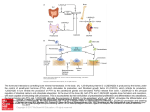
* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Question: 6
Hormone replacement therapy (male-to-female) wikipedia , lookup
Hypothyroidism wikipedia , lookup
Vasopressin wikipedia , lookup
Osteoporosis wikipedia , lookup
Hormonal breast enhancement wikipedia , lookup
Hyperandrogenism wikipedia , lookup
Graves' disease wikipedia , lookup
Androgen insensitivity syndrome wikipedia , lookup
Signs and symptoms of Graves' disease wikipedia , lookup
Growth hormone therapy wikipedia , lookup
January Question: 1 Abbreviations ACTH=corticotrophin, CAH=congenital adrenal hyperplasia, DHEA= dehydroepiandrosterone, DHEAS=dehydroepiandrosterone sulfate, FSH=follicle-stimulating hormone, GH=growth hormone, GnRH=gonadotropinreleasing hormone, hCG=human chorionic gonadotropin, IGF-1=insulinlike growth factor-1, LH=luteinizing hormone, MEN-1=multiple endocrine neoplasia-1, MRI=magnetic resonance imaging A mother brings in her 6-year-old daughter because she is worried that the girl is showing signs of early puberty. She noticed her daughter developing underarm odor and pubic hair 6 months ago. Her past year’s growth curve shows mildly accelerated linear growth. The mother had menarche at 11 years of age and believes that the child’s father and paternal aunt had early puberty. On physical examination, the well-appearing white child is at the 50th percentile for weight and 75th percentile for height, has no breast budding or clitoromegaly, but does have Tanner 3 pubic hair and underarm odor. She has no abdominal mass or birthmarks. Initial laboratory results include a morning DHEA-S of 180 mg/dL (4.9 mmol/L), total testosterone of 38 ng/dL (1.3 nmol/L), and 17-hydroxyprogesterone (17-OHP) of 210 ng/dL. Her bone age is advanced by 2.5 years, which is greater than 2 standard deviations. Of the following, the MOST appropriate next step in evaluation of this child is A. ACTH stimulation test B. adrenal ultrasonography C. clinical reassessment in 4 to 6 months D. cranial MRI E. GnRH agonist stimulation test Correct Adult body odor, pubic hair (pubarche), and acne are all signs of increased adrenal androgens (adrenarche). Premature adrenarche manifests as the presence of pubic hair in girls younger than 8 years of age (and boys younger than 9 years of age) without other evidence of puberty. Pubarche may occur approximately 6 months to 1 year earlier in non-Hispanic African American and Mexican American girls (as early as 7 years of age). An excess of vellous hair can be confused with Tanner stage 2 pubic hair, which is sparse, fine, and straight. Most affected children have isolated typical premature adrenarche and require only observation of clinical findings and growth velocity. Premature adrenarche is nearly 10 times more common in girls than boys. It is also more common in children who have a history of being small for gestational age, having a low birthweight, and having obesity with insulin resistance. Children who have a family history of early adrenarche, rapid tempo early pubic hair changes, or a bone age more than 1 year advanced, such as the girl described in the vignette, are at higher risk for an underlying disorder such as late-onset (nonclassical) CAH, adrenal tumors (rare), or true precocious puberty. Baseline testing for premature adrenarche often includes assessment of DHEA-S, testosterone, 17-OHP, and bone age. Laboratory values of pubertal range DHEA-S consistent with Tanner 2 to 3 pubic hair but minimal bone age advancement consistent with height age are reassuring. Assessment of serum adrenal androgens at baseline and 60 minutes after ACTH stimulation can determine if this child has late-onset adrenal hyperplasia. Steroid 21-hydroxylase deficiency accounts for 95% of CAH. Ultrasonography may be indicated if androgens values suggest the need for further evaluation of the adrenal gland, ovary, or testes. This girl has no evidence of breast tissue, but her advanced bone age puts her at risk for early central puberty. GnRH stimulation testing may be used to differentiate central from peripheral precocious puberty in a child presenting with breast or testicular development. Once central precocious puberty has been identified, cranial MRI may be indicated. Central precocious puberty is 5 times more common in girls and 9 times more likely to be idiopathic than for boys. Adrenarche is attributed to a pubertal maturation and expansion of the adrenal cortex zona reticularis, which is poorly understood. Adrenarche is likely regulated by increases in GH, IGF-1, insulin, and leptin coinciding with increasing body mass. The zona reticularis DHEA biosynthesis is upregulated while 3ß-hydroxysteroid dehydrogenase type 2 expression falls to adult activity during adrenarche. Virilization can result from androgen exposure (such as oral DHEA supplements or transdermal androgen gel exposure) as well as from endogenous androgen overproduction. Androgen-secreting tumors of adrenal glands are rare causes of hyperandrogenism. Pediatric adrenocortical carcinomas present with markedly elevated DHEA-S and testosterone values in a bimodal age distribution. Most occur before 3 years of age, with the remainder typically diagnosed after age 14 years. Adrenal carcinomas are associated with Li-Fraumeni syndrome, BeckwithWiedemann syndrome, Carney complex, MEN-1, and hemihypertrophy syndrome. Cushing syndrome may be suspected when virilization coincides with centripetal weight gain, hypertension, muscle weakness, and wide purplish striae. Virilizing gonadal tumors are very rare causes of precocious puberty. Similarly, ß-hCG from germ cell tumors in boys can stimulate Leydig cell production of testosterone. Typical female puberty begins with thelarche, defined by the presence of subareolar breast budding. Thelarche is independent of adrenarche and often precedes the development of pubic hair. Thelarche occurs at a mean age of 10.2 years (range, 8.2 to 12.1 years). According to the most recent guidelines, thelarche occurring before 8 years of age is considered premature, but it may be a variant of normal, particularly in certain racial/ethnic groups. Obesity may underlie the trends of earlier pubertal onset in girls and may accelerate pubertal onset by approximately 0.5 years. Premature thelarche can occur following exposure to topical formulations of estrogen or to lavender or tea tree oil. McCune-Albright syndrome is a rare condition causing gonadotropin-independent precocious puberty, with cyclic signs of premature thelarche and advanced bone age in girls who often have café au lait birth marks. Question: 2 Abbreviations: GnRH=growth hormone-releasing hormone, MRI=magnetic resonance imaging A 13-year-old boy presents with concerns related to growth. His height has crossed from the 75th to 50th percentiles over the past 2 years, with weight crossing from the 50th to 25th percentiles. On physical examination, he has 4-mL testes and Tanner stage 2 pubic hair. His mother had menses at 16 years of age but was a competitive gymnast, and the rest of her family had “normal” puberty. His father recalls growing a few inches in college. Of the following, the course of action that is MOST likely to confirm the diagnosis of delayed puberty for this boy is A. assessment of cranial nerve I B. bone age evaluation C. leuprolide stimulation test D. MRI of the hypothalamus and pituitary E. reassessment in 6 months Correct Answer: E The boy described in the vignette presents a common conundrum. His growth pattern, with crossing percentiles around the time of puberty, and the family history are most consistent with constitutional delay in puberty. Unfortunately, other than “tincture of time,” there is no diagnostic maneuver that can confirm this diagnosis. Although a delayed bone age and prepubertal bone age are consistent with the diagnosis, they may also be consistent with hypothalamic hypogonadism. Without overt signs of true dysfunction, MRI findings are likely to be normal. Anosmia may be consistent with the hypothalamic dysfunction often seen in Kallmann syndrome, but normal olfaction may also be present. Although a GnRH agonist test may confirm the lack of pubertal activation of the hypothalamic-pituitary-gonadal axis, it is not sensitive enough to discriminate between constitutional delay and hypothalamic hypogonadism. In 95% of the population, initial signs of puberty may occur as late as 13 years in girls and 14 years in boys. When signs do not occur by this age, delayed puberty or sexual infantilism may be diagnosed. Ultimately, the exclusion of underlying causes as well as the passage of time is the only way to reliably differentiate true pubertal delay from hypothalamic hypogonadism (gonadotropin deficiency). However, certain clinical clues can point to a diagnosis of delayed puberty versus gonadotropin deficiency. Family history is an important factor. Up to 80% of the control of timing of puberty is estimated to be due to genetic factors. Hence, in some series, up to 75% of patients who have delayed puberty have been noted to have a family history of delayed puberty, with inheritance patterns resembling autosomal dominance with variable penetrance. Nonetheless, kindreds who have hypothalamic hypogonadism may also have varied presentations, with some members having merely delayed puberty. Similarly, because some men who have idiopathic hypothalamic hypogonadism (IHH) experience spontaneous reversal after testosterone therapy, a history of subsequent puberty cannot completely rule out IHH. As with any other growth evaluation, underlying nonendocrine diseases should be considered when assessing the patient in whom delayed puberty is suspected. Chronic disease and malnutrition may lead to a functional hypothalamic gonadotropin deficiency. Among the diagnostic possibilities are diseases related to undernutrition, including inflammatory bowel and celiac diseases, poorly controlled diabetes, cystic fibrosis, and eating disorders. In addition, other systemic chronic diseases such as significant congenital heart disease or active or previous malignancies, and cancer therapy should be considered. Energy intake may be inadequate in the setting of excessive exercise to allow for puberty to progress. One commonality of these diseases is decreased body fat. Leptin both reflects adiposity and is permissive for puberty. Hence, whether via malnutrition or increased energy utilization, decreased body fat may result in pubertal delay. Finally, delayed puberty can be the first recognizable manifestation of previously unrecognized genetic syndromes or underlying pituitary dysfunction Question: 3 Abbreviations: AVP=arginine vasopressin, BUN=blood urea nitrogen, c-AMP=cyclic adenosine monophosphate You receive a consultation request for a 6-month-old adopted infant who was admitted for dehydration. He had an uncomplicated term birth, and his birthweight and height were at the 60th percentile. He was breastfed initially but was switched to formula after his weight and length fell to 5th percentile by 4 months of age. His mother had reported to the emergency medicine physician that her son has had frequent vomiting with fussing, 10 to 12 soaked diapers daily, and hard and pebblelike stools for the past 6 weeks. He drinks at least 14 bottles per day, with a preference for water. Physical examination revealed dry skin, sunken eyes, no skin tenting, and palpable abdominal stool. His laboratory results from specimens drawn upon arrival to the emergency department showed: Sodium, 154 mEq/L (155 mmol/L) Potassium, 5.3 mEq/L (5.3 mmol/L) Chloride, 119 mEq/L (119 mmol/L) Bicarbonate, 21 mEq/L (21 mmol/L) BUN, 83 mg/dL (29.6 mmol/L) Creatinine, 0.8 mg/dL (70.7 mmol/L) Calcium, 10.4 mg/dL (2.6 mmol/L) Urine osmolality, 70 mOsmol/kg Plasma vasopressin, 27 pg/mL (24.9 pmol/L) Of the following, the MOST typical inheritance pattern of this infant’s condition is A. autosomal dominant B. autosomal recessive C. imprinting D. sporadic E. X-linked Correct Answer: E The infant described in the vignette is unable to concentrate urine adequately in the midst of progressive dehydration and hypernatremia, and he has symptoms of polyuria, polydipsia, and extreme thirst. Vasopressin unresponsiveness should be suspected when plasma vasopressin values are elevated for the concurrent values of plasma osmolality. Vasopressin unresponsiveness can result from mutations altering the vasopressin AVPR2 receptor or the aquaporin-2 water channel. Although at least 7 aquaporins are expressed in the human kidney, only AQP2 abnormalities are associated with diabetes insipidus (DI). Nephrogenic (vasopressin-resistant) DI is most often inherited as an X-linked recessive defect of the vasopressin AVPR2 receptor caused by errors in folding, truncation, or location (MIM #222000). This receptor defect is responsible for approximately 90% of patients who have congenital nephrogenic DI. A typical family history for those whose nephrogenic DI is caused by a defective AVPR receptor includes similar symptoms in a male sibling or a maternal uncle. Female carriers experience milder symptoms. A spectrum of partial to complete DI can be present from birth in male offspring. Because the resistance to AVP is often partial, this form of nephrogenic DI can be difficult to distinguish from mild vasopressin deficiency or primary polydipsia. The G protein-coupled vasopressin receptor is responsible for vasopressin-related antidiuretic effects in the kidney. Conformational changes induced by the binding of vasopressin to its receptor in the collecting ducts trigger a cascade of increased c-AMP, activation of the protein kinase A (PKA) pathway, and phosphorylated AQP2 tetramer channel formation. This shuttle and repositioning of the AQP2 channels on the apical plasma membrane allows for reabsorption of water from the urine into the cells of the collecting duct. Any defect in this process impairs the ability to concentrate the high-volume dilute urine that originally entered the collecting ducts from the ascending limb of the loop of Henle. Fewer than 10% of nephrogenic DI cases are caused by mutations of the AQP2 water channel gene on chromosome 12q13 (MIM #125800 and #25700), conditions that usually have an autosomal recessive pattern of inheritance. Affected patients can develop severe hypertonic dehydration following mild impairment of fluid intake. A milder and extremely rare form of autosomal dominant vasopressin resistance is caused by abnormal AQP2 trafficking to the apical membrane. This defect impairs regeneration of functioning AQP2 channels. Vasopressin unresponsiveness can be acquired due to exposure to lithium and less commonly, other drugs, including amphotericin B and tetracycline. Lithium