* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Chapter 10 - Clayton State University
Survey
Document related concepts
Fatty acid metabolism wikipedia , lookup
Photosynthesis wikipedia , lookup
Mitochondrion wikipedia , lookup
Evolution of metal ions in biological systems wikipedia , lookup
Biochemistry wikipedia , lookup
Adenosine triphosphate wikipedia , lookup
Microbial metabolism wikipedia , lookup
Photosynthetic reaction centre wikipedia , lookup
NADH:ubiquinone oxidoreductase (H+-translocating) wikipedia , lookup
Citric acid cycle wikipedia , lookup
Light-dependent reactions wikipedia , lookup
Transcript
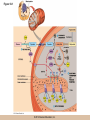
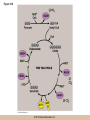
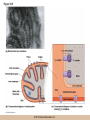
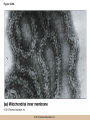
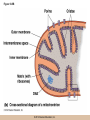
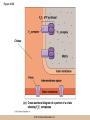
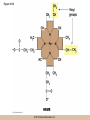
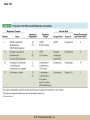
Cellular Respiration CHAPTER 10 © 2012 Pearson Education, Inc. Chemotrophic Energy Metabolism: Aerobic Respiration • Some cells meet their energy needs through anaerobic fermentation • However, fermentation yields only modest amounts of energy due to the absence of electron transfer • ATP yield is much higher in cellular respiration © 2012 Pearson Education, Inc. Cellular Respiration: Maximizing ATP Yields • Cellular respiration (or respiration) uses an external electron acceptor to oxidize substrates completely to CO2 • External electron acceptor: one that is not a by-product of glucose catabolism © 2012 Pearson Education, Inc. Cellular respiration defined • Respiration is the flow of electrons through or within a membrane, from reduced coenzymes to an external electron acceptor usually accompanied by the generation of ATP • Coenzymes such as FAD (flavin adenine dinucleotide) and coenzyme Q (ubiquinone) are involved © 2012 Pearson Education, Inc. The terminal electron acceptor • In aerobic respiration, the terminal electron acceptor is oxygen and the reduced form is water • Other terminal electron acceptors (sulfur, protons, and ferric ions) are used by other organisms, especially bacteria and archaea • These are examples of anaerobic respiration © 2012 Pearson Education, Inc. Mitochondria • Most aerobic ATP production in eukaryotic cells takes place in the mitochondrion • In bacteria, the plasma membrane and cyotoplasm are analogous to the mitochondrial inner membrane and matrix with respect to energy metabolism © 2012 Pearson Education, Inc. Figure 10-1 © 2012 Pearson Education, Inc. Aerobic Respiration Yields Much More Energy than Fermentation Does • With O2 as the terminal electron acceptor, pyruvate can be oxidized completely to CO2 • Aerobic respiration has the potential of generating up to 38 ATP molecules per glucose • Oxygen provides a means of continuous reoxidation of NADH and other reduced coenzymes © 2012 Pearson Education, Inc. Respiration Includes Glycolysis, Pyruvate Oxidation, the TCA Cycle, Electron Transport, and ATP Synthesis • Respiration will be considered in five stages – Stage 1: the glycolytic pathway – Stage 2: pyruvate is oxidized to generate acetyl CoA – Stage 3: acetyl Co A enters the tricarboxylic acid cycle (TCA cycle), where it is completely oxidized to CO2 © 2012 Pearson Education, Inc. The five stages, continued • Respiration will be considered in five stages – Stage 4: electron transport, the transfer of electrons from reduced coenzymes to oxygen coupled to active transport of protons across a membrane – Stage 5: The electrochemical proton gradient formed in step 4 is used to drive ATP synthesis (oxidative phosphorylation) © 2012 Pearson Education, Inc. The Mitochondrion: Where the Action Takes Place • The mitochondrion is called the “energy powerhouse” of the eukaryotic cell • These organelles are thought to have arisen from bacterial cells • Mitochondria have been shown to carry out all the reactions of the TCA cycle, electron transport, and oxidative phosphorylation © 2012 Pearson Education, Inc. Video: Mitochondria in 3-D © 2012 Pearson Education, Inc. Mitochondria Are Often Present Where the ATP Needs Are Greatest • Mitochondria are found in virtually all aerobic cells of eukaryotes • They are present in both chemotrophic and phototrophic cells • Mitochondria are frequently clustered in regions of cells with the greatest need for ATP, e.g., muscle cells © 2012 Pearson Education, Inc. Are Mitochondria Interconnected Networks Rather than Discrete Organelles? • In electron micrographs, mitochondria usually appear