers are not resonance forms • Resonance forms are representations of contributors to a • single structure Tautomers interconvert rapidly while ordinary isomers do not Spring, 2011 6 Enols • The enol tautomer is usually present to a very small extent and cannot be isolated • However, since it is formed rapidly, it can serve as a reaction intermediate Spring, 2011 7 Acid Catalysis of Enolization • Brønsted acids catalyze keto-enol tautomerization by protonating the carbonyl and activating the protons Spring, 2011 8 Base Catalysis of Enolization • Brønsted bases • • catalyze keto-enol tautomerization The hydrogens on the carbon are weakly acidic and transfer to water is slow In the reverse direction there is also a barrier to the addition of the proton from water to enolate carbon Spring, 2011 9 Reactivity of Enols • Enols have very e- rich double bonds • Very Nucleophilic • React with variety of electrophiles Spring, 2011 10 Generalized Reaction • When an enol reacts with an electrophile the intermediate cation immediately loses the OH proton to give a substituted carbonyl compound Spring, 2011 11 Halogenation • Aldehydes and ketones can be halogenated at their positions by reaction with Cl2, Br2, or I2 in acidic solution Spring, 2011 12 Kinetics • Rate is independent of identity of X2 – Cl2, Br2, I2 react at same rate • Rate is independent of [X2] • Rate determining step occurs before X2 • Rate determining step is enolization step • Rate = k[ketone][H+] Spring, 2011 13 Ramification of Enolization • Optically active ketones can lose optical activity O CH3 H O O OH CH3 CH3 H H CH3 racemic Spring, 2011 14 Elimination Reactions of -Bromoketones • -Bromo ketones can be dehydrobrominated by base treatment to yield ,b-unsaturated ketones Spring, 2011 15 Alpha Bromination of Carboxylic Acids: The Hell–Volhard–Zelinskii Reaction • Carboxylic acids do not react with Br2 (Unlike • aldehydes and ketones) They are brominated by a mixture of Br2 and PBr3 (Hell–Volhard–Zelinskii reaction) Spring, 2011 16 Acidity of Alpha Hydrogen Atoms: Enolate Ion Formation • Carbonyl compounds can act as weak acids (pKa • of acetone = 19.3; pKa of ethane = 60) The conjugate base of a ketone or aldehyde is an enolate ion - the negative charge is delocalized onto oxygen Spring, 2011 17 Enolate Formation - Bases • Ketones are weaker acids than the OH of • • alcohols so a a more powerful base than an alkoxide is needed to form the enolate NaOH and NaOCH3) are too weak Sodium hydride (NaH), sodamide (NaNH2), or lithium diisopropylamide [LiN(i-C3H7)2] are strong enough to form the enolate Spring, 2011 18 Lithium Diisopropylamide (LDA) • LDA is made from butyllithium (BuLi) and • • diisopropylamine (pKa 40) Soluble in organic solvents and effective at low temperature with many compounds (see Table 22.1) Not nucleophilic Spring, 2011 19 Similar Bases • Lithium tetramethylpiperdide Li+ N Li+ • Lithium N - dicyclohexylamide Spring, 2011 20 Acidity of the alpha Hydrogen O O O H Cl H H H 19 O H O 25 O O OR H 9 Spring, 2011 OR 17 16 O O H 11 O RO OR H 13 21 Acidity of b-Dicarbonyls • When a hydrogen atom is flanked by two carbonyl groups, its acidity is enhanced • Negative charge of enolate delocalizes over both carbonyl groups Spring, 2011 22 Acidities of Organic Compounds Spring, 2011 23 Reactivity of Enolate Ions • The carbon atom of an enolate ion is electronrich and highly reactive toward electrophiles (enols are not as reactive) Spring, 2011 24 Enolate Reactions • Enolate has two resonance forms • Reacts at both sites with E+ depending upon conditions O - O - E+ E+ O Spring, 2011 E O E 25 Two Reactions Sites on Enolates • Reaction