* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download O 2
IUPAC nomenclature of inorganic chemistry 2005 wikipedia , lookup
Artificial gene synthesis wikipedia , lookup
Calcium looping wikipedia , lookup
Electrochemistry wikipedia , lookup
Biochemistry wikipedia , lookup
Inorganic chemistry wikipedia , lookup
Marcus theory wikipedia , lookup
Rate equation wikipedia , lookup
Radical (chemistry) wikipedia , lookup
Supramolecular catalysis wikipedia , lookup
Photoredox catalysis wikipedia , lookup
Multi-state modeling of biomolecules wikipedia , lookup
Photosynthesis wikipedia , lookup
Asymmetric induction wikipedia , lookup
George S. Hammond wikipedia , lookup
Organic chemistry wikipedia , lookup
Physical organic chemistry wikipedia , lookup
Transition state theory wikipedia , lookup
Discodermolide wikipedia , lookup
Photosynthetic reaction centre wikipedia , lookup
Process chemistry wikipedia , lookup
Enantioselective synthesis wikipedia , lookup
Ring-closing metathesis wikipedia , lookup
Bioorthogonal chemistry wikipedia , lookup
Chemical thermodynamics wikipedia , lookup
Hydrogen-bond catalysis wikipedia , lookup
Chemical reaction wikipedia , lookup
Stoichiometry wikipedia , lookup
Lewis acid catalysis wikipedia , lookup
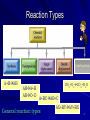
Synthesis, Decomposition & Combustion Mr. Shields Regents Chemistry U11 L05 1 Reaction Types Chemical reactions occur all around us everyday both in industry and in the home. Unimaginable numbers of these unique and specific reactions exist. With so many reactions occurring in our lives the Chemist needs a way to help organize them into some sort of manageable scheme. There are 5 general types of chemical reactions that Make up this scheme 2 Reaction Types Combustion A+BAB ABA+B ABC+D CH4+O2CO2+H2O A+BCAB+C General reaction types AX+BYAY+BX 3 Synthesis Synthesis is also known as direct combination; it is the combining together of several elements and/or compounds into one new compound. Synthesis takes the general format: + A+BAB One product There can also be more + than 2 elements and/or compounds That combine together. For example…. A+B+CABC One product 4 Synthesis Here’s some examples of synthesis: 2Fe + 3O2 2Fe2O3 (red Ochre; rust) 2Mg + F2 2MgF2 C + Cl2 + F2 CCl2F2 (Freon) 2H2O + Cl2 2H2CLO (hypochlorus acid) 2H2 + O2 2H20 In many of these reactions heat and light may also be released. 5 Decomposition What is a Decomposition reaction? A breaking apart of a larger molecule into several smaller molecules and/or elements ABC+D ABA+B Ex: H2CO3 CO2 + H20 Ex: HgO Hg + 02 Can you think of an example where Decomposition reactions are extensively utilized? 6 Decomposition Examples 1. Explosions (the rapid evolution of gases and heat) 2. H2O2 Decomposition when poured on a wound - The bubbles are O2 2H2O2 2H20 + O2 (catalyzed by the Fe in hemoglobin) 3. Another example is the decomposition of limestone when heated CaCO3 + heat CaO + CO2 (g) 7 Decomposition Examples 4. Another example of a decomposition reaction is one Involving Mercury (II) sulfide. It is used to produce liquid mercury. 2HgS + heat 2Hg + S (cinnabar) So what’s common in all these decomposition reactions? A more complicated molecule breaks down into several Simpler molecules and/or elements 8 COMBUSTION CH4+O2 CO2+ H2O + HEAT & LIGHT 9 Combustion (definition): A chemical process that involves the reaction of an Organic Compound with OXYGEN producing several Specific substances as well as heat and light AN UNCONTROLLED COMBUSTION REACTION Bitterroot Montana - 2000 10 Complete combustion reaction takes the form C2H6 + O2 CO2 + H2O Examples of reactions that looks like Combustion but is not… Reactions such as these Will often release heat 2Mg + O2 2MgO And light 4Fe + 3O2 2Fe2O3 4Na + O2 2Na2O Mg H2CO3 CO2 + H2O + O2 The 1st three are direct synthesis Because there is just one product Formed. The last is decomposition. Heat + Light NOT COMBUSTION 11 Typically, COMBUSTION is used to describe the Reaction of O2 and some Organic compound. When there is Sufficient O2 and the organic compound is a simple hydrocarbon there will be COMPLETE COMBUSTION The reaction products will be just H20 and CO2 If there is insufficient Oxygen present then there is INCOMPLETE COMBUSTION and the products are more varied. 12 Ex: * Incomplete combustion: CH4 + O2 H2O + CO2 + CO + C (not balanced) * Complete combustion: What’s this ? 2C6H14 + 19O2 14H20 + 12CO2 This is not synthesis; more than 1 product is formed Always 2 products for complete combustion: Water and Carbon Dioxide 1 2 13 Problems What are the following reaction types? 2Na + S Na2S H2CO3 H20 + CO2 Na2O + H2O 2NaOH 2C2H6 + 7O2 4CO2 + 6H2O + Heat 4Na + O2 2Na2O + Heat 3C02 C + 2CO + 2O2 14