accumulation in apical membrane cells dysregulates AQP2 formation. AQP2 disorders also occur in association with hypokalemia, hypercalcemia, ureteral obstruction, sickle cell disease, and metabolic acidosis. If the vasopressin unresponsiveness is partial, high doses of desmopressin may produce a modest improvement in urine osmolality, polyuria, and thirst. Management of vasopressin unresponsiveness requires sodium restriction to reduce solute load while compensating with extra free water. Thiazides both upregulate AQP2 and reduce sodium reabsorption to reduce urine volume. Thiazides may be combined with amiloride diuretic or indomethacin for synergistic improvement of AQP2 shuttling, which helps to decrease urine output. Therapeutic strategies to identify vasopressin 2 receptor-independent mechanisms and alternative regulation of AQP2 trafficking are emerging. Question: 4 Abbreviations: GH=growth hormone, T4=triiodothyronine, TSH=thyrotropin An 8-year-old girl transfers to your practice. Her records indicate that she presented at 3 years of age with severe growth failure and weight gain and was found to have GH deficiency and hypothyroidism. DNA analysis revealed a Pit-1 mutation. She was treated with both GH and L-thyroxine until about 6 weeks ago, when she moved to your community. Her mother misplaced her medicine bag and consequently the girl has had neither GH nor L-thyroxine for the past 6 weeks. Laboratory tests confirm frank hypothyroidism, and you order 75 mg of L-thyroxine. Of the following, the MOST appropriate test(s) to monitor her thyroid function is(are) A. free T4 in 1 week B. free T4 in 2 weeks C. free T4 in 6 weeks D. free T4 and TSH in 2 weeks E. T4 and TSH in 2 weeks Correct Answer: C The girl described in the vignette has a Pit-1 mutation, which results in central hypothyroidism and GH deficiency. Because she has central hypothyroidism, free T4 (and not TSH) is the best indicator of her thyroid status. Adequacy of the L-thyroxine dose should only be assessed once a steady state has been reached (ie, after at least 5 half-lives). Accordingly, because the half-life of L-thyroxine is 5 to 7 days, free T4 should be measured in 5 to 6 weeks. Measuring free T4 in 1 or 2 weeks is too soon. When a fixed dose of a drug is administered at regular intervals, the drug accumulates during the absorption phase until it reaches steady state, at which point the rate of drug intake equals the rate of drug elimination. The time required to reach steady state depends on the elimination half-life of the drug, defined as the time required for the serum drug concentration to decrease by 50%. In the absence of a loading dose, at least 5 half-lives are required to achieve a steady state. For primary hypothyroidism (ie, normal central regulation), the dose of L-thyroxine can be adjusted based on a goal of maintaining the TSH concentration in the normal reference range (ideally, in the lower half of the reference range). The TSH cannot be used for central hypothyroidism, and dose adjustments are based on the free T4 values. The goal is to maintain the free T4 concentration in the mid to upper normal range. Using the free T4 rather than total T4 minimizes the effect of any binding abnormalities. However, free T4 assays in general use are affected by interfering substances and significant binding abnormalities. These effects can often be minimized by using free T 4 by equilibrium dialysis. In addition, the free T4 has a narrower normal range than total T4. Children are generally monitored every 4 to 6 months while growing and going through puberty. Younger age is associated with more rapid metabolism of thyroxine, necessitating a higher daily replacement dose. The recommended starting doses of thyroxine are as follows: Age (yrs) Dose (µg/kg per day) Newborn 10 to 15 1 to 3 4 to 6 3 to 10 3 to 5 10 to 16 2 to 4 Adults 1.7 An alternative replacement dose is approximately 100 mg/m2 per day. Several substances may interfere with optimal L-thyroxine absorption, particularly soy, calcium, and iron supplements. In addition, certain medications, especially anticonvulsants, alter thyroid hormone metabolism and clearance, making interpretation of laboratory tests difficult (Table). Question: 5 Abbreviations: LH=luteinizing hormone A 17-year-old boy is diagnosed with Hodgkin lymphoma. Because he has an underlying cardiac condition, his oncologist decides to use MOPP [Mustargen (mechlorethamine, nitrogen mustard), Oncovin (vincristine), Procarbazine, and Prednisone] combination chemotherapy rather than ABVD (Adriamycin, Bleomycin, Vinblastine, and Dacarbazine) combination chemotherapy. He is planning to administer 6 cycles of chemotherapy. Of the following, the MOST correct statement about the effect of the chemotherapy on the boy’s gonadal function is that A. both protocols place him at significant risk for gonadal dysfunction B. he has an equal likelihood of developing hypoandrogenism as infertility C. he is more likely to have testicular injury than a prepubertal boy D. his risk for infertility is similar to that seen with cyclophosphamide plus total body irradiation E. there would be no gonadal concerns if he had been female Correct Answer: D Alkylating agents (Table - see below) damage DNA and induce apoptosis through covalent binding of alkyl groups to cellular macromolecules. The risk of gonadal injury is related to the specific alkylating agent, cumulative dose, and length of treatment. Leydig cell function is generally preserved, but germ cell failure is common. The highest risk factors are associated with: · 3 or more cycles of MOPP chemotherapy · Busulfan dose of 600 mg/m2 or more · Cumulative cyclophosphamide dose of 7.5 g/m2 or more or as part of conditioning for hematopoietic cell transplant (HCT) · Ifosfamide dose of 60 mg/m2 or more · Any alkylating agent chemotherapy combined with testicular radiation, pelvic radiation, or total body irradiation (TBI) Table. Alkylating Agents Used in Chemotherapy Classic Alkylators Busulfan Carmustine (BCNU) Chlorambucil Cyclophosphamide Ifosfamide Lomustine (CCNU) Mechlorethamine Melphalan Procarbazine Thiotepa Heavy Metal Alkylators Carboplatin Cisplatin Nonclassic Alkylators Dacarbazine (DTIC) Temozolomide Accordingly, the 6 cycles of MOPP therapy described for the boy in the vignette place him at a similar risk for infertility as would cyclophosphamide and TBI as conditioning for HCT. The ABVD regimen is associated with a lesser risk for gonadal dysfunction than the MOPP regimen. Heavy metal alkylating agents only have a medium risk relative to classic alkylators. Prepubertal status is not protective in males. In males with Hodgkin disease, one study found 70% of patients had semen abnormalities before treatment. Radiation therapy also can affect gonadal function. Radiation doses as low as 0.1 to 1.2 Gy can injure the progenitor type and differentiating spermatogonia, resulting in impaired spermatogenesis. Doses of 2 to 3 Gy also affect stem cell spermatogonia and lead to long-term azospermia, although this may be reversible. Doses greater than 6 Gy deplete the spermatogonial stem cell pool, resulting in likely permanent infertility. Fractionated doses appear to be more toxic than an equivalent single-dose exposure; some have hypothesized that reserve stem cells become activated and mitotically active after gonadotoxic damage. The Leydig cells of the testis are more resistant to radiation damage than the germ cells. Leydig cell failure does not occur until doses of 20 Gy in prepubertal boys and 30 Gy in postpubertal males. Although testosterone production may be normal at lower doses of fractionated testicular radiation, elevated LH concentrations suggest subclinical injury. Because ovarian hormone production is linked to maturation of primordial follicles, depletion of follicles by alkylating agent chemotherapy can potentially affect both hormone production and fertility. Any insult that reduces the number of follicles leads to an increased risk of premature ovarian failure (menopause before 41 years of age). The younger the child, the greater the complement of primordial follicles and, therefore, the greater the ovarian reserve. After alkylating agent chemotherapy, ovarian function can be retained or recover, with the risk of acute ovarian failure greatest after combined modality treatment with alkylating agent chemotherapy and abdominal or pelvic radiation or after dose-intensive alkylating agents for myeloablative conditioning before HCT. The LD50 for the ovary is less than 2 Gy. The highest risk factors for girls are high-dose radiation to the ovaries (>10 Gy), with permanent ovarian failure associated with doses of 20 Gy or more (70% of girls in 1 study); treatment with procarbazine; and treatment with cyclophosphamide in adolescence (13 to 20 years). Ovarian function may be retained in up to 50% of prepubertal girls who undergo conditioning with TBI; if the girl is older than 10 years at the time of TBI, acute ovarian failure generally occurs. Childhood cancer survivors who retain ovarian function are at increased risk of premature menopause. Question: 6 You are seeing a 16-year-old boy in clinic for recurrent fractures. He presented with a right femur fracture at the age of 7, which progressively deformed and did not heal well despite surgery. Since then, he has had numerous fractures, including femoral fractures requiring rodding (Figure 1). He also has had bilateral vision loss. The child’s parents are first cousins, and he has an older brother who has had similar limb deformity and fracture. On physical examination, the boy’s weight is 55 kg, height is 142 cm, and head circumference is 70 cm. He has notable cranial asymmetry with left maxillary prominence and malalignment of his teeth, horizontal nystagmus with limited visual acuity, and a broad asymmetric chest with sternal prominence. Cardiac and abdominal examinations yield no findings of note. The boy is fully pubertal. He has dextroscoliosis. Results of laboratory studies include: Hemoglobin, 10 mg/dL (100 g/L) Calcium, 8.6 mg/dL (2.15 mmol/L) Phosphorus, 4.2 mg/dL (1.36 mmol/L) Alkaline phosphatase, 1,799 U/L (30.0 mkat/L) Of the following, the MOST likely explanation for this boy’s high alkaline phosphatase value is A. excess osteoprotegerin B. increased bone formation C. osteosarcoma D. transient hyperphosphatasemia E. vitamin D deficiency Correct Answer: B The boy described in the vignette has juvenile Paget disease, an autosomal recessively inherited disease caused by mutations (homozygous or compound heterozygous) in the TNFRSF11B gene. TNFRSF11B codes for osteoprotegerin (OPG), a soluble member of the tumor necrosis factor (TNF) receptor family. OPG serves as a decoy receptor for RANK (receptor activator of nuclear factor kappa B)-ligand. When OPG concentrations are low, more RANK-ligand stimulates RANK on osteoclasts, leading to increased bone resorption (Figure 2). Because bone resorption and formation are coupled activities, when osteoclastic resorption increases, bone formation by osteoblasts also increases, leading to high circulating concentrations of alkaline phosphatase. Paget disease in adulthood can present as a focal disorder of bone remodeling (often affecting just the spine, pelvis, femurs, or skull), but juvenile Paget disease (MIM #23900) presents in infancy or early childhood and affects the skeleton more globally. Affected children have rapidly remodeling woven bone, with associated osteoporosis, fractures, and progressive bony deformity. They may also have macrocephaly, hearing loss, and early tooth loss. Although transient hyperphosphatasemia also is associated with increased circulating alkaline phosphatase, transient hyperphosphatasemia is generally only reported in children younger than 5 years of age and is not accompanied by skeletal abnormalities, as seen in this boy. Vitamin D deficiency can also increase alkaline phosphatase concentrations but values of greater than 1,000 units/L (16.7 mkat/L) would be unusual in a 16-yearold patient. Although adults who have Paget disease are at risk of osteosarcomas and alkaline phosphatase concentrations can be elevated in persons with osteosarcoma, a Paget-associated osteosarcoma would be extremely rare in a pediatric patient. Bone is an intricate living tissue, composed of outer compact cortical bone and inner meshlike cancellous bone. Both cortical and cancellous bone contain organic mineralized matrix made of a collagen scaffolding impregnated with calcium hydroxyapatite and calcium phosphate. The protein comprising this matrix is more than 90% type 1 collagen, a triple helix of 2 a-1 polypeptides encoded by COL1A1 and 1 a-2 polypeptide encoded by COL1A2. Within and upon this bone matrix are numerous cells. Osteoblasts derive their names from the Greek words for “bone” and “embryonic layer.” These tall, plump, boneforming cells have large, elaborate Golgi apparati and arise from mesenchymal precursor cells in the bone marrow. Their differentiation is controlled by growth factors, including a number of bone morphogenic proteins. Osteoblasts line up on surfaces where bone is formed and initiate bone formation by laying down osteoid. Osteoid is unmineralized bone that primarily consists of type 1 collagen but also contains other types of collagen, osteocalcin, osteonectin, and glycoproteins. Osteocalcin can be measured in serum as a gauge of bone formation. Once the scaffolding is laid down, osteoblasts mineralize it by depositing hydroxyapatite crystals. This process requires calcium, phosphate, and the enzyme alkaline phosphatase, which is secreted in large amounts by osteoblasts and can be measured in serum to assess bone formation. Osteoclasts derive their names from the Greek words for “bone” and “broken.” They are multinucleated giant cells that arise from monocyte-derived mononuclear hematopoietic precursors and resorb bone. Osteoclasts require RANK-ligand to differentiate and become active. Maturation is accelerated by PTH and vitamin D. As osteoclasts mature, they fuse into multinucleated cells and produce osteoclast-specific enzymes. These cells are relatively rare in bone. To resorb bone, osteoclasts settle on a bone surface, seal off an area, and develop an invaginated plasma membrane adjacent to the bone surface known as the “ruffled border.” This ruffled border works as a huge lysosome, secreting acid onto the bone surface and breaking down the matrix by secreting collagenase and proteases. The resulting collagen fragments have pyridinoline cross-links that can be assayed in serum or urine as a measure of bone resorption. The most abundant bone cells are osteocytes, whose name comes from the Greek words for “bone” and “cell.” Osteocytes are