as oval structures • However they can take various shapes and sizes, depending on the cell type • Their appearance under EM suggests that they are large, and numerous discrete entities © 2012 Pearson Education, Inc. The Outer and Inner Membranes Define Two Separate Compartments and Three Regions • A distinctive feature of mitochondria is the presence of both outer and inner membranes • The outer membrane contains porins that allow passage of solutes with molecular weights up to 5000 • The intermembrane space between the inner and outer membranes is thus continuous with the cytosol © 2012 Pearson Education, Inc. The inner membrane • The inner membrane of the mitochondria is impermeable to most solutes, partitioning the mitochondrion into two separate compartments – The intermembrane space – The interior of the organelle, or mitochondrial matrix © 2012 Pearson Education, Inc. The inner boundary membrane and cristae • The portion of the inner membrane adjacent to the intermembrane space is called the inner boundary membrane • The inner membrane is about 75% protein by weight; the proteins include those involved in solute transport, electron transport, and ATP synthesis © 2012 Pearson Education, Inc. The cristae • The inner membrane of most mitochondria has many infoldings called cristae • They increase surface area of the inner membrane, and provide more space for electron transport to take place • The cristae provide localized regions, intracristal spaces, where protons can accumulate during electron transport © 2012 Pearson Education, Inc. The cristae (continued) • The cristae are thought to be tubular structures that associate in layers • They have limited connections to the inner boundary membrane through small openings, crista junctions • Cells with high metabolic activity seem to have more cristae in their mitochondria © 2012 Pearson Education, Inc. The mitochondrial matrix • The interior of the mitochondrion is filled with a semi-fluid matrix • The matrix contains many enzymes involved in mitochondrial function as well as DNA molecules and ribosomes • Mitochondria contain proteins encoded by their own DNA as well as some that are encoded by nuclear genes © 2012 Pearson Education, Inc. Mitochondrial Functions Occur in or on Specific Membranes and Compartments • Specific functions and pathways have been localized within mitochondria by fractionation studies • Most of the enzymes involved in pyruvate oxidation, the TCA cycle, and catabolism of fatty acids and amino acids are found in the matrix • Most electron transport intermediates are integral inner membrane components © 2012 Pearson Education, Inc. Table 10-1 © 2012 Pearson Education, Inc. Localization of Specific Mitochondrial Functions • Knoblike spheres called F1 complexes protrude from the inner membrane into the matrix • These are involved in ATP synthesis • Each complex is an assembly of several different polypeptides, and can be seen in an electron micrograph using negative staining © 2012 Pearson Education, Inc. F1 complexes • Each F1 complex is attached by a short protein stalk to an Fo complex • This is an assembly of hydrophobic polypeptides embedded in the mitochondrial inner membrane • This FoF1 complex is an ATP synthase that is responsible for most of the ATP generation in the mitochondria (and in bacterial cells as well) © 2012 Pearson Education, Inc. The Tricarboxylic Acid Cycle: Oxidation in the Round • In the presence of oxygen pyruvate is oxidized fully to carbon dioxide with the released energy used to drive ATP synthesis • This involves the TCA (tricarboxylic acid) cycle, in which citrate is an important intermediate • The TCA cycle is also called the Krebs cycle after Hans Krebs, whose lab played a key role in elucidating the cycle © 2012 Pearson Education, Inc. The Tricarboxylic Acid Cycle • The TCA cycle metabolized acetyl CoA, produced from pyruvate decarboxylation • Acetyl CoA can also arise from fatty acid oxidation • Acetyl CoA transfers its acetate group to a fourcarbon acceptor called oxaloacetate, generating citrate © 2012 Pearson Education, Inc. The fate of citrate • After its formation, citrate undergoes two successive decarboxylations • It also goes through several oxidation steps • Eventually oxaloacetate is regenerated, and can accept two more carbons from acetyl CoA and the cycle begins again © 2012 Pearson Education, Inc. The overall cycle • Each round of the TCA cycle involves the entry of two carbons, the release of two CO2, and the regeneration of oxaloacetate • Oxidation occurs at five