on oxygen yields an enol derivative • Reaction on carbon yields an -substituted carbonyl compound Spring, 2011 26 Enol Ether Formation • Enolates react with trimethylsilyl chloride to form the enol ether O Spring, 2011 TMSCl O-TMS 27 Alkylation • Alkylation occurs at C O CH3Br O CH3 Spring, 2011 28 Revisit Halogenation of the Enolate The Haloform Reaction • Base-promoted reaction occurs through an enolate ion intermediate Spring, 2011 29 Further Reaction: Cleavage • Monohalogenated products are themselves rapidly • turned into enolate ions and further halogenated until the trihalo compound is formed from a methyl ketone The product is cleaved by hydroxide with -CX3 as the leaving group, stabilized anion Spring, 2011 30 Halogenation: Summary • Base catalyzed process – Can’t be stopped at one halogen – Cleaves methyl ketones to give HCX3 • Acid catalyzed process – Second halogenation is very slow – Useful reaction for monohalo ketones Spring, 2011 31 Alpha Selenylation • Used to make unsaturated ketones O O - LDA O Spring, 2011 Ph Se PhSeCl O Se O H2O2 O - Ph O Se + Ph 32 Alkylation of Enolate Ions • Alkylation occurs when the nucleophilic enolate ion reacts with the electrophilic alkyl halide or tosylate and displaces the leaving group Spring, 2011 33 Constraints on Enolate Alkylation • SN2 reaction:, the leaving group X can be • • chloride, bromide, iodide, or tosylate R should be primary, methyl, allylic or benzylic Secondary halides react poorly, and tertiary halides don't react at all because of competing elimination Spring, 2011 34 Alkylation of Ketones • Base must be weaker acid than ketone – Complete proton abstraction • Base cannot be nucleophilic – Adds to carbonyl • LDA meets both criteria • Consider: Spring, 2011 35 Deprotonation by LDA O O - O - LDA + CH3I O O + 6% Spring, 2011 56% 36 Analysis • Least hindered product predominates – Steric size of base – Approach to more substituted side restricted – Increase in the size of the base increases the predominance of the less substituted side product Spring, 2011 37 The Malonic Ester Synthesis • For preparing a carboxylic acid from an alkyl halide while lengthening the carbon chain by two atoms Spring, 2011 38 Formation of Enolate and Alkylation • Malonic ester (diethyl propanedioate) is easily • converted into its enolate ion by reaction with sodium ethoxide in ethanol The enolate is a good nucleophile that reacts rapidly with an alkyl halide to give an substituted malonic ester Spring, 2011 39 Dialkylation • The product has an acidic -hydrogen, allowing the alkylation process to be repeated Spring, 2011 40 Hydrolysis and Decarboxylation • The malonic ester derivative hydrolyzes in acid and loses CO2 (“decarboxylation”) to yield a substituted monoacid Spring, 2011 41 Malonic Acid Syntheses • Very useful reaction, • For example Br CO2Et Br Br H2C(CO2Et)2 Base HC(CO2Et)2 CO2Et Br CO2Et CO2Et Br Br CO2Et CO2Et • All can be decarboxylated to the mono acids Spring, 2011 42 Decarboxylation of b-Ketoacids • Decarboxylation requires a carbonyl group two atoms • • away from the CO2H The second carbonyl permit delocalization of the resulting enol The reaction can be rationalized by an internal acid-base reaction Spring, 2011 43 Decarboxylation • Beta-dicarboxylic acids readily lose CO2 • Other beta carboxy carbonyls should also • Look at Ethyl acetoacetate (pKa = 11) O O OEt H Spring, 2011 44 Acetoacetate Synthesis O O base OEt H O O O OEt RX R R base RX O after loss of CO2 R R Spring, 2011 45 Reminder of Overall Conversion • The malonic ester synthesis converts an alkyl halide into a carboxylic acid while lengthening the carbon chain by