small, flat, star-shaped cells that are terminally differentiated osteoblasts. They have a half-life of 25 years. They are connected to each other and to osteoblasts by an extensive canalicular network. Canaliculi are used for exchange of nutrients and waste through gap junctions. Osteocytes have an important role as mechanoreceptors. They detect bone strains and help coordinate bone modeling and remodeling. Question: 7 Abbreviations ACTH=corticotrophin, GH=growth hormone, IM=intramuscular, IV=intravenous, MRI=magnetic resonance imaging, TSH=thyrotropin It is the Friday before New Year’s and you are seeing a 12-year-old girl for follow-up evaluation in clinic. She had Hashimoto thyroiditis diagnosed at the age of 5 and Addison disease diagnosed at the age of 7. Over the past year she had been clinically stable, taking levothyroxine 88 mg/day; hydrocortisone doses of 5 mg in the morning, 2.5 mg in the afternoon, and 2.5 mg in the evening; and fludrocortisone 0.1 mg/day. During the previous summer, her TSH was 8.1 mIU/mL, and you increased her levothyroxine dose to 100 mg/day. She has not yet had repeat laboratory assessments on this new dose. In the fall, she had 3 generalized tonic-clonic seizures that were not believed to be related to her autoimmune disease. Her neurologic examination and MRI yielded normal results, but electroencephalography showed paroxysmal activity in her right occiput. Her primary care physician prescribed phenytoin at 200 mg/day. Two weeks ago she was hospitalized overnight with what was considered an acute viral illness involving vomiting, fatigue, and hyponatremia. On physical examination, the girl’s height is 151 cm, weight is 42 kg, blood pressure is 90/60 mm Hg, and heart rate is 100 beats/min. She has generalized hyperpigmentation. Laboratory results include: Serum sodium, 129 mEq/L (129 mmol/L) Potassium, 3.3 mEq/L (3.3 mmol/L) Glucose, 54 mg/dL (3.0 mmol/L) Phenytoin, 13 mg/L (normal, 10 to 20 mg/L) Of the following, the MOST likely explanation for this girl’s current findings is that her A. fludrocortisone dose is too low due to drug-induced inhibition of 11ß-hydroxylase B. levothyroxine dose is too low because of changes in absorption C. hydrocortisone dose is too low due to body growth D. hydrocortisone dose is too low due to increased CYP3A4 metabolism E. hydrocortisone dose is too low due to increased metabolism at higher L-thyroxine dose Correct Answer: D The girl described in the vignette has clinical and laboratory signs of cortisol insufficiency (hyperpigmentation, hyponatremia, hypokalemia, and hypoglycemia). This is most likely due to more rapid hydrocortisone metabolism because of P-450 cytochrome CPY3A4 induction by phenytoin. She may also have had more rapid fludrocortisone metabolism through the same pathway. Although certain cortisol synthesis inhibitors (aminoglutethimide and metyrapone) block 11ß-hydroxylase, there is no evidence that phenytoin acts to inhibit this pathway. The Table lists causes of drug-induced cortisol deficiency that can be seen in pediatric patients. Moderate-to-severe hypothyroidism can be associated with hyponatremia, but this girl’s hypothyroidism is relatively well controlled, and hypothyroidism would not explain her hyperpigmentation or other laboratory abnormalities. Conversely, although treatment of profound hypothyroidism can unmask previously undiagnosed adrenal insufficiency, minor adjustments in thyroid hormone doses in a child already receiving steroid hormone replacement is not likely to precipitate adrenal crisis. Although she is a growing adolescent, her body surface area is 1.3 m2 and, therefore, her current hydrocortisone dose of approximately 7.5 mg/m2 per day is adequate when calculated for her body size. Causes of primary adrenal insufficiency in children include abnormalities of adrenal gland development, disorders of steroidogenesis, unresponsiveness to ACTH (familial glucocorticoid deficiency), peroxisomal defects, and damage to previously normal glands. In boys, a diagnosis of adrenoleukodystrophy should also be entertained. In addition, acquired central adrenal insufficiency is common after a period of supraphysiologic glucocorticoid therapy. Adrenal insufficiency in this context can be seen after as few as 2 to 3 weeks of corticosteroid therapy, and the risk of insufficiency can be minimized or avoided by tapering corticosteroid doses before discontinuation. The signs and symptoms of glucocorticoid deficiency are wide-ranging and reflect the importance of glucocorticoids to multiple body systems. Some of the more important relationships include the effects of glucocorticoids on intermediary metabolism at the levels of the liver, fat, and skeletal muscle. Therefore, cortisol deficiency can lead to hypoglycemia, decreased adiposity, and muscle weakness. Cortisol also affects the immune system. Persons who have adrenal insufficiency can have relative lymphocytosis, monocytosis, granulocytopenia, and eosinophilia. Decreased cortisol leads to increased renal calcium reabsorption and may result in hypercalcemia. Skin darkening only occurs in those persons who have high ACTH concentrations and is due to ACTH-mediated stimulation of epidermal melanocortin 1-receptors. Question: 8 Abbreviations CT=computed tomography, PTH=parathyroid hormone, T4= thyroxine, TSH=thyrotropin, 25-OH vitamin D=25hydroxyvitamin D, 1,25 (OH)2 vitamin D=1,25 dihydroxyvitamin D Paramedics bring a 16-year-old boy to the emergency department at 08:35 am because of a generalized seizure that lasted 10 minutes following a short bout of gastroenteritis. He has a history of seizures, the first of which occurred at age 10 years and was found to be due to hypocalcemia resulting from hypoparathyroidism. He was prescribed calcium carbonate and calcitriol supplements, but his compliance has been poor, and previous assessments have yielded calcium values in the range of 5 to 6 mg/dL (1.25 to 1.50 mmol/L). The boy was diagnosed with Graves disease at 9 years of age when he presented with hyperthyroidism, a goiter, and mild bilateral proptosis. Six weeks after starting methimazole, he developed neutropenia, and underwent radioactive ablation of the thyroid gland. He has been receiving thyroid hormone replacement with levothyroxine since 2 months after thyroid ablation. Two months ago, his TSH value was 8.23 mIU/L and total T 4 was 12.3 mg/mL (210.3 nmol/L). On physical examination, the sleepy patient is easily aroused, does not have focal neurologic deficits, and is alert and oriented to person, place, and time. He has no signs of meningism. He has a Chvostek sign, is jittery, and has mild proptosis. His extraocular movements are intact and optic disks are sharp. Laboratory testing yields: Sodium, 141 mEq/L (141 mmol/L) Potassium, 4.2 mEq/L (4.2 mmol/L) Chloride, 117 mEq/L (117 mmol/L) Blood urea nitrogen, 12 mg/dL (4.3 mmol/L) Creatinine, 0.7 mg/dL (61.9 mmol/L) Glucose, 124 mg/dL (6.9 mmol/L) Calcium, 5.4 mg/dL (1.4 mmol?/L) Phosphorus, 7.5 mg/dL (2.4 mmol/L) Magnesium, 1.9 mEq/L (0.95 mmol/L) Albumin, 4.2 g/dL (42 g/L) Intact PTH, 22 pg/mL (22 ng/L) TSH, 10.25 mIU/L T4, 11.4 mg/dL (194.9 nmol/L) Of the following, a CT scan without contrast is MOST likely to reveal A. macrocephaly B. subarachnoid hemorrhage C. subdural hemorrhage D. thalamic calcifications E. ventriculomegaly Correct Answer: D Hypocalcemia has numerous causes. However, the combination of hypocalcemia, hyperphosphatemia, and an inappropriate-for-hypocalcemic normal range intact PTH unequivocally indicates primary hypoparathyroidism. Decreased activation of parathyroid chief cell membrane localized calcium-sensing receptors by extracellular calcium induces PTH secretion (Figure). PTH facilitates renal 1a-hydroxylation of 25-OH vitamin D (calcidiol) to 1,25 (OH)2 vitamin D (calcitriol). Calcitriol increases intestinal absorption of calcium and phosphorus. PTH also increases bone resorption, which results in release of calcium and phosphorus from bones into the circulation. Finally, PTH acts in the kidneys to increase calcium and decrease phosphorus renal tubular reabsorption. The net effect of these actions is to increase serum calcium and decrease serum phosphorus concentrations. Lack of PTH decreases circulating calcitriol and, thus, calcitriol-mediated gastrointestinal calcium and phosphorus absorption. PTH deficiency also leads to decreased renal tubular reabsorption of calcium, causing hypocalcemia accompanied by hyperphosphatemia. Yet, counterintuitively, hypoparathyroidism causes calcification in various organ systems, including the brain. The classic pattern found in the brain includes basal ganglia calcifications. Thalamic calcifications may occur in association with basal ganglia calcifications. Neither acute nor chronic hypocalcemia is known to cause macrocephaly or subarachnoid or subdural hemorrhages. Hypoparathyroidism may cause pseudotumor cerebri, but pseudotumor cerebri is defined as increased intracranial pressure without ventriculomegaly. Increased intracranial hypertension with ventriculomegaly is hydrocephalus, which is not known to be caused by hypoparathyroidism. There are many causes of primary hypoparathyroidism (Table 1). The primary hypoparathyroidism for the boy in the vignette is most likely due to an autoimmune process because the presence of another autoimmune condition, Graves disease of the thyroid, increases his risk for a second autoimmune condition. The autoimmune process may involve T-cell- and/or humoral-mediated destruction of the parathyroid gland and/or the presence of antibodies that activate calcium-sensing receptors on parathyroid chief cells. Collateral damage of the parathyroid glands by radioactive iodine is not a likely cause of this boy’s hypoparathyroidism. Evidence that radioactive iodine ablation of the thyroid gland causes hypoparathyroidism is from rare isolated case reports and has not been verified by large observational studies. Also, it is not unexpected for an occasional patient who has a history of Graves disease to develop autoimmune parathyroid disease. However, thyroidectomy for the treatment of Graves disease has been associated with iatrogenic hypoparathyroidism due to removal or damage of the parathyroid glands during surgery or interruption of their blood supplies. There are numerous potential adverse effects of acute and chronic hypocalcemia due to hypoparathyroidism (Table 2 and Table 3). Many patients who have chronic hypocalcemia do not manifest the adverse effects of acute hypocalcemia unless their serum calcium concentrations rapidly drop. Such a rapid decrease can result from restricted enteral intake due to surgery or acute illness; increased rate of passage of calcium through the gut due to diarrhea; increased calcium demand due to pregnancy, breastfeeding, or rapid skeletal growth; an alkalotic state; and use of drugs that cause hypocalcemia or impair intestinal calcium absorption. In addition, states of increased phosphate load such as trauma, surgery, and the use of phosphate enema can depress serum calcium acutely. The duration of hypoparathyroidism and low serum calcium-phosphorus ratios are associated with the risk for developing basal ganglia calcifications. Therapeutic regimens leading to increased serum calcium-phosphorus ratios may slow progression of basal ganglia calcifications as its possible neurologic consequences (Table 3). Such regimens may need to include approaches for lowering circulating phosphorus concentrations as well as increasing serum calcium concentrations to the lower end of the normal range. The efficacy and safety of such approaches, which include oral phosphate binders and recombinant injectable PTH formulations, is still being assessed in the pediatric age range. Nephrocalcinosis, nephrolithiasis, and renal failure are believed to be potential adverse consequences of treatment of hypocalcemia due to hypoparathyroidism, rather than a consequence of hypocalcemia. Individuals who have autosomal dominant hypoparathyroidism due to activating calcium-sensing receptor mutations appear to be at particularly high risk for iatrogenic calcification-related renal failure from calcium and vitamin D treatment of their condition. February Question: 1 Abbreviations GH=growth hormone, IGF-1=insulinlike growth factor 1 You are considering potential therapy for an otherwise healthy 9-year-old boy whose height is -3 standard deviations and who has abnormally low IGF-1 concentrations but a peak GH value of 13 ng/mL (13 mg/L) on stimulation testing with clonidine and arginine. You recall that the growth response of children who have GH deficiency treated with GH is better than the response of children who have primary IGF deficiency treated with IGF-1 and that this has been attributed to direct, non-IGF-mediated GH actions at the growth plate. Of the following, the growth plate layer that is MOST likely stimulated by GH rather than IGF-1 is the A. calcified zone B. hypertrophic zone C. proliferative zone D. resorption front E. resting or reserve zone Correct Answer: E Longitudinal bone growth occurs at the growth plate by endochondral bone formation in which cartilage is created and then remodeled into bone tissue (chondrogenesis and ossification). The sequence of events in bone formation occurs in the direction from the epiphysis toward the metaphysis, with new bone elongation appearing from the metaphysis outward (Figure). The resting or reserve zone contains prechondrocytes, whose differentiation into chondrocytes is GH-dependent and IGF-1- independent. This differentiation involves expression of new genes, including IGF1 and IGF1R. Chondrocytes then undergo IGF-dependent clonal expansion (cell division) in the proliferative zone. Subsequently, the cells change from flat to long in the hypertrophic zone, which accounts for more than half of the growth plate length. The hypertrophic chondrocytes secrete extracellular matrix, including collagen type X, as well as matrix vesicles that lead to calcification. The hypertrophic cells only live a few days, undergoing apoptosis or programmed cell death, which leaves behind lacunae within the calcified matrix. The lacunae are invaded by bone marrow, which provides blood vessels, osteoblasts, and osteoclasts (resorption front). Thus, cartilage is laid down first and replaced by bone. Both GH and IGF-1 stimulate osteogenesis. Question: 2 Abbreviations AMH= anti-Müllerian hormone, FSH=follicle-stimulating hormone, GH=growth hormone, hCG=human chorionic gonadotropin, IM=intramuscular, LH= luteinizing hormone A 12-year-old boy comes to the endocrinology clinic for follow-up evaluation of a small penis, which originally was evaluated when he was 7 years old. Physical examination of the boy