steps, four in the cycle itself and one when pyruvate is converted to acetyl CoA • In each case, electrons are accepted by coenzymes © 2012 Pearson Education, Inc. Figure 10-6 © 2012 Pearson Education, Inc. Figure 10-5 © 2012 Pearson Education, Inc. Figure 10-5A © 2012 Pearson Education, Inc. Figure 10-5B © 2012 Pearson Education, Inc. Figure 10-5C © 2012 Pearson Education, Inc. Pyruvate Is Converted to Acetyl Coenzyme A by Oxidative Decarboxylation • The glycolytic pathway ends with pyruvate, which is small enough to enter the intermembrane space of the mitochondrion • At the inner mitochondrial membrane, a specific symporter transports pyruvate into the matrix, along with a proton • Then, pyruvate is converted to acetyl CoA by pyruvate dehydrogenase complex (PDH) © 2012 Pearson Education, Inc. Conversion of pyruvate • The conversion is a decarboxylation because one carbon is liberated as CO2 • It is also an oxidation because two electrons (and one proton) are transferred to NAD+ to form NADH • Coenzyme A contains the B vitamin • Coenzyme A has a SH group that makes it a good carrier of acetate (and other organic acids) © 2012 Pearson Education, Inc. Conversion of pyruvate • The conversion of pyruvate to acetyl CoA can be summarized as follows: – © 2012 Pearson Education, Inc. The TCA Cycle Begins with the Entry of Acetate as Acetyl CoA • With each round of the TCA cycle, two carbon atoms enter in organic form as acetate and leave in inorganic form as carbon dioxide • In the first reaction, TCA-1, the two-carbon acetate group is transferred from acetyl CoA to oxaloacetate (4C) to form citrate (6C) • This reaction is catalyzed by citrate synthetase © 2012 Pearson Education, Inc. Figure 10-10 © 2012 Pearson Education, Inc. Summing Up: The Products of the TCA Cycle Are CO2, ATP, NADH, and FADH2 • The TCA cycle accomplishes the following: 1. Two carbons enter the cycle as acetyl CoA, which joins oxaloacetate to form the six-carbon citrate 2. Decarboxylation occurs at two steps to balance the input of two carbons by releasing two CO2 3. Oxidation occurs at four steps, with NAD+ the electron acceptor in three steps and FAD in one © 2012 Pearson Education, Inc. The Products of the TCA Cycle Are CO2, ATP, NADH, and FADH2 (continued) • The TCA cycle accomplishes the following: 4. ATP is generated at one point, with GTP as an intermediate in the case of animal cells 5. One turn of the cycle is completed as oxaloacetate, the original 4C acceptor, is regenerated © 2012 Pearson Education, Inc. Summing up the TCA cycle • The TCA cycle can be summarized as follows: • Including glycolysis, pyruvate decarboxylation, and the TCA cycle © 2012 Pearson Education, Inc. Several TCA Cycle Enzymes Are Subject to Allosteric Regulation • Like all metabolic pathways, the TCA cycle must be carefully regulated to meet cellular needs • Most of the control of the cycle involves allosteric regulation of four key enzymes by specific effector molecules • Effector molecules may be activators or inhibitors © 2012 Pearson Education, Inc. Regulation of PDH • PDH is reversibly inactivated by phosphorylation and activated by dephosphorylation of one of its protein components • PDH is inhibited by ATP, which is abundant where energy is plentiful © 2012 Pearson Education, Inc. The TCA Cycle Also Plays a Central Role in the Catabolism of Fats and Proteins • The TCA cycle represents the main conduit of aerobic energy metabolism for a variety of substrates besides sugar, in particular, fats and proteins © 2012 Pearson Education, Inc. Fat as a Source of Energy • Fats are highly reduced compounds that liberate more energy per gram upon oxidation than do carbohydrates • They are a long-term energy storage form for many organisms • Most fat is stored as deposits of triacylglycerols, neutral triesters of glycerol and long-chain fatty acids © 2012 Pearson Education, Inc. Catabolism of triacylglycerols • Triacylglycerol catabolism begins with their hydrolysis to glycerol and free fatty acids • The glycerol is channeled into the glycolytic pathway • Fatty acids are linked to coenzyme A, to form fatty acyl CoAs, then degraded by b-oxidation © 2012 Pearson Education, Inc. b-oxidation • b-oxidation is a catabolic process that generates acetyl CoA and the reduced coenzymes NADH and FADH2 • b-oxidation occurs in different compartments in different organisms • Here, we will focus on the mitochondrion of animals using saturated fatty acids with an even number of carbons as an energy source © 2012 Pearson Education, Inc. Fatty acid degradation • Most