* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download medical necessity letter
Behavioural genetics wikipedia , lookup
Cancer epigenetics wikipedia , lookup
Designer baby wikipedia , lookup
DNA paternity testing wikipedia , lookup
Population genetics wikipedia , lookup
Nutriepigenomics wikipedia , lookup
Microevolution wikipedia , lookup
Pharmacogenomics wikipedia , lookup
Medical genetics wikipedia , lookup
Public health genomics wikipedia , lookup
BRCA mutation wikipedia , lookup
Genetic testing wikipedia , lookup
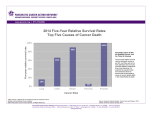
LETTER OF MEDICAL NECESSITY FOR HEREDITARY PANCREATIC CANCER GENETIC TESTING Date: Date of service/claim To: Utilization Review Department Insurance Company Name, Address, City, State Re: Patient Name, DOB, ID # ICD-9 Codes: (list codes) This letter is in regards to my patient and your subscriber, First, Last Name, to request full coverage for medically-indicated hereditary pancreatic cancer genetic testing to be performed by Ambry Genetics Corporation (TIN 33-0892453 / NPI 1861568784), a CAP-approved and CLIA-certified laboratory located at 15 Argonaut, Aliso Viejo, CA 92656. Pancreatic cancer is thought to have a familial component in approximately 10% of cases; evaluating personal and family histories is a major part of this assessment. Significant aspects of my patient’s personal and/or family medical history that suggest an inherited predisposition to pancreatic cancer are below: Based on the above, along with the PancPRO probability estimate of XXX, this history is suggestive of hereditary pancreatic cancer and warrants germline genetic testing. There are many genes known to predispose an individual to pancreatic cancer, and the ordered genetic test analyzes 13 clinically relevant genes for this: APC, ATM, BRCA1, BRCA2, CDKN2A, EPCAM, MLH1, MSH2, MSH6, PALB2, PMS2, STK11, and TP53. These genes substantially increase the risk for pancreatic and other cancers. Given the substantial clinical overlap between many of these genes, this multi-gene test is the most efficient and cost-effective way to make a definitive genetic diagnosis. The results of this test will help clarify my patient’s risk for pancreatic and other cancers and will directly impact my patient’s care and management. Mutations in tested genes will specify different medical management based on clinical guidelines. For example: Mutations in MLH1, MHS2, MSH6, PMS2, and EPCAM cause Lynch syndrome, a hereditary colorectal cancer syndrome. Those with Lynch syndrome have up to a 9- to 11-fold increased risk to develop pancreatic cancer (NCCN, 2014). Specific high-risk screening guidelines exist for Lynch syndrome, which could then be implemented. Mutations in STK11 cause Peutz-Jeghers syndrome, which includes an increased lifetime risk for pancreatic cancer. This risk is as much as 132-fold higher than usual (NCCN, 2014). Mutations in CDKN2A are associated with melanoma-pancreatic cancer syndrome, which includes a 20-fold to 47-fold increased lifetime risk for pancreatic cancer (NCCN, 2014). This may develop at earlier ages than in the general population. Aggressive pancreatic cancer screening measures could be implemented for those with STK11 and CDKN2A mutations. If my patient is found to carry a mutation in one of the genes included in this test, an aggressive approach to reduce the risk of cancer would be recommended to reduce the patient’s chance of developing and subsequently dying of an advanced stage cancer. Screening modalities for pancreatic cancer include endoscopic ultrasound and/or MRI/MRCP. Depending on the gene involved, risk reduction measures for other cancer types may also be recommended. As such, I am ordering this genetic test as medically necessary care, and affirm that the patient has provided informed consent for genetic testing. I am specifying Ambry Genetics Corporation because this laboratory has highly sensitive and cost-effective testing for hereditary pancreatic cancer, along with a large database of tested patients to ensure the most validated, accurate, and informative test interpretation. I recommend that you support this request for coverage of diagnostic genetic testing for hereditary pancreatic cancer for my patient. Genetic testing can take up to four months to complete and the laboratory will not bill until testing is concluded. Therefore, we are requesting that the authorization be valid for 6 months. Thank you for your time and please don’t hesitate to contact me with any questions. Sincerely, Ordering Clinician Name (Signature Provided on Test Requisition Form) (MD/DO, Clinical Nurse Specialist, Nurse-Midwives, Nurse Practitioner, Physician Assistant, Genetic Counselor*) *Authorized clinician requirements vary by state Test Details CPT codes: 81201x1, 81211x1, 81213x1, 81292x1, 81294x1, 81295x1, 81297x1, 81298x1, 81300x1, 81317x1, 81319x1 Laboratory: Ambry Genetics Corporation (TIN 33-0892453 / NPI 1861568784), a CAP-accredited and CLIA-certified laboratory located at 15 Argonaut, Aliso Viejo, CA 92656 Reference: National Comprehensive Cancer Network Clinical Practice Guidelines in Oncology (NCCN Guidelines®), “Pancreatic Adenocarcinoma,” Version 2.2014.