on the initial evaluation documented normal stature and weight (both around the 25th percentile), a stretched penile length of 3.7 cm, otherwise normal findings on genitourinary examination, and bilaterally descended testes that were 1 mL in volume. There were no other findings of note on his history and the remainder of his physical examination. Laboratory evaluation at that time yielded: Total testosterone, 0.8 ng/dL (0.03 nmol/L) Stimulated testosterone (after 1,500 units hCG intramuscularly every other day x 7), 8.5 ng/dL (0.29 nmol/L) LH, 0.2 mIu/mL (0.9 IU/L) FSH, 0.6 mIu/mL (0.6 IU/L) Karyotype, 46,XY Bone age, 6 years Physical examination findings today show height at the 25th percentile, weight just below the 50th percentile, penile length of 3.9 cm, and testes volume remaining at 1 mL. He has Tanner 2 pubarche. Laboratory results include: Total testosterone, 6.2 ng/dL (0.22 nmol/L) LH, 8.9 miIu/mL (8.9 IU/L) FSH, 18.6 miIu/mL (18.6 IU/L) Inhibin B, 28 pg/mL (normal, 50 to 250 pg/mL) Bone age, 10 years Of the following, the MOST likely diagnosis for this boy is A. 5-alpha reductase deficiency B. mutation of steroidogenic factor 1 gene (SF1) C. mutation of the FGFR1 gene D. mutation of the KAL1 gene E. partial androgen insensitivity syndrome (PAIS) Correct Answer: B Patients who have an 46,XY genotype and an SF1 mutation can present with a variable phenotype that ranges from female to ambiguous genitalia and possibly associated adrenal insufficiency. Much milder defects of virilization have now been documented, such as described for the boy in the vignette, for whom an SF1 mutation is the only plausible explanation of those provided. Affected patients usually also show hormonal characteristics of gonadal dysgenesis, such as hypergonadotropism and low inhibin B concentrations. Basal and post-hCG testosterone as well as AMH values are also low. A male affected by 5-alpha reductase deficiency has a small phallus associated with hypospadias (often perineal) and a urogenital sinus. Testosterone concentrations should rise after hCG stimulation and are usually elevated beyond childhood. The defect lies in the SRD5A2 gene, expressed in androgen target tissues and responsible for the conversion of testosterone to dihydrotestosterone (DHT) required for normal virilization of the external genitalia. Those who have PAIS, either with ambiguous genitalia or microphallus plus penoscrotal hypospadias, have preserved inhibin B secretion from the Sertoli cells. Accordingly, FSH is normal or only mildly elevated in affected children older than 10 years of age. In complete AIS, the phenotype is female. Mutations of the KAL1 gene and of the FGFR1 gene are known causes of congenital hypogonadotropic hypogonadism, with resulting low LH and FSH concentrations. Micropenis is an anatomically correct phallus that is abnormally short. Some use a penile length at -2 SD as the lower limit of normal variance; others use -2.5 SD. Normative values for penile length with SD scores and the lower limit of normal are provided in Table. True micropenis is also characterized by reduced diameter and can be assessed by measuring the width at the mid-shaft. Micropenis can be one of the presenting features of hypopituitarism associated with gonadotropin deficiency (Table 2). Micropenis may be the only phenotypic abnormality in hypopituitarism and should prompt the clinician to look into the possibility of other pituitary hormone deficiencies. During the first trimester, hCG controls fetal androgen production needed for differentiation of the male external genitalia and penile development. After that time, fetal pituitary hormones control androgen production. Insufficient androgen production due to gonadotropin insufficiency results in suboptimal penile growth. In addition, significant GH deficiency can lead to a small penis. Some conditions are occasionally considered as consistent with micropenis when they truly represent variants of normal. A hidden penis, also named concealed penis, is a normally-sized penis that is not easily visible by inspection, primarily due to a large overlying suprapubic fat pad. Several other terms have been used instead of hidden penis. The term buried penis has been used to describe an otherwise normal phallus further concealed in the pubic (adipose) tissue due to inadequate fixation of the skin at the base of the penis. On the other hand, if the penoscrotal angle is poorly defined, often due to an extension of the scrotal skin distally on the ventral surface of the penile shaft, the condition should be termed a webbed penis. When a penis is trapped, scar tissue frequently develops due to prior surgery on the organ, usually after circumcision on a smaller or previously hidden penis. All of these forms of normal penile variation must be differentiated from a true micropenis by accurate measurement of the stretched penile length with a ruler (from the symphysis of the pubic bone to the distal tip of the glans). The goal of therapy for micropenis is to provide the patient with a penile size large enough to allow normal urination while standing, have a normal body image, and allow normal sexual function later in life. Intervention usually involves IM administration of synthetic androgens to bring the penis size within the normal range. Testosterone esters (enanthate or cypionate) at a dose of 25 mg IM monthly are often administered in 3 injections, which usually results in a significant increase in the penile size, followed by some reduction in the months after the last injection. In general, the overall penile size remains larger than before therapy, obviating the need for repeated courses of testosterone. If needed, additional treatment can be provided during the years of puberty. Such short-term use of testosterone is well-tolerated without any inadvertent advancement of the bone maturation. Some evidence suggests that prepubertal administration of testosterone may negatively affect the ultimate adult penile size that can be achieved with androgen therapy later during adolescence or adult life. Most available information seems to indicate that intervention during infancy is safe (likely because it often coincides with the time of mini-puberty). Nonetheless, more studies are needed. Question: 3 Abbreviations GH=growth hormone, HbA1c=glycolated hemoglobin, T1D=tyipe 1 diabetes, T2D=type 2 diabetes You are asked to discuss the possible adverse effects of GH treatment with a nurse who will be training patients how to start on therapy. To help her better understand risks of therapy and direct her discussions to specific subgroups of patients, you review the likelihood of adverse effects based on disease. Of the following United States Food and Drug Administration (FDA)-approved indications for GH treatment, the MOST likely slightly increased adverse effects are A. adrenal insufficiency and scoliosis in Noonan syndrome B. glucose intolerance and pseudotumor cerebri in children born small for gestational age C. glucose intolerance and scoliosis in Turner syndrome D. hypothyroidism and slipped capital femoral epiphysis in short stature homeobox deficiency E. type 1 diabetes and thyroid disease in idiopathic short stature Correct Answer: C GH therapy was developed in the middle of the last century to treat patients who had severe GH deficiency. Initially cadaveric in origin, the limited supply of GH, which was regulated by the National Pituitary Agency, led to limitations in treatment to patients who had the greatest need for such therapy. With the advent of recombinant DNA technology and subsequent approval of recombinant human GH (rHGH) in 1985, a theoretically limitless supply of GH became available and the FDA-approved indications expanded. Generally believed to be safe, GH may accentuate predisposition to diseases in specific patient subgroups. For example, the incidence of scoliosis and glucose intolerance is increased with GH administration to patients with Turner syndrome, who already have an increased likelihood of these 2 conditions. After the initial approval for GH therapy for GH deficiency, pediatric approvals expanded to: Chronic renal failure Short stature associated with Turner, Noonan, and Prader-Willi syndromes Short stature in those children who were born small for gestational age and did not experience catch-up growth Short stature homeobox deficiency Idiopathic short stature (ISS) ISS is defined as a condition in which the height of an individual is more than 2.25 standard deviations (SD) below the corresponding mean height for a given age, sex, and population group without evidence of systemic, endocrine, nutritional, or chromosomal abnormalities. ISS is believed to reflect myriad underlying disorders, many of which have yet to be defined, as well as familial growth patterns. Of all patients presenting with short stature below -2 SD in height, most are considered to fall into this “nondiagnostic” category. This subgroup of patients has variable responses to therapy, with a mean expected response of approximately a gain of 1 to 2 inches in height over pretreatment predicted height. Initiation of GH therapy should be pursued after appropriate evaluation to exclude nonendocrine and non-GHremediable causes of short stature. When that evaluation leads to categorization of a patient into one of the previously noted diagnostic groups, a full discussion of possible adverse effects of GH therapy should be undertaken with the family. Adverse effects of GH therapy fall into those that occur during treatment and possible long-term sequelae. The most often seen are local injection site reactions, mild headache, and muscle and joint pain. More severe headaches, a manifestation of pseudotumor cerebri, may occur with an incidence of up to 27 in 100,000. Generally occurring within the first few months of therapy, this is more common in children who have congenital GH deficiency and Turner syndrome. It typically resolves with discontinuation of GH, which may then be reinitiated with dose titration to avoid headaches. Some reports suggest that the likelihood of slipped capital femoral epiphysis may be greater in patients treated with GH than in the general population. Generally associated with obesity and rapid growth rate, slipped capital femoral epiphysis is more likely to occur in the first few months of therapy. Similarly, the growth response to GH may exacerbate a preexisting tendency to scoliosis, which is even more likely in children who have Prader-Willi or Turner syndromes, who are already predisposed to this condition. Children who have Prader-Willi syndrome should be evaluated for sleep apnea before initiation of GH treatment because this therapy has to potential to cause adenotonsillar hypertrophy. Because GH is a counterregulatory hormone for glucose metabolism, abnormalities in glucose homeostasis are a theoretical adverse effect of therapy that has garnered much attention. Children who are born small for gestational age and girls who have Turner syndrome have inherent risks of T2D, so these populations have been evaluated closely. Mean HbA1c values may increase in patients receiving GH, as does insulin production. Although frank T1D and T2D have been seen in children receiving GH treatment, the mild increase in T2D is believed to be an acceleration of the already ongoing process of insulin resistance. When specifically analyzed in a large controlled trial of GH in a small-for-gestational age population, the insulin resistance resolved once GH was discontinued. Central hypothyroidism may be unmasked in patients receiving GH who have hypothalamic-pituitary-thyroid axis abnormalities. Similarly, because GH inhibits 11ß-hydroxysteroid dehydrogenase-1, increased cortisol replacement doses may be required in patients receiving GH therapy. The concern for long-term effects of GH therapy has always been present. At this time, taking into account the limitations of long-term follow-up in this group of patients, there appears to be no increased risk of malignancy among those who had no previously identified risk. There does not appear to be an increased likelihood of recurrence of primary malignancy among patients treated with GH. Because many treated patients are survivors of childhood cancers, the risk of second malignancies as well as primary cancers has been studied. A history of cranial irradiation may be associated with an increased risk of meningioma, and acute lymphocytic leukemia may be associated with an increased risk for bone cancer among patients treated with GH. Recently, concern has been raised about an increase in mortality among adults treated with GH during childhood. At this time, data from retrospective studies are conflicting, regulatory agencies have not recommended changes in prescribing habits, and further data are being collected. Question: 4 Abbreviations ACTH=corticotropin, CAH=congenital adrenal hyperplasia, T4=thyroxine, TSH=thyrotropin A 16-year-old girl is admitted to the hospital after she presented to the emergency department with dizziness and near-syncope. She has been relatively healthy, except for the diagnosis of hypothyroidism at 13 years of age for which she has been taking 50 mg levothyroxine daily. She has had a few episodes of dizziness during the last 3 months, which were attributed to stress by her pediatrician. Her schedule consists of 3 advanced placement classes besides her usual school work, cheerleading, running track, and Irish dancing. She is involved in school theater and heads the school’s environmental club. On physical examination, the slightly tan adolescent is in no acute distress, her blood pressure is 90/65 mm Hg, and her heart rate is 90 beats/min. Her height and weight are at the 50th and 25th percentiles, respectively. Her thyroid gland measures 3 cm in length for each of the lobes, and she has Tanner 5 breasts and pubic hair. Laboratory testing is performed before any therapeutic interventions, and results include: Sodium, 130 mEq/L (130 mmol/L) Potassium, 5.6 mEq/L (5.6 mmol/L) Chloride, 110 mEq/L (110 mmol/L) Bicarbonate, 19 mEq/L (19 mmol/L) Urea nitrogen, 16 mg/dL (5.7 mmol/L) Creatinine, 0.7 mg/dL (61.9 mmol/L) Glucose, 72 mg/dL (4 mmol/L) T4, total, 7.4 mg/mL (126.5 nmol/L) Free T4, 1.2 ng/dL (15.5 pmol/L) TSH, 5.9 mIu/L Erythrocyte sedimentation rate, 29 mm/hr White blood cell count, 6.5x10³/mL (6.5x109/L) Hemoglobin, 14.7 g/dL (147 g/L) Hematocrit, 44% Platelets, 173x10³/mL (173x109/L) Urinalysis, normal, except for 3 to 5 red blood cells/high-power field and trace protein Of the following, the MOST likely diagnosis is A. autoimmune adrenalitis B. autoimmune hypophysitis C. Gordon syndrome D. Liddle syndrome E. type 1 diabetes mellitus Correct Answer: A The