fatty acids are oxidatively converted to acetyl CoA in the mitochondrion • These can be further catabolized in the TCA cycle • The fatty acids are degraded in a series of repetitive cycles, which removes two carbons at a time until the fatty acid is completely degraded © 2012 Pearson Education, Inc. Steps of b-oxidation • Each cycle involves – – – – 1.Oxidation (ATP hydrolysis) 2. Hydration (add water) 3. Reoxidation (NADH production) 4. Thiolysis (conversation to acetyl CoA) • The result is the production of one FADH2, one NADH, and one acetyl CoA per cycle © 2012 Pearson Education, Inc. Protein as a Source of Energy and Amino Acids • Besides their other numerous functions, proteins can be catabolized to produce ATP if necessary when carbohydrate and lipid stores are depleted • Eventually cells undergo turnover of proteins and protein-containing structures • The resulting amino acids can be used to generate new proteins or degraded for energy © 2012 Pearson Education, Inc. Protein catabolism • Protein catabolism begins with hydrolysis of the peptide bonds that link amino acids together • This is called proteolysis and the enzymes responsible for it are called proteases • The products of proteolysis are small peptides and free amino acids © 2012 Pearson Education, Inc. Endopeptidases and exopeptidases • Further digestion of peptides is catalyzed by peptidases • Endopeptidases hydrolyze internal peptide bonds • Exopeptidases remove successive amino acids from the end of the peptide © 2012 Pearson Education, Inc. Amino acid catabolism • Free amino acids can be catabolized for energy • These are converted into intermediates of mainstream catabolism in as few steps as possible • The pathways differ for individual amino acids, but all eventually lead to acetyl CoA, pyruvate, or a few key TCA cycle intermediates © 2012 Pearson Education, Inc. Electron Transport: Electron Flow from Coenzymes to Oxygen • Chemotrophic energy metabolism through the TCA cycle accounts for synthesis of 4 ATP per glucose (2 from glycolysis and 2 from the TCA cycle) • This accounts for only a small portion of the energy in the original glucose molecule • The remainder is stored in NADH and FADH2 © 2012 Pearson Education, Inc. The Electron Transport System Conveys Electrons from Reduced Coenzymes to Oxygen • Coenzyme reoxidation by transfer of electrons to oxygen is called electron transport • Electron transport and ATP generation are not independent processes; they are functionally linked to each other © 2012 Pearson Education, Inc. Electron Transport and Coenzyme Oxidation • Electron transport involves the highly exergonic oxidation of NADH and FADH2 with O2 as the terminal electron acceptor and so accounts for the formation of water • • © 2012 Pearson Education, Inc. The Electron Transport System • Electron transfer is carried out as a multistep process involving an ordered series of reversibly oxidized electron carriers functioning together • This is called the electron transport system, ETS • The ETS contains a number of integral membrane proteins that are found in the inner mitochondrial membrane (or plasma membrane of bacteria) © 2012 Pearson Education, Inc. Focus • What are the major electron carriers? • What is the sequence of these carriers? • Understand the role of the organization of carriers in the flow of electrons from reduced coenzymes to oxygen is coupled to pumping of proton across the membrane and ATP synthesis. © 2012 Pearson Education, Inc. The Electron Transport System Consists of Five Kinds of Carriers • Flavoproteins • Iron-sulfur proteins • Cytochromes • Copper-containing cytochromes • Coenzyme Q © 2012 Pearson Education, Inc. Features of electron carriers • Flavoproteins and coenzyme Q pump protons and electrons. • All except coenzyme Q are proteins with prosthetic groups capable of being reversibly oxidized and reduced – Most are hydrophobic – Most of these intermediates occur in the membrane as large assemblies of proteins called respiratory complexes © 2012 Pearson Education, Inc. Flavoproteins • Membrane-bound flavoproteins use either (FAD) or flavin adenine mononucleotide (FMN) as the prosthetic group – For example, NADH dehyrogenase • What is the function? • Flavoproteins transfer both electrons and protons and are reversibly oxidized and reduced © 2012 Pearson Education, Inc. Figure 10-9 © 2012 Pearson Education, Inc. Iron-Sulfur Proteins • Iron-sulfur proteins are also called nonheme iron proteins and have an iron-sulfur (Fe-S) center complexed with cysteine groups of a protein – The