* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download MACROPHAGE - immunology.unideb.hu
Survey
Document related concepts
Gluten immunochemistry wikipedia , lookup
Hygiene hypothesis wikipedia , lookup
Herd immunity wikipedia , lookup
Molecular mimicry wikipedia , lookup
Cancer immunotherapy wikipedia , lookup
Adoptive cell transfer wikipedia , lookup
Social immunity wikipedia , lookup
Complement system wikipedia , lookup
Immune system wikipedia , lookup
Adaptive immune system wikipedia , lookup
Psychoneuroimmunology wikipedia , lookup
Polyclonal B cell response wikipedia , lookup
Transcript
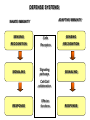
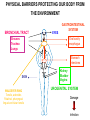
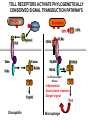
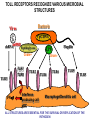
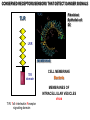
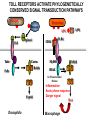
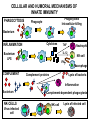
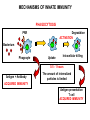
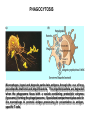
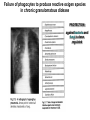
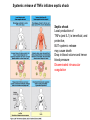
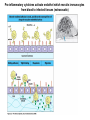
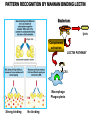
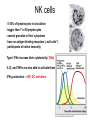
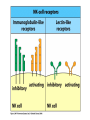
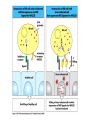
THE TWO „ARMS” OF THE IMMUNE SYSTEM • INNATE/NATURAL IMMUNITY • ACQUIRED IMMUNITY WHY IS THE IMMUNE SYSTEM SO IMPORTANT? Biomass: 90% microbes Animal mass PATHOGENS < 5 – 25x microbes Virus Viruses Bacteria Monocellular parazites 3 hours 3 hours 18 - 30 years Multicellular parazites (helminths) VARIABILITY Rapid evolution Adaptation Selection CHARACTERISTICS OF INNATE IMMUNITY • • • • • • NATURAL/INNATE Rapid, prompt response (hours) No variable receptors No improvement during the response No memory Not transferable Can be exhausted, saturated • • • • • • ADAPTIVE/ACQUIRED Time consuming (several days) Variable antigen receptors Efficacy is improving during the response Memory Can be transferred Regulated, limited COMMON EFFECTOR MECHANISMS FOR THE ELIMINATION OF PATHOGENS TWO LINES OF IMMUNE DEFENSE INNATE/NATURAL IMMUNITY ACQUIRED/ADAPTIVE IMMUNITY Phagocytes (monocyte/macrophage, neutrophil, dendritic cell) Killer cells (NK cell, δ T cell) B lymphocytes (B2) T lymphocytes CELLS helper T cell cytotoxic T cell B1 lymphocytes (CD5+) Enzymes (lysozyme, pepsin, trypsin) Antibacterial peptides HUMORAL Complement system FACTORS Cytokines, chemokines Antibodies DEFENSE SYSTEMS ADAPTIVE IMMUNITY INNATE IMMUNITY SENSING Cells SENSING RECOGNITION Receptors RECOGNITION SIGNALING Signaling pathways SIGNALING Cell-Cell collaboration RESPONSE Effector functions RESPONSE PHYSICAL BARRIERS PROTECTING OUR BODY FROM THE ENVIRONMENT BRONCHIAL TRACT EYES GASTROINTESTINAL SYSTEM Sinuses Trachea Lungs Oral cavity esophagus Stomach Intestines SKIN WALDEYER RING Tonsils, adenoids Palatinal, pharyngeal lingual and tubar tonsils Kidney Bladder Vagina UROGENITAL SYSTEM Damage Infection EPITELIAL SURFACES ARE IMPORTANT IN THE FIRST LINE OF DEFENSE α2-macroglobulin inhibits potentially damaging proteases About 10% of serum proteins are protease inhibitors. Human defensins are variable antimicrobial peptides Peptides of 30-40 amino acids, amphipathic character They penetrate microbial membranes Ongoing race between pathogens and the immune system of the host Normal flora Cells of human body: 90% microbes, 10% human Symbiotic, non-pathogenic microbes – mucosal membrane, skin Bacteria, Fungi, Protozoa Gut – colonalization after birth 1012 bakteria/g (1.5 kg) intestinal content 1000 species 100-times more bacterial genes then eukaryotic „peaceful” commensalisms vitamins (i.e. K1 vitamin) real ecosystem, survival of the fittest, competition with pathogenic organism the few who brake in through the gut epithelium induce local immune response Important role in: - development of mucosal and systemic immunity - normal development of peripheral lymphoid organs - maintenance of basic level of immunity RECOGNITION BY THE INNATE IMMUNE SYSTEM INNATE/NATURAL IMMUNITY RECOGNITION Richard Pfeiffer, a student of Robert Koch – ENDOTOXIN There must be a receptor that recognizes endotoxin Lipopolysaccharide (LPS) receptor remained elusive The Dorsoventral Regulatory Gene Cassette Spätzle/Toll/Cactus controls the potent antifungal response in Drosophila adults Bruno Lemaitre, A Hoffmann et al, Cell, 1996 Spätzle: Toll ligand Toll: Receptor Cactus: I-kB Dorsal: NF-kB Drosomycin TOLL RECEPTORS ACTIVATE PHYLOGENETICALLY CONSERVED SIGNAL TRANSDUCTION PATHWAYS Fungus Bacterium Protease LPB LPS Toll Tube Spätzel CD14 Cactus Relish Pelle TLR4 MyD88 NFkB IRAK IL-1R associated Kinase Peptid Drosophila Inflammation Acute phase response Danger signal IL-6 Macrophage WHAT IS RECOGNIZED BY INNATE AND ACQUIRED IMMUNITY? RECEPTORS Common pattern of groups of pathogens Pathogen Associated Molecular Pattern PAMP Recognition by receptors Pattern Recognition Receptor PRR 9-13 various Toll-receptors TLR family Innate immunity Ancient Unique structural elements Antigenic determinant Recognition by highly specific antigen receptors B cell receptor BCR (sIg) T cell receptor TCR Several millions antigen receptors Acquired immunity 450 million years TOLL RECEPTORS RECOGNIZE VARIOUS MICROBIAL STRUCTURES Bacteria Virus CpG DNA ssRNS dsRNA Peptidoglycane Gram+ TLR3 IFN TLR7 TLR8 TLR2 Interferon producing cell pDC Flagellin LPS Gram- TLR4 TLR6 TLR9 TLR5 Macrophage/Dendritic cell ALL STRUCTURES ARE ESSENTIAL FOR THE SURVIVAL OR REPLICATION OF THE PATHOGEN CONSERVED RECEPTORS/SENSORS THAT DETECT DANGER SIGNALS TLR3 Fibroblast Epithelial cell DC TLR LRR MEMBRANE TIR domain CELL MEMBRANE Bacteria MEMBRANES OF INTRACELLULAR VESICLES virus TIR: Toll-Interleukin Receptor signaling domain PHAGOCYTES ARE ABLE TO RECOGNIZE PATHOGENS Toll receptormediated signaling FcR, CR Toll receptor PHAGOCYTES (macrophages, dendritic cells, neutrophil granulocytes) RECOGNIZE PATHOGENS BY PATTERN RECOGNITION RECEPTORS RECOGNITION IS ESSENTIAL RECOGNITION CYTOPLASMIC SENSORS VESZÉLYT ÉRZÉKELŐ KONZERVÁLT RECEPTOROK NLR: NOD-like receptor RLR: RIG-like receptor CONSERVED RECEPTORS SENSING DANGER SIGNALS NLR nod-like receptors Nucleotide binding domain Leucin rich repeats TLR N C NBD NLRP1 – ASC NLRP3 – ASC – CARDINAL PYR NBD NOD1/2, IPAF/NLRC4 CARD NBD IPAF BIR TLR3 Fibroblast Epithelial cell DC CYTOPLASM RLH CARD-CARD-helicase MEMBRAN DANGER SIGNALS ARE TRANSLATED TO CYTOKINE SECRETION THROUGH VARIOUS MOLECULAR SENSORS IN DC SUBTYPES 4 2 1 5 6 6 3 1 7 NLR 7 9 10 8 RLH RLH NLR=NOD/NALP (IL-1β) RLH=RIG-1/MDA5 (IFN) Conventional DC TLR1 – TLR2 – Plasmacytoid DC bacterial lipoprotein (together with TLR2) bacterial lipoprotein, peptidoglycane, lipoteicholic acid IL-1β (heteromer with TLR1 and TLR6) IL-12/23 TLR3 – viral dsRNS, polyI:C IL-10 TLR4 – bacterial LPS TLR5 – bacterial flagellin TLR6 – bacterial lipoprotein (with TLR2) TLR7 – viral ssRNA TLR8 – GU rich viral ssRNS, imidazoquinolin (antiviral drug) TLR9 – unmethylated CpG DNA Th1/Th17/Th2 TLR10 – modified viral nucleotides IFNαβ NK/DC SIGNALING IN INNATE IMMUNITY TOLL RECEPTORS ACTIVATE PHYLOGENETICALLY CONSERVED SIGNAL TRANSDUCTION PATHWAYS Fungus Bacterium Protease LPB LPS Toll Tube Spätzel CD14 Cactus Relish Pelle TLR4 MyD88 NFkB IRAK IL-1R associated Kinase Peptid Drosophila Inflammation Acute phase response Danger signal IL-6 Macrophage TOLL RECEPTOR MEDIATED SIGNALLING NEW THERAPEUTIC TARGET Figure 3 The 'hourglass' shape of the innate immune response. Although microbial stimuli are chemically complex and although the innate immune response ultimately involves the activation of thousands of host genes, innate immune signals traverse a channel of low complexity. Ten Toll-like receptors (TLRs), four TIR (Toll/interleukin-1 receptor homologous region) adaptors and two protein kinases are required for most microbial perception. This circumstance lends itself to effective pharmacotherapeutic intervention. NF-B, nuclear factor-B; STAT1, signal transducer and activator of transcription 1. EFFECTOR MECHANISMS OF INNATE IMMUNITY CELLULAR AND HUMORAL MECHANISMS OF INNATE IMMUNITY PHAGOCYTOSIS Phagocytosis Intracellular killing Phagocyte Bacterium INFLAMMATION