girl described in the vignette most likely has hypoaldosteronism (hyperkalemia, hyperchloremic metabolic acidosis) associated with primary adrenal insufficiency due to autoimmune adrenalitis. The mild hyperpigmentation, hypotension, history of dizziness, and prior diagnosis of primary hypothyroidism all suggest primary adrenal insufficiency as a cause for her hypoaldosteronism. In addition, because isolated hypoadrenalism is usually not associated with significant sodium wasting, primary adrenal insufficiency should be considered as a cause for hypoaldosteronism when mild hyponatremia is present. Cortisol deficiency indirectly stimulates vasopressin release, causing water retention and a reduction in serum sodium. Thus, significant hyponatremia is usually associated with combined aldosterone and cortisol deficiency. Mineralocorticoid replacement therapy with fludrocortisone is effective to correct the electrolyte abnormalities. Hypoaldosteronism should be considered in any patient presenting with hyperkalemia and mild hyperchloremic metabolic acidosis not caused by renal disease/failure or the use of potassium–sparing diuretics. Isolated hypoaldosteronism is most often seen in those who have either renal insufficiency due to diabetic nephropathy or interstitial nephritis and glomerulonephritis. Other causes include a variety of medications: nonsteroidal antiinflammatory drugs, cyclosporine, angiotensin-converting enzyme inhibitors, heparin, and certain antibiotics (trimethoprim). Hypoaldosteronism can also be caused by a number of inherited disorders. Congenital isolated hypoaldosteronism is caused by a defect in aldosterone synthase (CYP11B2), an enzyme in the aldosterone biosynthetic pathway. Two specific abnormalities have been described, illustrative of the 3 different enzymatic functions exerted by aldosterone synthase. Congenital isolated hypoaldosteronism type 1 is characterized by impaired hydroxylation of corticosterone at the 18-carbon position. Congenital isolated hypoaldosteronism type 2 is characterized by impaired conversion of the 18-hydroxyl group to an aldehyde. Both lead to deficient aldosterone production (Figure 1). Determination of the precursor-to-end product ratio allows for the differential diagnosis between the two types (18-hydroxycorticosterone-to-aldosterone is <10 in type 1 versus >100 in type 2 congenital isolated hypoaldosteronism). Clinically, these disorders present in infancy with failure to thrive, salt wasting, and recurrent hypovolemia; the clinical picture may improve with age. Hypopituitarism does not cause hypoaldosteronism because ACTH does not play a major role in the regulation of aldosterone secretion; mild hyponatremia can be seen in hypopituitarism due to secondary cortisol deficiency. Liddle syndrome and Gordon syndrome are 2 forms of pseudohypoaldosteronism: type 1 and type 2, respectively. In both, resistance to the actions of aldosterone is the cause of loss of aldosterone effect. Pseudohypoaldosteronism type 1 or Liddle syndrome can be inherited in 2 patterns. In the autosomal recessive form, the defect lies in the epithelial sodium channel of the renal collecting tubules (Figure 2). Autosomal recessive pseudohypoaldosteronism type 1 presents with excessive loss of salt in the urine and high sodium concentrations in saliva, sweat, and stool. Pseudohypoaldosteronism type 1 is especially serious in the neonatal period and can be life-threatening. Hyponatremia, hyperkalemia, and increased plasma renin activity make the diagnosis. Serum aldosterone concentrations are also high. Respiratory tract infections are common as is a lung disease similar to cystic fibrosis. Clinical improvement can be seen at older ages. Therapy involves aggressive salt supplementation plus control of hyperkalemia. Autosomal dominant pseudohypoaldosteronism type 1 is a milder variant caused by a loss-of-function mutation in the gene encoding for the mineralocorticoid receptor. Although often clinically much less symptomatic than the autosomal recessive form, severe cases have presented in neonates with hyponatremia, hyperkalemia, and failure to thrive. Gordon syndrome or pseudohypoaldosteronism type 2 is characterized by hyperkalemia, metabolic acidosis, and low to low-normal plasma renin activity and aldosterone concentrations. It is caused by mutations affecting either WNK4 or WNK1, both proteins important for normal function of the thiazide-sensitive Na-Cl cotransporter (Figure 3). Affected patients become hypertensive. Treatment, including dietary sodium restriction and hydrochlorothiazide administration, is very effective. Fludrocortisone (in higher doses) is also an effective therapy in isolated hyporeninemic hypoaldosteronism, although the coexistence of hypertension and edema may prohibit its use. In that case, the use of a thiazide diuretic and a low-potassium diet help to control the hyperkalemia. Question: 5 Abbreviations GH=growth hormone, GHRH=growth hormone-releasing hormone, IGF-1=insulinlike growth factor 1, MRI=magnetic resonance imaging, T3=triiodothyronine, TSH=thyrotropin A 4-1/12-year-old boy comes to your clinic for evaluation of short stature. You construct his growth curve using all available past data (Figure). The boy appears well and has no abnormal physical examination findings or neurologic delay, but his grandmother reports that initially when he returned to her, he was hoarding food. He lived with his biological mother and maternal grandmother until age 3 years. He then lived in another state with his mother and her boyfriend for 15 months but was legally placed back into the grandmother’s custody 2 months ago. Laboratory test results from a health maintenance visit 7 weeks ago include an IGF-1 of 45 ng/mL (5.9 nmol/L), free T4 of 1.2 ng/dL (12.5 pmol/L), and TSH of 1.1 mIU/L. His bone age is read as typical for a 4-year-old male. Of the following, the MOST appropriate next step in endocrine evaluation for this boy is A. administration of bedtime tryptophan to increase overnight melatonin B. brain MRI with contrast C. complete blood count, urinalysis, and repeat IGF-1 D. GH stimulation test E. morning fasting cortisol assessment and repeat thyroid function tests Correct Answer: C Psychosocial dwarfism is a rare disorder in which a pathologic environment of caregiver neglect leads to endocrine dysfunction and growth failure. Diagnosing the condition can be challenging because results of evaluation often mimic those of hypopituitarism. There is likely a continuum ranging from an affective deprivation syndrome to the extreme of psychosocial dwarfism with nonorganic failure to thrive. The child described in the vignette hopefully is recovering from a 15-month situation of poor nutrition and suspected maternal neglect. He requires close monitoring of his growth, discussion of concerns with his custodial grandmother, and careful observation of his IGF-1 trends and linear growth. Further endocrinologic assessment (GH stimulation, cortisol, thyroid function, and MRI) will likely be unnecessary if the boy experiences rapid growth in a secure setting. Tryptophan is not a therapeutic option. Accurate family height information or medical histories may be unavailable in difficult social or foster care situations. Furthermore, children who have experienced neglect or abuse are at higher risk for past exposures to human immunodeficiency virus, fetal alcohol syndrome, malnutrition, head trauma, and sleep deprivation. A careful physical examination that includes review of the growth curve is required, with attention to dysmorphisms and body proportions to rule out underlying genetic syndromes. Among the findings suggestive of psychosocial growth retardation are long bone growth arrest lines and temporary cranial suture widening. Further laboratory evaluations are guided by history and physical examination findings. Potential additional laboratory tests include a complete blood count, urinalysis, stool for ova and parasites, fecal fat, and possibly a sweat chloride test. Laboratory findings can overlap for children who have failure to thrive due to inadequate calories or protein in their diet and those who have psychosocial dwarfism. Children who have familial short stature track along the growth curve within the range of their mid-parental height. In addition, they have normal growth velocity as well as IGF-1 and IGF binding protein-3 values and their bone ages are consistent with chronologic age. Children who have constitutional growth delay typically experience a deviation from the growth curve by 2 years of age, followed by relatively normal growth velocity parallel to the growth curve. Their short stature is more apparent at puberty when the delayed pubertal growth spurt and delayed bone age are clues to the cause of the short stature. Children who have psychosocial dwarfism are often diagnosed in hindsight when their spontaneous rapid recovery of GH production coincides with a growth spurt that occurs in response to removal from their stressful environment. They typically experience a suspicious rapid deceleration from their improved growth rate if they return to the stressful environment. Early diagnosis and removal from the pathologic environment can allow for catch-up growth and normalization of sleep patterns and behavioral disorders if recognized promptly. The duration of the stress correlates with the reversibility of its negative effects on behavior. Psychosocial dwarfism has been categorized into several subtypes (Table). Neglected or abused infants (up to 18 months of age) tend to have growth failure that is primarily related to caloric deprivation, no bizarre behavior, and often parental depression. Older children are prone to alterations of GH synthesis or release. The classic behavior findings include food hoarding and stealing or rummaging for food in trash. The prognosis for type II is guarded when it is diagnosed late in childhood. The exact mechanism of the impact of a severely pathologic caregiver on the hypothalamic GHRH secretion as well as the response to GH has not been elucidated. It is likely modulated by hypothalamic dopamine, catecholamine, and neuropeptide Y and attenuation of GHRH secretion during stress. Children who experience greater growth delays related to severe emotional maltreatment tend to have higher early morning cortisol values and alteration of melatonin, which contribute to mood disorders. There is also evidence that intracerebral concentration of tryptophan and serotonin influence the behavioral neuroendocrine response to stress and melatonin production. Historical accounts dating back to May 26, 1828, describe a 17-year-old male named Kasper Hauser who exhibited findings of severe dwarfism and flat affect with no language as he exited the Haller Gate of a concentration camp in Nuremburg, Germany. Following his release, the young man experienced a dramatic growth spurt accompanied by social development to become a “peculiar” adult with limited intellect. This is likely one of the earliest accounts of the constellation of findings now known as psychosocial dwarfism. Question: 6 Abbreviations: ACTH=corticotrophin, CT=computed tomography, MEN=multiple endocrine neoplasia, TSH=thyrotropin, T4=thyroxine An 11-year-old girl is referred to you for evaluation of a thyroid nodule. She sustained a concussion at a soccer match last week and underwent a head CT scan as part of her evaluation. Although results of the scan were negative, the imaging revealed a thyroid nodule measuring approximately 0.5 X 0.3 X 0.2 cm in the left lobe of the gland. Her pediatrician ordered ultrasonography, which confirmed the presence of a 0.4 X 0.3 X 0.2-cm nodule that had a heterogeneous appearance without calcifications. There was no surrounding lymph node enlargement in the central or lateral neck compartments. Thyroid function tests revealed a TSH of 1.84 mIU/mL and free T 4 of 1.46 ng/dL (18.8 pmol/L). The girl has no history of known radiation exposure. Her paternal grandfather had thyroid cancer and underwent repeated neck surgeries for “calcium problems.” The girl’s father has been healthy but recently developed high blood pressure that has not responded well to treatment. Physical examination of the girl reveals a blood pressure of 104/68 mm Hg. You are unable to definitively palpate any nodules within the thyroid gland and find no accompanying lymphadenopathy. Of the following, the MOST appropriate initial management for this girl is to A. measure serum calcitonin B. measure thyroid autoantibodies C. perform fine-needle aspiration D. perform total thyroidectomy E. repeat ultrasonography in 6 months The family history for the girl described in the vignette is very suspicious for a MEN syndrome. Specifically, the repeated neck surgeries for “calcium problems” and thyroid cancer in the grandfather and the hypertension unresponsive to therapy in the father suggest possible diagnoses of hyperparathyroidism, medullary thyroid carcinoma (MTC), and pheochromocytoma. This combination of endocrine tumors is found in MEN-2A. This family history warrants an aggressive screening approach for MTC. Along with genetic testing for RET mutation analysis, assessment of serum calcitonin is a critical part of the initial diagnostic evaluation and may aid in determining whether additional imaging studies are needed as well as the potential surgical approach. Measurement of thyroid autoantibodies is a consideration during evaluation of thyroid nodules in children because nodules can rarely occur as part of autoimmune thyroiditis. However, this assessment has no bearing on the diagnostic and therapeutic considerations for MTC. Fine-needle aspiration (FNA) is an important component of the evaluation of some thyroid nodules in children. The diagnostic accuracy of FNA varies widely in clinical practice and involves additional risks of sedation for the procedure for children. Sensitivity rates for detecting MTC using FNA vary from 30% to 90% in adults. In addition, the risk for false-negative results is increased during procedures on smaller nodules such as the one presented in the vignette. Total thyroidectomy is recommended for patients who have known high-risk RET mutations, low-risk mutations accompanied by elevation in calcitonin, or in situations of known tumor involvement following FNA. Although some low-risk thyroid nodules may be followed clinically with ultrasonography, this girl should undergo additional evaluation based on her family history. MTC arises from the parafollicular C-cell of the thyroid gland. MTC represents approximately 4% of all thyroid cancers in the United States and is identified in approximately 0.3% of all thyroid nodules. MTC that occurs in childhood is almost