iron atoms in the center of the proteins are the actual electron carriers; these alternate between the Fe2+ (oxidized) and Fe3+ (reduced) states • They transfer one electron at a time, and no protons © 2012 Pearson Education, Inc. Cytochromes • Cytochromes also contain iron, but as part of a porphyrin prosthetic group, heme. – Where else in the body do you fine heme? • There are five types: b, c, c1, a, and a3 – The iron atom of the heme group serves as the electron carrier and transfers one electron at a time and no protons – Cytochrome c is a peripheral membrane protein loosely associated with the outer surface of the membrane. It is not part of a large complex and can diffuse more rapidly • Important for transferring electrons between complexes © 2012 Pearson Education, Inc. Figure 10-13 © 2012 Pearson Education, Inc. Copper-Containing Cytochromes • In addition to their iron atoms, cytochromes a and a3 contain a single copper atom bound to their heme group • It associates with the iron atom to form a bimetallic iron-copper (Fe-Cu) center – Copper ions can be reversibly converted from the oxidized (Cu2+) to the reduced form (Cu+) by accepting or donating single electrons © 2012 Pearson Education, Inc. Copper-Containing Cytochromes (continued) • The iron-copper center plays a critical role in keeping an O2 molecule bound to the cytochrome oxidase complex • The oxygen is held there until it has picked up four electrons and four protons, at which point two water molecules are released © 2012 Pearson Education, Inc. Coenzyme Q • The only nonprotein component of the ETS is coenzyme Q (CoQ), a quinone • Because of its ubiquitous occurrence in nature, it is also called ubiquinone • CoQ is reduced in two successive (one electron plus one proton) steps to semiquinone (CoQH) and then dihydroquinone (CoQH2) © 2012 Pearson Education, Inc. Coenzyme Q (continued) • Unlike the proteins of the ETS, most of the CoQ is freely mobile in the inner mitochondrial membrane • CoQ is the most abundant electron carriers in the membrane and occupy a central position in the ETS – They serve as a collection point for electrons from the reduced FMN and FAD-linked dehydrogenases in the membrane – A portion of the CoQ is tightly bound to specific respiratory complexes © 2012 Pearson Education, Inc. Coenzyme Q (continued) • CoQ accepts both protons and electrons when it is reduced and releases both protons and electrons when it is oxidized • This is vital to its role in the active transport of protons across the inner mitochondrial membrane – It accepts protons on one side of the membrane, diffuses across and releases the protons- proton pump © 2012 Pearson Education, Inc. Most of the Carriers Are Organized into Four Large Respiratory Complexes • Although many electron carriers are part of the ETS, most are organized into multiprotein complexes • Most are thought to be organized into four different kinds of respiratory complexes © 2012 Pearson Education, Inc. Figure 10-15 © 2012 Pearson Education, Inc. Table 10-3 © 2012 Pearson Education, Inc. Properties of the Respiratory Complexes • Each respiratory complex consists of distinctive assembly of polypeptides and prosthetic groups • Complex I transfers electrons from NADH to CoQ and is called the NADH-coenzyme Q oxidation complex (or NADH dehydrogenase complex) © 2012 Pearson Education, Inc. The respiratory complexes • Complex II transfers to CoQ the electrons derived from succinate (FAD) in Reaction TCA-6 and it is called the succinate-coenzyme Q oxidoreductase complex, or succinate dehydrogenase • Complex III is called the coenzyme Q-cytochrome oxidoreductase complex because it accepts electrons from coenzyme Q and passes them to cytochrome c © 2012 Pearson Education, Inc. The respiratory complexes (continued) • Complex III is also called cytochrome b/c1 complex • Complex IV transfers electrons from cytochrome c to oxygen and is called cytochrome c oxidase • For each pair of electrons transported through complexes I through IV, 10 protons are pumped from the matrix to the intermembrane space © 2012 Pearson Education, Inc. The Role of Cytochrome c Oxidase • Cytochrome c oxidase (complex IV) is the terminal oxidase, transferring electrons directly to oxygen • Cyanide and azide are toxic to most aerobic cells because they bind the Fe-Cu center of cytochrome c oxidase, blocking electron transport © 2012 Pearson Education, Inc. Incomplete reduction of oxygen • Complexes I and III can also transfer electrons to oxygen, resulting in its incomplete reduction • This can