Cytokines IL-12 Bacterium LPS COMPLEMENT TNF IFN Complement proteins Neutrophil NK-cell Macrophage Lysis of bacteria Inflammation Bacterium NK-CELLS Virus-infected cell Complement-dependent phagocytosis NK-cell Lysis of infected cell MECHANISMS OF INNATE IMMUNITY PHAGOCYTOSIS PRR Degradation ACTIVATION Bacterium Phagocyte Uptake Intracellular killing 0.5 - 1 hours Antigen + Antibody The amount of internalized particles is limited ACQUIRED IMMUNITY Antigen presentation T cell ACQUIRED IMMUNITY PHAGOCYTE SYSTEM NEUTROPHIL GRANULOCYTE MONOCYTE – MACROPHAGE – DENDRITIC CELL Gatekeeper function Sensing commensals and pathogens Rapid activation of innate immunity Priming adaptive immune responses Maintenance of self tolerance Defence against infectious diseases Elimination of tumor cells PHAGOCYTOSIS Macrophages ingest and degrade particulate antigens through the use of long pseudopodia that bind and engulf bacteria. The engulfed bacteria are degraded when the phagosome fuses with a vesicle containing proteolytic enzymes (lysosome), forming the phagolysosome. Specialized compartments also exist in the macrophage to promote antigen processing for presentation to antigenspecific T cells. Opsonization enhances the efficiency of phagocytosis of pathogens by phagocytes Killing of bacteria by neutrophils: azurophilic and specific granules azurofil ic Lyzozyme Defensins Mieloperoxidase Cathepsin G elastase specific granuls NADPH oxidase Lyzozyme Phagocyte oxidase (Phox) produces reactive oxidative species (ROS) that help destroy pathogens Failure of phagocytes to produce reactive oxigen species in chronic granulomatous didease PROTECTION against bacteria and fungi is down regulated MECHANISMS OF INNATE IMMUNITY INFLAMMATION – ACUTE PHASE RESPONSE PRR TNF- neutrophil LPS IL-12 DANGER SIGNAL ACTIVATION IFN Few hours LPS (endotoxin) (Gram(-) bacteria) ACUTE PHASE RESPONSE Kinetics of the release of proinflammatory citokines in bacterial infection macrophage cytokines TNF- IL-1 Plasma level Bacterium NK-cell TNF- IL-1 IL-6 IL-6 1 2 3 4 5 hrs INFLAMMATORY RESPONSE The classic symptoms of inflammation: redness (rubor) - vasodilation, swelling (tumor) - edema, heat (calor) – increased perfusion, pain (dolor) – factors stimulating nociceptors, loss of function (functio laesa) CONSEQUENCES OF MACROPHAGE ACTIVATION SYNTHESIS OF CYTOKINES Systemic effects of pro-inflammatory cytokines Systemic release of TNFa initiates septic shock Septic shock Local production of TNFα (and IL1) is beneficial, and protective, BUT systemic release may cause death Drop in blood volume and hence blood pressure Disseminated intrvascular coagulation Pro-inflammatory cytokines activate endothel which recruits immunocytes from blood to infected tissues (extravasatio) THE ACUTE PHASE RESPONSE IL- 6 C-reactive protein Phosphocolin binding (e.g.fungi) COMPLEMENT Mannose binding lectin/protein MBL/MBP COMPLEMENT Liver Phosphocoline binding Fungi, bacterial Cell wall. Fibrinogen Serum Amyloid Protein (SAP) Mannose/galactose binding IL-6 induces the production of acute phase protiens MECHANISMS OF INNATE IMMUNITY COMPLEMENT ACTIVATION COMPLEMENT Complement-proteins Lysis of bacteria Inflammation Chemotaxis Bacterium Lectin pathway Alternative pathway Complement-dependent phagocytosis Antigen + Antibody Few minutes – 1 hour ACQUIRED IMMUNITY Enzymes get fragmented, complement activity can be exhausted RECOGNITION BY SOLUBLE MOLECULES MANNOSE BINDING LECTIN GLYCOSYLATION OF PROTEINS IS DIFFERENT IN VARIOUS SPECIES Prokariotic cells Eukariotic cells Mannose Glucoseamin Mannose Galactose Neuraminic acid PATTERN RECOGNITION BY MANNAN BINDING LECTIN Bacterium lysis Complement activation LECTIN PATHWAY CR3 Macrophage Phagocytosis Strong binding No binding NK cells - 5-10% of lymphocytes in circulation - bigger than T or B lymphocytes - several granules in their cytoplasm - have no antigen binding receptors („null cells”) - participants of native immunity Type I IFNs increase their cytotoxicity (100x) IL12, and TNFα are also able to activate them IFNγ production --- MF, DC activation