always due to germline activating mutations of the RET pro-oncogene. The estimated incidence of MTC among the pediatric population is 0.5 cases per 1 million per year. Early diagnosis and aggressive surgical resection are crucial to reducing morbidity and mortality. MTC occurs in the context of the MEN-2A phenotype in 90% to 95% of cases, with a minority comprising part of MEN-2B or as part of a familial form of isolated inherited MTC, although most of these cases are now considered to be part of the spectrum of MEN-2A. MTC in MEN-2A tends to be identified in the fourth to sixth decades of life but may occur among children. MTC develops in nearly 100% of individuals who have MEN-2B, but unlike in MEN-2A, tends to occur at younger ages and is more aggressive due to a specific Met918Thr mutation of the RET gene. Guidelines for diagnosis and management of MTC were recently updated by the American Thyroid Association. Presently, RET gene mutations are classified into 1 of 4 levels (A through D) (Table). All mutations associated with MEN-2B are considered to be in the level D (most severe) category, and prophylactic thyroidectomy is recommended within the first postnatal year. The most common MEN-2A mutation (codon 634) is associated with MTC in 50% of children and classified as a level C risk. In this grouping of mutations, thyroidectomy is recommended by 5 years of age. Class A and B mutations are generally considered lower risk, and prophylactic thyroidectomy is considered on a case-by-case basis. Calcitonin is the most sensitive marker for diagnosis of MTC and its precursor cell type, C-cell hyperplasia. In adults, concentrations less than 10 pg/mL (2.9 pmol/L) are considered negative, values of 10 to 20 pg/mL (2.9 to 5.8 pmol/L) are indeterminate, values of 20 to 100 pg/mL (5.8 to 29.2 pmol/L) are suspicious, and greater than 100 pg/mL (29.2 pmol/L) values are strongly suggestive of MTC. Other studies have demonstrated that MTC is found in an intrathyroid location only when basal calcitonin concentrations are 60 pg/mL (17.5 pmol/L) or less among known gene carriers; values above this are generally associated with metastasis outside the thyroid capsule. In the postoperative patient, basal and pentagastrin-stimulated values of less than 10 pm/mL (2.9 pmol/L) are considered consistent with surgical cure, 10 pg/mL to 30 pg/mL (2.9 to 8.8 pmol/L) is indeterminate, and more than 30 pg/mL (8.8 pmol/L) is considered to represent residual disease. The doubling time of calcitonin values from the immediate postoperative period is highly predictive of disease-free survival. Although calcitonin concentrations are generally higher in larger nodules, cases of normal calcitonin values in the setting of large MTC nodules have been described, so calcitonin values must be interpreted in the context of other clinical and laboratory variables. In addition, knowledge of the assay used is critical because variability in calcitonin values, depending upon the assay employed, is common. Current assays using a 2-step chemiluminescent immunometric assay are more specific for the intact calcitonin peptide and reduce interference from other cross-reacting substances. Thus, calcitonin is an important diagnostic marker, both for detecting MTC in children who have sporadic nodules and monitoring children who have known RET mutations (risk levels A and B) in whom prophylactic thyroidectomy is not clearly indicated at a young age. Current guidelines suggest that a basal calcitonin concentration of less than 40 pg/mL (11.7 pmol/L) in a child who has a nodule that measures less than 0.5 cm in diameter is not indicative of metastatic disease. Results of calcitonin assays should be interpreted using age-appropriate norms. Calcitonin concentrations are higher in newborns up to the age of 6 months, in whom values less than 40 pg/mL are considered normal. From 6 months to 3 years, a normal range is less than 15 pg/mL and beyond 3 years, values are indistinguishable from adults (normal, <10 pg/mL). Carcinoembryonic antigen (CEA), chromogranin A, vasoactive intestinal peptide, somatostatin, and ACTH can all be expressed and secreted by the C cell of the thyroid gland, but calcitonin remains the primary biochemical marker used in detection, tumor staging, and follow-up surveillance. CEA concentrations are frequently used in conjunction with calcitonin in postoperative surveillance. Procalcitonin and calcium-stimulated calcitonin values are promising alternatives but need further study, particularly in the pediatric population, before they are used in the laboratory evaluation of MTC. MTC typically has an ovoid appearance on ultrasonography, and some studies have demonstrated increased cystic components compared to other forms of thyroid cancer. They are otherwise very difficult to distinguish on ultrasonography from other benign and malignant thyroid nodules. Assessing calcitonin concentrations in a routine thyroid nodule or in the presence of nodular goiter remains controversial. Expert panels from the United States have recommended neither for nor against measuring calcitonin in this situation, but most European agencies are proponents. Much of the controversy surrounds the cutoff points for diagnosis and variability in the type of assay used. Lowering the diagnostic cutoff to improve sensitivity affects the specificity of the test, resulting in an increase in unnecessary procedures in individuals without disease or with C-cell hyperplasia only. The addition of pentagastrin-stimulated calcitonin concentrations can add to the positive predictive value of MTC diagnosis, but pentagastrin is not available in the United States. Question: 7 Abbreviations DHEA= dehydroepiandrosterone, FSH=follicle-stimulating hormone, hCG=human chorionic gonadotropin, LH=luteinizing hormone You are evaluating a 16-year-old girl who has irregular menstrual periods occurring every 6 to 10 weeks. She achieved menarche at about age 13 years. Of the following, the finding MOST characteristic of her cycles is the she has A. a greater than two-thirds risk of persistent oligoovulation B. a longer luteal phase than follicular phase C. a typical pattern for an adolescent girl D. low androstenedione concentrations E. normal LH concentrations Correct Answer: A The normal adult menstrual cycle averages 28 days, with a 90% confidence interval of 22 to 40 days. During the first 2 years after menarche, menstrual cycles are more irregular and longer by several days; about 50% of the cycles are anovulatory or have attenuated ovulation. Two thirds of adolescent girls have mature menstrual cyclicity by 1 year postmenarche and about 90% by 2 years postmenarche. Failure to establish a mature menstrual pattern by 2 years postmenarche or to sustain a normal pattern after one has been established carries a greater than twothirds risk of persistent oligoovulation. Even within the first year postmenarche, no menses for more than 90 days for even one cycle, intervals of less than 21 days, bleeding for more than 7 days, or heavy flow requiring pad or tampon changes more than every 1 to 2 hours are abnormal findings and should be investigated. Adolescents who have anovulatory cycles have higher serum LH, testosterone, and androstenedione concentrations, but it is not clear if this is causal of or the result of anovulation. The menstrual cycle has 2 phases: the follicular and the luteal (Figure). The follicular phase starts with the onset of menses. Gonadotropin concentrations increase, and in the early follicular phase, FSH predominates while nocturnal LH pulsation is low. As LH pulsation increases, the preovulatory follicles slowly begin to secrete estradiol, with estradiol concentrations gradually rising while FSH concentrations fall. When the plasma concentration of estradiol rises to greater than 200 to 300 pg/mL (734 to 1,101 pmol/L) for 36 hours, positive feedback results in the midcycle gonadotropin surge. Estradiol induces progesterone receptor expression in the hypothalamus and pituitary. Progesterone increasing to about 100 ng/dL assists the LH surge, with LH pulses becoming larger in amplitude and slower in frequency, bringing about the FSH surge. The LH surge results in ovulation from the dominant ovarian follicle. The follicular phase varies in duration, averaging 14 days. Menstrual cycle variability is almost entirely due to variations in follicular phase length. The empty ovarian follicle converts into a corpus luteum, which initiates the luteal phase. Estrogen concentrations decrease when the follicle is disrupted. As the corpus luteum forms, progesterone concentrations increase steadily and are sustained at greater than 500 ng/dL for several days in the mid-luteal phase, along with lesser but significant increases in estradiol and 17-hydroxyprogesterone. In response to the high progesterone concentrations, gonadotropin pulsation slows. The progesterone and estradiol maintain the endometrial layer. In the absence of hCG production from a pregnancy, the corpus luteum is destroyed, progesterone and estradiol concentrations decrease, and the endometrium sloughs, resulting in menses. As FSH begins to rise out of proportion to LH, FSH induces follicular growth, and the next menstrual cycle begins. The luteal phase lasts 14±1 (standard deviation) days but does not occur in anovulatory cycles. The ovary has 3 regions. The outer cortex contains the germinal epithelium and the follicle-containing oocytes, which are embedded in stroma. Stromal tissue appears to respond to LH and hCG and produce androstenedione. The medulla is central and also consists of stroma. The hilum, where the ovary attaches to the mesovarium, contains hilus cells. Hilus cells are similar to the Leydig cells of the testis and can become active in steroidogenesis or form androgen-secreting tumors. The ovaries secrete pregnenolone, progesterone, 17alpha-hydroxyprogesterone (17alpha-OHP), DHEA, androstenedione, testosterone, estrone, and estradiol, although the specific ovarian cells involved are not known. Pregnenolone is the precursor of all the steroid hormones. Estrone and estradiol are the major products of the preovulatory follicles; progesterone and 17alpha-OHP are the major products of the corpus luteum. The biologically active ovarian steroids are estradiol and progesterone. 17alpha-OHP is believed to have little or no biologic activity, and DHEA and androstenedione are not biologically active. DHEA is converted to androstenedione in the ovary and in peripheral tissues. Androstenedione acts as a precursor for estrone and testosterone in extraglandular tissues such as adipose tissue and skin as well as for estrogen in the granulosa cells. Estrone and testosterone may be converted to estradiol and dihydrotestosterone, respectively, in target tissues for biologic effect. Question: 8 Abbreviations DXA=dual-energy x-ray absorptiometry, PTH=parathyroid hormone You are contacted by a state agency regarding a 3-year-old toddler who has a history of several fractures. Because of concern about potential child abuse, the primary care physician asks for guidance regarding the next step in this patient’s evaluation. Of the following, the BEST test to perform is A. alkaline phosphatase B. DXA C. PTH D. skeletal survey E. urine telopeptide of type I collagen Correct Answer: D Not every child who has fractures requires a complete evaluation, but in the proper clinical circumstance, an initial evaluation should rule out diseases of bone and mineral metabolism and look specifically at markers of collagen production as well as bone turnover. Radiographic findings on skeletal survey that indicate a need for further evaluation include multiple fractures in various stages of healing, wormian bones, codfish vertebrae, and osteopenia. Among the underlying bone diseases that may be related to repeat fractures is osteogenesis imperfecta (OI). This group of diseases leads to greater bone fragility, with an increased propensity to fracture, as well as decreased bone mass. Contrary to the more common bone diseases of childhood, the hallmark of OI is defects in collagen synthesis, not in bone and mineral metabolism/calcification, as is seen in various types of rickets. Specific collagen-related markers of bone formation may be elevated in subtypes of OI. Urinary n-terminal telopeptide of type I collagen can be elevated in OI but is a nonspecific finding. As in rickets, healing fractures, or metabolic bone disease, alkaline phosphatase concentrations are elevated. However, this cannot be assessed in the presence of recent fracture or bone healing. Because OI is not a specific disorder in bone mineralization or calcium metabolism, vitamin D and PTH values cannot aid in diagnosing OI. Findings of bone densitometry are nonspecific. Ultimately, genetic testing for specific mutations in type I collagen via analysis of the COL1A1 and COL1A2 genes can yield an OI diagnosis in up to 90% of patients. Skin biopsy for analysis of collagen in fibroblasts may also be diagnostic but is less frequently indicated. With an incidence of approximately 1 in 20,000, OI is divided into 9 subtypes that have varying clinical presentations and severity. All of the subtypes have defects in either synthesis of the collagen chains or posttranslational modifications of type 1 collagen. Because type 1 collagen is also expressed in a variety of connective tissues, the other occasionally prominent clinical finding of blue sclerae, due to a defect in corneal collagen, is also seen in subtypes of the disease. Most patients who have OI have an autosomal dominant mutation in COL1A1 or COL1A2. Although an absolute correlation between disease type and severity does not exist, the specific classes of mutations correlate may predict the degree of impairment of bone formation. The disease is often subclassified as mild (type I), moderate (types III through IX), severe (III), or lethal perinatal (type II) (Table). Disease can also be categorized in subgroups of infantile, childhood, or adult presentation. Familial disease is common, but not every family member who has a specific mutation has the same disease severity. In addition to a family history of fractures or OI, other clues in either the patient or family members that may point to the diagnosis of OI include easy bruising, conductive hearing loss, hyperextensibility, and early and significant osteoporosis. A history of poor dentition may also be present. Over the past decade, treatment has evolved to include the use of bisphosphonates in the treatment of OI. These potent inhibitors of bone resorption and turnover encompass analogs of pyrophosphate. They have been used in all classes of OI except type VI, which is a defect in bone mineralization. Marked