generate toxic superoxide anion (O2) or hydrogen peroxide (H2O2), both of which contribute to cellular aging © 2012 Pearson Education, Inc. The Respiratory Complexes Move Freely Within the Inner Membrane • The protein complexes of the mitochondrial inner membrane are mobile, free to diffuse within the membrane • The inner membrane has no cholesterol and is very fluid, so the protein mobility is high © 2012 Pearson Education, Inc. Respirasomes • Recent work suggests that the multiprotein respiratory complexes are organized into supercomplexes called respirasomes • The association of several of the TCA cycle dehydrogenases within the respirasomes suggests that they function to minimize diffusion distances © 2012 Pearson Education, Inc. Coenzyme Q and cytochrome c • Coenzyme Q is the “funnel” that collects electrons from virtually every oxidation reaction in the cell • Coenzyme Q and cytochrome c are both small molecules that can diffuse rapidly within the membrane (coenzyme Q) or on its surface (cytochrome c) • They are both quite numerous, which accounts for observed rates of electron transfer © 2012 Pearson Education, Inc. Electron Transport and ATP Synthesis Are Coupled Events • The crucial link between electron transport and ATP production is an electrochemical proton gradient • It is established by the directional pumping of protons across the membrane in which electron transport is occurring • ATP synthesis is coupled to electron transport © 2012 Pearson Education, Inc. ATP synthesis is dependent on electron transport • Certain chemicals known as uncouplers can abolish the interdependence of the two processes • These allow continued electron transport and O2 consumption in the absence of ATP synthesis • Treatments that stop electron transport also inhibit ATP synthesis, so ATP synthesis is dependent on electron transport but the reverse is not true © 2012 Pearson Education, Inc. Respiratory Control of Electron Transport • The availability of ADP regulates the rate of oxidative phosphorylation and thus of electron transport • This is called respiratory control • Electron transport and ATP generation will be favored when ADP concentration is high and inhibited when ADP concentration is low © 2012 Pearson Education, Inc. The Chemiosmotic Model: The “Missing Link” Is a Proton Gradient • In 1961 Peter Mitchell proposed the chemiosmotic coupling model • The essential feature of the model is that the link between electron transport and ATP formation is the electrochemical potential across a membrane • The electrochemical potential is created by the pumping of protons across a membrane as electrons are transferred through the respiratory complexes © 2012 Pearson Education, Inc. Coenzyme Oxidation Pumps Enough Protons to Form 3 ATP per NADH and 2 ATP per FADH2 • The transfer of two electrons from NADH is accompanied by the pumping of a total of 10 protons (12 if the Q cycle is operating) • The number of protons required per molecule of ATP is thought to be 3 or 4, with 3 regarded as most likely • So, about 3 molecules of ATP are synthesized per NADH oxidized © 2012 Pearson Education, Inc. Number of ATP generated is an estimate • FADH2 donates electrons to complex II with higher reduction potential, pumping 6 protons (8 if the Q cycle is operating) • So about 2 ATP are synthesized per FADH2 • These values are estimates, affected by an organism’s specific ATP synthase and other factor © 2012 Pearson Education, Inc. The Chemiosmotic Model Is Affirmed by an Impressive Array of Evidence • Since its initial formulation, the chemiosmotic model has become universally accepted as the link between electron transport and ATP synthesis • Several lines of evidence support the mode © 2012 Pearson Education, Inc. 1. Electron Transport Causes Protons to Be Pumped Out of the Mitochondrial Matrix • Mitchell and Moyle demonstrated experimentally that the flow of electrons through the ETS is accompanied by the unidirectional pumping of protons across the inner mitochondrial membrane © 2012 Pearson Education, Inc. 5. Uncoupling Agents Abolish Both the Proton Gradient and ATP Synthesis • Dinitrophenol (DNP) is known to uncouple ATP synthesis from electron transport • When membranes are treated with DNP, they allow protons to cross the membrane freely, so that no proton gradient can be formed • ATP synthesis is abolished as well © 2012 Pearson Education, Inc. 6. The Proton Gradient Has Enough Energy to Drive ATP Synthesis • The electrochemical proton gradient across the membrane involves both a membrane potential