two atoms Spring, 2011 46 Decarboxylation of Acetoacetic Acid b-Ketoacid from hydrolysis of ester undergoes decarboxylation to yield a ketone via the enol Spring, 2011 47 Generalization: b-Keto Esters • The sequence: enolate ion formation, alkylation, • hydrolysis/decarboxylation is applicable to bketo esters in general Cyclic b-keto esters give 2-substituted cyclohexanones Spring, 2011 48 Carbonyl Condensation Reactions • Carbonyls behave – Electrophile or Nucleophile • Electrophilic Behavior – Nu: add to C=O • Nucleophilic Behavior – Enols or enolates react with E+ • Condensation Reactions – – – – Combine both reactions Uses two carbonyl compounds One is the Nu: through an enolate Second is the E+ Spring, 2011 49 The Aldol Reaction enolate (nucleophile) O O O R - R R R R - OH R R R R O R R R O aldol • Equilibrium reaction • Products favored for aldehydes • Reactants favored for ketones Spring, 2011 50 Choice of Base • LDA • Enolate forms completely • No aldol products observed • NaOCH3 • [enolate] is low • Aldol observed Spring, 2011 51 Dehydration of Aldols • Dehydration of the aldol occurs readily • Conjugated enones are formed R R OH acid or base R R R R O R R O aldol Spring, 2011 52 Intramolecular Aldol • Dicarbonyl compounds • React with base • Cyclic aldols result • Prepare Cyclopentanones • Prepare Cyclohexanones • Smaller rings can’t form • TS is too high in energy Spring, 2011 53 Intramolecular Aldol Reactions O O base O O - O O O O OH Spring, 2011 54 Claisen Condensation • Esters are weakly acidic • Esters undergo a reversible condensation reaction: O OCH2CH3 O O NaOCH2CH3/CH3CH2OH - OCH2CH3 OCH2CH3 - O O OCH2CH3 CH3CH2O CH3CH2O O O Ethyl acetoacetate O O OCH2CH3 Spring, 2011 55 Dieckmann Cyclization • Intramolecular Claisen Condensation O O NaOEt OEt O OEt OEt O Spring, 2011 56 Decarboxylation after Cyclization • beta-ketoester can be alkylated • Hydrolyzed and decarboxylated O O O 1. base OEt R 2. RX 3. NaOH(aq) 4. HCl Spring, 2011 57 The Michael Reaction • Unsaturated ketones react with nucleophiles • 1,2 reaction to yield alcohols • 1,4 reaction to yield substituted ketones • Nucleophile used determines the result obtained • Enolates also react 1,4 with unsaturated ketones • The Michael Reaction Spring, 2011 58 The Michael Reaction O R O O O - O O R O R O Spring, 2011 R R O R 59 Stork Enamine Reaction • Secondary amines react with aldehydes and ketones to form enamines O acid N N H Spring, 2011 60 Enamines are similar to enolates • Both structures have a nucleophilic carbon N N+ nucleophilic carbon atom O O - Spring, 2011 61 Stork Enamine Reaction • Enamine reacts with unsaturated carbonyl • 3 step process: • 1. form enamine • 2. react with unsaturate ketone • 3. hydrolyze the enamine back to carbonyl Spring, 2011 62 The Stork Enamine Reaction H N O N acid O N N O N O O O acid Spring, 2011 63 The Robinson Annulation • Enolates react 1,4 with unsaturated ketones or aldehydes • Enolates also react 1,2 with ketones or aldehyes • Combine both reactions in a single process Spring, 2011 64 Robinson Annulation O O O O NaOEt OEt + O O - OEt + H most acidic proton Spring, 2011 65 Robinson Annulation O O O O - OEt + (Michael Addition) OEt O O Spring, 2011 66 Robinson Annulation O O - - OEt OEt O O O O Spring, 2011 67 Robinson Annulation O ketone C=O is more electrophilic than ester - OEt O O O O O O OEt O OH O Spring, 2011 OEt O OEt 68 Robinson Annulation • Provides facile entry into functionalized six-membered rings • This process has been used successfully to prepare a number of important 6 membered ring systems • e.g. many steroids have been prepared in this manner Spring, 2011 69