decreases in bone pain and reduction in fracture rate have been noted in treated patients, and ongoing clinical trials have also noted positive effects on normalizing radiographic findings of bone. Increased mobility and return to function has also been reported. March Question: 1 Abbreviations: CAH=congenital adrenal hyperplasia, DHEA-S=dehydroepiandrosterone sulfate, FSH=follicle-stimulating hormone, GnRH=gonadotropin-releasing hormone, hCG=human chorionic gonadotropin, LH=luteinizing hormone, 17-OH progesterone=17-hydoxyprogesterone, TSH=thyrotropin, T4=thyroxine You are seeing an 8-year-old boy in the endocrinology clinic because he has developed acne, body odor, and Tanner stage 3 pubic hair. On physical examination, he has asymmetric testes with volumes of 2 to 3 mL on the right and 5 mL on the left. No abnormal masses are palpable, but he reports discomfort upon palpation of the left testis. Testicular ultrasonography confirms a solid mass in the left testis. Of the following, the laboratory test results that are MOST consistent with this boy’s presentation are A. LH FSH Testosterone Androstenedione DHEA-S 0.02 mIU/mL 0.36 305 ng/dL (10/58 nmol/L) 165 ng/dL (5.76 nmol/L) 65 mg/dL (1.76 (0.02 IU/L) mIU/mL mmol/L) (0.36 IU/L) B. C. D. E. 0.02 mIU/mL 0.6 mIU/mL 320 ng/dL (11.10 nmol/L) (0.02 IU/L) (0.6 IU/L) 175 ng/dL (6.11 nmol/L) 673 mg/dL (18.17 mmol/L) 0.1 mIU/mL (0.1 0.9 mIU/mL 18 ng/dL (0.62 nmol/L) IU/L) (0.9 IU/L) 1.9 mIU/L (1.9 1.8 mIU/mL 156 ng/dL (5.41 nmol/L) IU/L) (1.8 IU/L) 0.02 mIU/mL 0.36 (0.02 IU/L) mIU/mL 74 ng/dL (2.58 nmol/L) 134 mg/dL (3.62 mmol/L) 97 ng/dL (3.39 nmol/L) 65 mg/dL (1.76 mmol/L) 10 ng/dL (0.35 nmol/L) 42 ng/dL (1.47 nmol/L) 65 mg/dL (1.76 mmol/L) (0.36 IU/L Correct Answer: A The laboratory measurement of sex steroids plus gonadotropins can often help distinguish virilizing gonadal tumors from adrenal tumors or increased sex steroids associated with central precocious puberty (PP). In boys, aside from a detailed history, auxology, and physical examination, the evaluation of PP associated with advanced bone age is further guided by the testicular examination. When both testes appear prepubertal (<4 mL), an adrenal source for the PP is more likely. Exposure to exogenous androgens is also possible. If one of the testes is larger than the other, further evaluation for a possible gonadal tumor is required. With bilateral enlarged testes, the PP more likely has a central cause. Measurements of hCG (in boys) and prolactin, 17-OH progesterone, T4, and TSH may also be helpful in the diagnosis of unexplained PP. For girls presenting with masculinizing symptoms, sex steroid determination can indicate the severity of the virilizing condition, and measuring 17-OH progesterone concentrations can screen for nonclassic CAH, a relatively common cause of androgen-mediated signs in girls. Measuring LH, FSH, and estradiol are not helpful in the evaluation of girls who have virilizing PP. The boy described in the vignette has precocious puberty with a unilateral testicular mass suggesting a testicular tumor. The laboratory findings listed in option A, with prepubertal LH and FSH, elevated testosterone and androstenedione, and normal DHEA-S, are most likely for this boy and suggest the presence of a Leydig cell tumor. An androgen-secreting tumor, such as a Leydig cell tumor, should be considered in any prepubertal male who has asymmetric testicular enlargement. If a mass cannot be detected on palpation or ultrasonography, the larger testis should probably still be considered for biopsy, especially if there is evidence of ongoing testis growth. The laboratory results in option B are most suggestive of an adrenal tumor, with elevated testosterone and DHEAS concentrations. Boys with PP who have an adrenal cause generally do not have testicular enlargement (testicular volume <4 mL or length <2.5 cm in longest diameter). The results shown in option C suggest premature adrenarche, with a mildly elevated DHEA-S appropriate for pubic hair Tanner stage. Values for other sex steroids are normal or very mildly elevated. The laboratory results listed in option D indicate a central cause for increased sex steroids. A randomly obtained LH that is greater than 0.3 mIU/mL (0.3 IU/L) is highly suggestive of central PP, and a stimulation test using GnRH (not currently available in the United States) or GnRH analog may further confirm LH-dominant gonadotropin secretion. The values listed in option E would be normal for an 8-year-old prepubertal boy. Question: 2 Abbreviations: CAH=congenital adrenal hyperplasia, GH=growth hormone, IGF-1=insulinlike growth factor-1 You are evaluating a 14-year-old girl who had ambiguous genitalia at birth. Postnatal evaluation excluded CAH as well as ovarian or adrenal tumor. She was found to have aromatase deficiency and was asked to return for evaluation at 9 to 10 years for induction of puberty, but she failed to do so. Today she expresses concern about her lack of breast development. Physical examination documents absent breast development, Tanner stage 4 pubic hair, abundant axillary hair, and clitoral enlargement. Testosterone measures 95 ng/dL (3.3 nmol/L) and estradiol measures less than 10 pg/mL (36.71 pmol/L). Of the following, compared to her age-matched peers, the MOST likely findings for this girl are Bone Age A. Height GH Secretion IGF-1 Advanced Tall Normal Normal B. Delayed Short Normal Normal C. Advanced Short Low Low D. Delayed Short Low Low E. Advanced Tall Increased Increased Correct Answer: D GH secretion rates are relatively stable in prepubertal children, but during puberty, a 1.5- to 3-fold increase occurs in the pulsatile secretion of GH, which is mirrored by at least a 3-fold increase in IGF-1. Several lines of evidence suggest that estrogen promotes the pubertal increase in GH in both males and females: Estrogen priming increases GH response to pharmacologic stimuli in both sexes. There is a positive correlation between GH and estrogen values in prepubertal boys and girls. Testosterone (an aromatizable androgen) induces an increase in GH secretion and IGF-1 concentrations in boys while nonaromatizable androgens (eg, oxandrolone and dihydrotestosterone) do not. The antiestrogen tamoxifen abrogates the stimulatory effect of testosterone on GH secretion. Because the girl described in the vignette has aromatase deficiency and consequent inability to convert testosterone to estradiol, her GH secretion and IGF-1 values would be expected to be low compared to her agematched peers (most of whom would be in late puberty). Furthermore, because estrogen plays the major role in the pubertal growth spurt and skeletal maturation, she would also be expected to be short relative to her peers, with a delayed bone age. Although estrogen stimulates GH secretion at the hypothalamic and pituitary levels, estrogen also modulates GH action at the level of the liver, in that oral estrogen appears to impair GH action (causing a decline in IGF-1) while transdermal estrogen increases circulating IGF-1 concentrations. Local generation of IGF-1 (by interaction of GH with the GH receptor at the chondrocyte) is the primary mechanism whereby GH induces linear growth. The original "somatomedin hypothesis" suggested that linear growth was mediated by circulating IGF-1 from the liver. However, later discoveries showed that many tissues produce IGF-1, and an autocrine/paracrine action of IGF-1 is a major determinant of postnatal body growth. Question: 3 Abbreviations: TSH=thyrotropin, T1D=type 1 diabetes You are consulted about a 2-month-old male infant who has severe intractable diarrhea, new-onset diabetes, and elevated TSH values on serum testing. He has had weight loss and mucoid, bloody diarrhea that worsened when he was switched from human milk to formula during his hospital admission 1 week ago. Serum glucose values have ranged from 200 to 300 mg/dL (11.1 to 16.7 mmol/L) since his admission. Genetic testing has confirmed an inactivating mutation of the FOXP3 gene. Of the following, the other clinical condition for which this patient is MOST at risk is A. chronic mucocutaneous candidiasis B. dermatitis C. eosinophilic enteropathy D. hypoparathyroidism E. myasthenia gravis Correct Answer: B The infant described in the vignette has a rare X-linked recessive syndrome with immunodysregulation, polyendocrinopathy, and enteropathy (IPEX). Of the clinical conditions listed, he is at greatest risk for dermatitis. Chronic mucocutaneous candidiasis, hypoparathyroidism, eosinophilic enteropathy, and myasthenia gravis are not described in IPEX. IPEX is caused by a loss-of-function mutation in FOXP3, a gene located on Xp11.23. FOXP3 is a member of the forkhead P family of transcription factors, encodes a protein with well-described functional domains, and plays an important role in CD4+T reg cell differentiation. A loss of FOXP3 leads to quantitative or functional T reg cell deficiencies, resulting in autoimmune disease and allergic inflammation. The family history in patients who have IPEX can be unremarkable because mutations are often sporadic and female carriers are frequently asymptomatic. The classic clinical triad in IPEX consists of autoimmune enteropathy, dermatitis, and autoimmune endocrinopathies. The enteropathy commonly is the initial manifestation and involves intractable, severe, watery diarrhea that is often mucoid or bloody. The diarrhea frequently worsens when the infant is switched from human milk to formula. Biopsy findings are nonspecific and include villous atrophy, crypt hyperplasia, and lymphocytic infiltration of the bowel mucosa. Eosinophilic predominance is not expected in this autoimmune enteropathy. The dermatitis seen in IPEX usually consists of an eczematous rash that is often severe and is seen in early infancy along with the enteropathy and endocrinopathies. Serum immunoglobulin E values can be markedly elevated. Psoriasis, alopecia universalis, and painful cheilitis are also described. The autoimmune endocrinopathies in IPEX most often include T1D and primary thyroiditis. The diabetes generally presents in the first postnatal year and may result in complete immune-mediated destruction of islet cells. Antiislet cell antibodies may be detected. Autoimmune thyroiditis is also common and can present at any age. Hypothyroidism with positive thyroid peroxidase and thyroglobulin antibodies is the most common thyroid disorder in IPEX, although cases of hyperthyroidism have been described. In addition to these classic findings, patients may have renal disease, neurologic abnormalities, pulmonary disease, immune-mediated cytopenias, and increased susceptibility to infections. Autoimmune polyglandular syndromes (APS) should be considered in any patient who has diabetes mellitus and primary thyroid disease. APS I, also known as autoimmune polyendocrinopathy, candidiasis, ectodermal dysplasia (APECED), is caused by an inactivating mutation in the gene coding for the autoimmune regulator transcription factor (AIRE). In this condition, autoreactive T-cells are not identified and deleted, resulting in autoimmune endocrinopathies and chronic mucocutaneous candidiasis. Primary adrenal insufficiency, primary hypogonadism, and hypoparathyroidism are the most common endocrinopathies, with diabetes mellitus and thyroiditis less commonly seen. Malabsorption is described in 25% of patients. APS II is much more prevalent than APS I. Primary adrenal insufficiency is the most common endocrinopathy and is the initial presentation in 50% of patients. T1D, autoimmune thyroid disease, and primary hypogonadism are also common. Vitiligo, myasthenia gravis, rheumatoid arthritis, and autoimmune hypophysitis are also described. Question: 4 Abbreviations PTH=parathyroid hormone, 25-OH vitamin D=25 hydroxyvitamin D, 1,25(OH)2 vitamin D=1,25 dihydroxyvitamin D You are evaluating a previously healthy 5-year-old girl who has a 6-month history of painful left lower leg swelling that started after a fall off of her bicycle. Tibial radiographs demonstrate periarticular calcified lesions. Laboratory evaluation shows Serum calcium, 9.8 mg/dL (2.45 mmol/L) Serum phosphorus, 8.1 mg/dL (2.61 mmol/L) PTH intact, 18 pg/mL (1.9 pmol/L) 25-OH vitamin D, 22 ng/mL (54.9 nmol/L) 1,25(OH)2 vitamin D, 75 pg/mL (180 pmol/L) Of the following, the genetic mutation that BEST accounts for this patient’s findings is A. activating mutation of GTP-binding protein that stimulates adenyl cyclase (GNAS1) B. activating mutation of UDP-N-acetyl-alpha-D-galactosamine: polypeptide N acetylgalactosaminyltransferase 3 (GALNT3) C. inactivating mutation of dentin matrix protein 1 (DMP1) D. inactivating mutation of fibroblast growth factor 23 (FGF23) E. inactivating mutation of phosphate regulating endopeptidase on the X chromosome (PHEX) Correct Answer: D The girl described in the vignette has hyperphosphatemia with elevated 1,25 (OH)2 vitamin D (calcitriol) values. Such findings may be caused by an inactivating mutation of the FGF23 gene, resulting in a condition termed familial tumoral calcinosis (MIM #211900). Familial tumoral calcinosis is an autosomal recessive disorder characterized by reduced activity of the FGF23 protein. Most often, this results from an inactivating mutation in the FGF23 gene. However, decreased FGF23 protein activity may also be seen with inactivating mutations of other genes crucial for normal FGF23 processing (GALNT3) and signaling (Klotho). Patients who have familial tumoral calcinosis have variable clinical findings. They may develop ectopic calcifications in the soft tissues around joints, in the long bones, and in the major blood vessels; may have ophthalmic or dental anomalies; or may be asymptomatic. Soft-tissue lesions may be precipitated by trauma. Laboratory evaluation reveals normal calcium and PTH concentrations, with elevated phosphorus and elevated or inappropriately normal calcitriol. Serum phosphorus concentrations are maintained through the actions of FGF23, PTH, and calcitriol. FGF23 is produced by the osteocytes and osteoblasts in response to calcitriol and increased serum phosphorus. After glycosylation by GALNT3, mature FGF23 is secreted into circulation. It acts through the FGF-Klotho coreceptor to increase renal phosphate excretion by reducing expression of the sodium-phosphate cotransporters (NaPi-IIa and NaPi-Iic) in the proximal renal tubule. FGF23 reduces serum phosphorus through its inhibitory effects on calcitriol synthesis and downregulation of renal phosphorus transporters. Accompanying the decreased FGF23 activity in familial tumoral calcinosis is increased renal phosphorus absorption and calcitriol synthesis. This latter finding is a distinguishing characteristic of this condition and