and a concentration gradient • A mitochondrion actively respiring has a membrane potential of about 0.16V (positive on the intermembrane space side) and a pH gradient of about 1.0 (higher on the matrix side) © 2012 Pearson Education, Inc. Proton motive force • The electrochemical gradient exerts a proton motive force (pmf), that tends to drive protons back down their concentration gradient (back into the matrix) • © 2012 Pearson Education, Inc. ATP Synthesis: Putting It All Together • Some of the energy of glucose is transferred to reduced coenzymes during glycolysis and the TCA cycle • This energy is used to generate an electrochemical proton gradient across the inner mitochondrial membrane • The pmf of that gradient is harnessed to make ATP © 2012 Pearson Education, Inc. Uncoupling synthesis of ATP and electron transport • F1 particles and membranous vesicles were separated by centrifugation • The membranes could still carry out electron transport, but could not synthesize ATP • The isolated F1 particles could synthesize ATP but could not carry out electron transport © 2012 Pearson Education, Inc. The FoF1 Complex: Proton Translocation Through Fo Drives ATP Synthesis by F1 • The F1 complex is not directly membranebound, but is attached to the Fo complex that is embedded in the inner membrane • Fo acts as a proton translocator, the channel through which protons flow across the membrane © 2012 Pearson Education, Inc. The FoF1 ATP synthase • Fo provides a channel for exergonic flow of protons across the membrane • F1 carries out the ATP synthesis, driven by the energy of the proton gradient • Together, they form a complete ATP synthase © 2012 Pearson Education, Inc. Video: Rotation of ATP Synthase © 2012 Pearson Education, Inc. Rotation of a and b with respect to g • Boyer proposed that at any time, each of the active sites is in a different conformation and the hexagonal ring of a and b subunits rotates relative to the central stalk containing the g subunit • The rotation was thought to be driven by the flow of protons through Fo • It is now known that it is the g subunit that actually rotates © 2012 Pearson Education, Inc. The Chemiosmotic Model Involves Dynamic Transmembrane Proton Traffic • There is continuous, dynamic two-way proton traffic across the inner membrane • NADH sends 10 protons across via complexes I, III, and IV; FADH2 sends 6 across, via complexes II, III, and IV • Assuming that 3 protons must return through FoF1 per ATP generated, this means 3 ATP per NADH and 2 per FADH2 are generated © 2012 Pearson Education, Inc. Aerobic Respiration: Summing It All Up • As carbohydrates and fats are oxidized to generate energy, coenzymes are reduced • These reduced coenzymes represent a storage form of the energy released during oxidation • This energy can be used to drive ATP synthesis as the enzymes are reoxidized by the ETS © 2012 Pearson Education, Inc. Summing it all up (continued) • As electrons are transported from NADH or FADH2 to O2, they pass through respiratory complexes where proton pumping is coupled to electron transport • The resulting electrochemical gradient exerts a pmf that serves as the driving force for ATP synthesis © 2012 Pearson Education, Inc. Video: ATP Synthase 3-D Structure © 2012 Pearson Education, Inc. The Maximum Yield of Aerobic Respiration Is 38 ATPs per glucose • The maximum ATP yield per glucose under aerobic conditions: • Including the summary reactions of glycolysis and the TCA cycle to this gives: © 2012 Pearson Education, Inc. 1. Why Does the Maximum ATP Yield in Eukaryotic Cells Vary Between 36 and 38 ATPs Per Glucose? • Glycolysis produces two NADH per glucose in the cytosol, and catabolism of pyruvate produces eight more in the mitochondrial matrix • NADH in the cytosol cannot enter the matrix to deliver its electrons to complex I • Instead the electrons and H+ ions are passed inward by an electron shuttle system © 2012 Pearson Education, Inc. Aerobic Respiration Is a Highly Efficient Process • To determine efficiency of respiration, we need to determine how much of the energy of glucose is preserved in the resulting 36–38 ATP • DGo for glucose CO2 + H2O is 686 kcal/mol • ATP hydrolysis under cellular conditions is about 10 to 14 kcal/mol © 2012 Pearson Education, Inc. Efficiency of aerobic respiration • For 36–38 ATP, assuming a value of 10 kcal/mol, the energy per mole of glucose is about 360–380 kcal conserved • This efficiency of 52–55% is well above that obtainable from the most efficient machines created © 2012 Pearson Education, Inc.