* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Chapter 7- Alcohols
Elias James Corey wikipedia , lookup
Physical organic chemistry wikipedia , lookup
George S. Hammond wikipedia , lookup
Asymmetric induction wikipedia , lookup
Baylis–Hillman reaction wikipedia , lookup
Stille reaction wikipedia , lookup
Tiffeneau–Demjanov rearrangement wikipedia , lookup
Kinetic resolution wikipedia , lookup
Wolff–Kishner reduction wikipedia , lookup
Hofmann–Löffler reaction wikipedia , lookup
Petasis reaction wikipedia , lookup
Hydroformylation wikipedia , lookup
Nucleophilic acyl substitution wikipedia , lookup
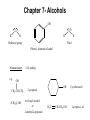
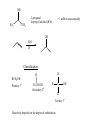
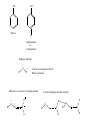
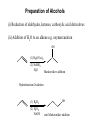
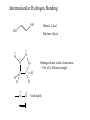
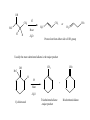
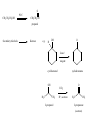
Chapter 7- Alcohols OH O R S H R H Thiol Hydroxyl group Phenol- Aromatic alcohol Nomenclature e.g. - OL ending OH OH CH3-CH-CH3 (CH3)3COH Cyclohexanol 2-propanol tert-butyl alcohol or 2-methyl-2-propanol H2C CH-CH2-OH 2-propen-1-ol Alcohols- commercially important Ethanol CH3CH2OH - Produced by fermentation or hydration of ethylene CH2=CH2 + H2O H2SO4 CH3-CH2OH Commercial EtOH 95% H2O 5% Can’t purify further by distillation Absolute alcohol use CaO To remove water CaO + H2O Ca(OH)2 Ethanol- Solvent, fuel, antiseptic, reagent OH H3C CH3 2-propanol Isopropyl alcohol (IPA) > 1 million tons annually OH H2O H+ Classification R R RCH2OH Primary 1o R-CH-OH Secondary 2o R C OH R Tertiary 3o Reactivity depends on the degree of substitution OH OH Phenol Cl 4-chlorophenol or p-chlorophenol Hydrogen bonding O R H O more electronegative than H. Bond is polarised Molecules can interact via hydrogen bonds R H H H H O O O O Can form hydrogen bonded networks R R H R O R H O R Covalent OH bond ~ 480 kJ mol-1 Hydrogen bond ~ 20-40 kJ mol-1 Much weaker but has important effects (i) Boiling points of alcohols are higher than would be predicted based on molecular weight - Extra energy required to break the intermolecular hydrogen bonds (ii) Lower molecular weight alcohols are soluble in water CH3CH2OH MW 46 B.P. +78.5oC CH3OCH3 46 -24oC CH3CH2CH3 44 -42oC Preparation of Alcohols (i) Reduction of aldehydes, ketones, carboxylic acid derivatives (ii) Addition of H2O to an alkene e.g. oxymercuration OH (1) Hg(OAc)2 (2) NaBH4 H2O Markovnikov addition Hydroboration-Oxidation (1) B2H6 (2) H2O2 NaOH OH anti-Markovnikov addition Intermolecular Hydrogen Bonding OH Ethan-1,2-diol HO Ethylene Gylcol H H O C H O C H Hydrogen bond- weak, electrostatic ~ 10% of O-H bond strength H H O H bond dipole Alcohols are weak acids ROH + B: base RO + BH alkoxide e.g CH3CH2OH + NaH CH3CH2OH Na (CH3)3COH + K CH3CH2O Na + H2(g) CH3CH2O Na + 1/2 H2(g) (CH3)3CO K + 1/2 H2(g) potassium tert-butoxide pKa MeOH/EtOH ~16 Phenol ~10 (more acidic) 7.8 Dehydration of alcohols- alkenes - Occurs by heating with a strong acid H + H2C CH2 HC CH H OH + H2O o 180 C H OH Example of an elimination reaction - can be used as a route to alkenes OH CH3 H H2SO4 + H2O Heat H3C C OH CH3 Cyclohexanol H2C C + CH3 2-methylpropene Cyclohexene Fastest 3o > 2o > 1o CH3 H+ Slowest Rate affected by substitution H2O OH H+ CH3 H3C Heat H H -H2O CH3 or H3C CH3 H2C Proton lost from either side of OH group Usually the more substituted alkene is the major product H3C CH2 CH3 OH H H+ + Heat -H2O Cyclohexanol Trisubstituted alkene - major product Disubstituted alkene Mechanism of dehydration Protonation of hydroxyl group occurs first This converts the OH group into a good leaving group (H2O) H+ CH3-CH2-OH H CH3-CH2-O H H O H2C H H2O C H2 H CH2=CH2 + H2O + H3O+ 7.9 Reaction of Alcohols with Hydrogen Halides Example of a substituted reaction (OH replaced by X) Good synthetic route to alkyl halides Rate depends on degree of substitution 3o > 2o > 1o R-OH + H-X R-X + H2O alcohol alkyl halide Cl, Br, I CH3 CH3 e.g. H3C C OH CH3 t-butyl alcohol + HCl H3C C Cl CH3 t-butyl chloride + H2O Reaction of 1o alcohols with HCl can be very slow. To increase rate add a lewis acid catalyst such as zinc chloride (ZnCl2) CH3CH2CH2CH2OH + HCl ZnCl 2 Heat CH3CH2CH2CH2-Cl + H2O 1-chlorobutane There are special reagents which can also be employed e.g. thionyl chloride (SOCl2) or phosphorus tri-bromide (PBr3) O CH3CH2OH + Cl-S-Cl CH3CH2-Cl + HCl + SO2 By-products are gases- Pure alkyl chloride formed OH 3 Br + PBr3 + H3PO3 3 (or PCl3) Usually use SOCl2 or PBr3 with 1o or 2o alcohols- reaction of 3o alcohols with H-X is so fast these reagents are unnecessary 7.12 Alcohol Oxidation Primary Alcohols R-CH2OH oxidising O R-C-H aldehyde agent oxidising agent O R-C-OH carboxylic acid Typical oxidising agents: (1) CrO3, H+ (H2SO4), acetone- Jones' reagent (2) KMnO4 Potassium permanganate With these strong oxidising agents it is not possible to isoalte the aldehyde - only the carboxylic acid is obtained Jones' CH3CH2CH2OH CH3CH2CO2H reagent propanoic acid There are special reagents which allow oxidation to the aldehyde level with no further oxidation to the carboxylic acid e.g. Pyridinium chlorochromate (PCC) CrO3 + HCl + N pyridine N H CrO3 Cl O CH3CH2CH2OH PCC CH3CH2CH propanal Secondary Alcohols Ketones e.g. H OH O Jones' reagent cyclohexanol cyclohexanone OH O CrO3 H3C CH3 2-propanol H+, acetone H3C CH3 2-propanone (acetone) Tertiary Alcohols- Not oxidised under usual conditions for alcohol oxidation CH3 Jones' H3C C OH no reaction reagent CH3 No H on carbon which can be removed Thiols: S (sulfur) analogues of alcohols e.g. SH CH3-S-H methanethiol thiophenol Thiols are more acidic than the alcohols RS Na + H2O R-S-H + NaOH pKa ~ 10 Sodium thiolate Thiolate anions - good nucleophiles e.g. CH3CH2-Br + CH3S Na CH3CH2-S-CH3 + NaBr methylethyl sulfide