may prevent the hypocalcemia and secondary hyperparathyroidism that is often seen in other cases of hyperphosphatemia (Table). High concentrations of circulating active FGF23 have been implicated in a number of diseases, including tumorinduced osteomalacia (TIO), X-linked hypophosphatemic rickets (XLHR) (MIM#307800), autosomal dominant hypophosphatemic rickets (ADHR) (MIM#193100), and autosomal recessive hypophosphatemic rickets (ARHR1) (MIM #241520) and (ARHR2) (MIM #613312). These disorders are characterized by low serum phosphorus, normal serum calcium, normal PTH, low calcitriol, and rickets or osteomalacia. TIO is caused by tumoral production of FGF23, which is often mesenchymal in origin. XLH results from an inactivating mutation in PHEX, a proteincleaving enzyme gene. A mutation in the PHEX gene indirectly alters the degradation and production of FGF23, causing increased circulating concentrations. ADHR results from activating mutations in a mutant FGF23 gene that is resistant to protease cleavage. Finally, ARHR is caused by an inactivating mutation in the DMP1 gene. Patients who have ARHR present with symptoms similar to XLH and ADHR. FGF23 values are either elevated or inappropriately normal for the low phosphate values. McCune Albright Syndrome (MIM #174800) is caused by an activating mutation of the a subunit of the trimeric GTP-binding protein that stimulates adenyl cyclase (GNAS1). Fibrous dysplasia is a common manifestation of McCune Albright syndrome, and local production of FGF23 from those lesions may cause hypophosphatemia in some patients. Question: 5 Abbreviations: AVP= arginine vasopressin, MRI=magnetic resonance imaging A 12-year-old boy is hospitalized for behavioral complications associated with his diagnosis of bipolar disorder. His medications include lithium carbonate, risperidone, and aripiprazole. You are asked to see him because his psychiatrist has concerns that he has developed diabetes insipidus. The boy’s mother reports that he has been drinking a lot of fluids at home. The family recently moved from Scotland, and the change in environment has contributed to his behavioral deterioration. On physical examination, the well-hydrated but noncooperative boy has a weight of 40 kg and height of 150 cm (both close to 50th percentile), heart rate of 90 beats/min, respiratory rate of 18 breaths/min, and blood pressure of 110/74 mm Hg. He has an easily palpable thyroid gland, normal heart and lung sounds on auscultation, and some palpable stools on deep abdominal palpation. He has Tanner stage 2 pubic hair growth and 5-mL testes bilaterally. He has 2 areas of vitiligo in his neck area, each patch about the size of a quarter. You order laboratory tests and request monitoring of his intake and output. The following laboratory test results were obtained at 5 pm, after your initial consultation. Serum glucose, 157 mg/dL (8.71 mmol/L) Serum osmolality, 275 mOsm/kg (275 mmol/kg) Serum sodium, 137 mEq/L (137 mmol/L) Urine sodium, 40 mEq/L (40 mmol/L) Urine osmolality, 127 mOsm/kg (127 mmol/kg) Urine specific gravity, 1.002, glucose-negative, trace ketones The next morning, his records reveal: Weight and height, unchanged from admission Heart rate, 90 beats/min Blood pressure, 108/76 mm Hg Fluid intake: 4,400 mL (water, soda, juice) Fluid output: 2,000 mL Of the following, the MOST likely diagnosis for this boy is A. central diabetes insipidus (autosomal dominant) B. central diabetes insipidus (autosomal recessive) C. nephrogenic diabetes insipidus (medication-induced) D. nephrogenic diabetes insipidus (X-linked) E. primary polydipsia Correct Answer: E Although primary polydipsia is a diagnosis of exclusion, a number of factors in the history of the boy described in the vignette suggest that he most likely has primary or psychogenic polydipsia (also called compulsive water drinking). Either central or nephrogenic diabetes insipidus (DI) is less likely, given his low serum osmolality and serum sodium values and high urine sodium value on random testing. DI can be diagnosed if laboratory testing reveals a serum osmolality greater than 300 mOsm/kg (300 mmol/kg), with a simultaneous urine osmolality less than 300 mOsm/kg (300 mmol/kg). On the other hand, a serum osmolality less than 270 mOsm/kg (270 mmol/kg) or a urine osmolality greater than 600 mosm/kg (600 mmol/kg) excludes the diagnosis of DI. The patient who has primary polydipsia may have relatively low serum sodium values at baseline. In contrast, those who have DI show a tendency toward high-normal sodium values or frank hypernatremia. In cases of potential DI, the history is very important. For example, the clinician should always inquire about coexisting psychiatric disease, whether the drinking interferes with normal activities, if certain medications are being taken, and if nocturia and enuresis are present. The boy in the vignette has documented polydipsia. However, the normal urine output recorded for him is most likely due to incomplete collection or inadequate recording, a common occurrence when trying to evaluate this parameter as part of a search for a water balance problem. If the initial screening tests indicate abnormally high fluid intake and urine volume, and the serum sodium osmolality is between 270 and 300 mOsm/kg (270 and 300 mmol/kg), a water deprivation test may be required to confirm or rule out DI, and the response to the administration of aqueous vasopressin can help to differentiate between central and nephrogenic DI. When interpreting the water deprivation test, the clinician should be aware that the patient who has a long-standing history of primary polydipsia may have developed mild nephrogenic DI due to washing out of the renal medullary interstitial concentration gradient. If psychogenic polydipsia is strongly considered as the explanation for the clinical presentation, a few days of supervised moderate fluid restriction may be helpful to facilitate interpretation of the water deprivation test results. There are many protocols for water deprivation testing and nuances to consider when testing younger versus older patients (eg, duration of the test, monitoring procedures). Primary polydipsia is characterized by excessive fluid intake lasting over a prolonged period and not initially due to abnormal AVP secretion or action. Primary polydipsia can be further divided into 3 subcategories: 1) Patients may demonstrate a nonthirst-driven pattern of excessive intake of water/fluids in the setting of a concurrent psychiatric disorder. This type of primary polydipsia, known as psychogenic polydipsia, has been reported in 6% to 17% of hospitalized psychiatric patients. Psychogenic polydipsia can be a manifestation of a patient’s inherent compulsive behavior (as part of the behavioral comorbidity) or can be a method for patients to decrease stress levels. 2) When the excessive water intake is actually driven by an altered thirst mechanism, it is called dipsogenic polydipsia. The cause of the polydipsia is usually idiopathic; no specific cause can be found in most patients. However, occasionally the excessive intake can be associated with pre-existing disease of the hypothalamus. 3) A third group of patients who have primary polydipsia exhibit excessive water intake described as habitual polydipsia that is not infrequently iatrogenic (eg, due to drug intake that results in dry oral mucosae or even be due to inappropriate “medical advice” to increase fluid intake for the sake of improving health “the natural way.”) These patients have no other signs of mental illness or increased thirst. The 3 subgroups of patients all demonstrate excessive consumption of fluids, resulting in excessive body fluid, a decrease in serum osmolality, and consequent suppression of AVP secretion. Water excretion is increased to compensate for the high fluid intake. The serum osmolality stabilizes at a new set point close to the osmotic threshold for AVP secretion. In addition, the increased vascular fluid volume suppresses aldosterone secretion, leading to increased urine sodium excretion. Because these patients share historical and laboratory characteristics with those who have DI (polydipsia, polyuria, dilute urine) and dipsogenic polydipsia can occasionally be caused by the same hypothalamic lesion as found in acquired central DI, it is important to at least consider brain imaging (MRI) before entertaining an idiopathic or psychiatric cause. Question: 6 Your local school board is concerned about the obesity epidemic among American children and charges the school nurse with monitoring weights of the students in the third-grade class. She weighs the 19 students and obtains the following values: 21 29 31 42 25 29 31 43 27 29 35 44 28 29 40 45 28 30 41 Of the following, the MOST appropriate description of student weights is Mean Median Mode 33 30 29 B. 29 30 33 C. 30 29 33 D. 30 33 29 E. 33 29 30 A. Correct Answer: A Summary statistics are used to describe a collection of continuous data such as body weight. Such descriptions require 2 features: a measure of the central tendency and a measure of the dispersion. The central tendency refers to the value around which the observed data tend to cluster. For example, in Figure 1A, the data represented by the black and red distributions each center on their respective C values, but the value of the red C differs from that of the black C. The dispersion or spread refers to the variability of observed values along the continuum. Because the black and red distributions have the same shape, they have the same degree of variability. In contrast, the black and red distributions of data in Figure 1B center on the same value, but the red distribution is wider, indicating that fewer of the observations cluster around the value of C than in the black distribution. Three measures of central tendency depict the “typical” value of a continuous variable for a given population: mean, median, and mode. The mean is the arithmetic average, calculated by summing all the observed values and dividing by the total number of observations. The median, or 50th percentile, is the value above or below which half of the observations fall (ie, the literal “middle” value). The median is calculated by sorting the observed values in ascending or descending order and finding the value that occurs half-way down the list. The mode refers to the most frequently observed value within the dataset. The mode is ascertained by counting the frequency of each observed value and choosing the value that occurs most often. Thus, for the set of student weights described in the vignette, the calculations for these measures are: 1) Mean = 627/19 = 33 2) Median = 30 3) Mode = 29 Selecting which measure of central tendency best describes a dataset depends on the distribution of observations in the dataset. For symmetric distributions around a single peak, the mean, median and mode are identical (Figure 2A). For such normal (or approximately normal) distributions, the mean is preferable because it allows more powerful statistical tests of significance. However, means are unduly influenced by outlier observations. Because outlier observations differ greatly in value from the central tendency but occur at lower frequency, they pull the mean away from the bulk of the observations. Thus, medians are better for describing skewed distributions (Figure 2B). In the vignette, the 6 obese students raise the mean, with the median more closely describing the weights of the majority of the class. Modes, which are least amenable to statistical testing, are useful when a distribution has more than 1 peak (Figure 2C). Such bimodal distributions, such as when a disease tends to occur more frequently at 2 age ranges or in 2 months of the year, may help provide clues as to its pathophysiology. Standard deviation (SD) describes the dispersion of data within a given distribution from its mean (M). Standard deviation is the square root of the variance (V). Variance is calculated by taking the difference between each observed value and the mean (X-M), squaring the differences (so the positive and negative deviations do not cancel out each other), summing those squares (S[X-M]2), then dividing the result by the total number of observations minus 1 (n-1). Thus, SD=square root of (S[X-M]2)/(n-1). Standard error of the mean (SEM) is sometimes confused with SD, but it is not a descriptive statistic. SEM refers to the precision of a mean calculated for a dataset as representing the true value of the mean of the larger population from which the dataset was drawn. SD describes the variability of observed values within the given dataset. Therefore, SD is the appropriate statistical measure to use for reporting a single dataset. Although SEM is sometimes reported because it is smaller, it is not appropriate for describing a single dataset; rather, it should be restricted to evaluating estimates of a population mean. Question: 7 Abbreviations: ACTH=corticotrophin, CRH=corticotropin-releasing hormone, DHEA-S=dehydroepiandrosterone sulfate, HPA=hypothalamic-pituitary-adrenal, 17-OH progesterone=17-hydroxyprogesterone A 16-year-old girl presents with fatigue, anxiety, acne, irregular menses, hypertension, and hirsutism. Prior laboratory studies found elevated cortisol and DHEA-S values, with normal thyroid function test results. Her mother has similar complaints. Physical examination of the adolescent shows her height and weight at the 10th percentile, normal heart rate, blood pressure of 150/95 mm Hg, hirsutism, and acne but no other clinical findings of Cushing syndrome. Results of laboratory evaluations include: - Serum cortisol (8 am), 40 mg/dL (1,103.52 nmol/L) - Serum cortisol (4 pm), 12 mg/dL (331.06 nmol/L) - Plasma ACTH (8 am), 70 pg/mL - Plasma ACTH (4 pm), 45 pg/mL - Testosterone (8 am), 75 ng/dL (2.6 nmol/L) - DHEA-S (8 am), 450 mg/dL (12.15 mmol/L) - 17-OH progesterone (8 am), 270 ng/dL - Deoxycorticosterone (8 am), 160 ng/dL (4,880 nmol/L) - Androstenedione (8 am), 240 ng/dL (8.38 nmol/L) - Aldosterone, 5 ng/dL (138.7 pmol/L) - Sodium, 142 mEq/L (142 mmol/L) - Potassium, 3.2 mEq/L (3.2 mmol/L) - Chloride, 108 mEq/L (108 mmol/L) - Carbon dioxide, 30 mEq/L (30 mmol/L) - Calcium, 9.2 mg/dL (2.3 mmol/L) Electrocardiography documents regular rate and rhythm, with evidence of mildly flattened T waves and ST depression. The girl returns for repeat blood pressure evaluations, which read 155/95 mm Hg and 160/85 mm Hg, and her electrolyte values remain abnormal despite initiation of oral potassium supplements. Two 24-hour urine cortisol excretion tests on consecutive days show values of 180 and 120 mg/dL (4,965 and 3,310 nmol/L). A lowdose dexamethasone suppression test (1 mg orally) produces an 8 am serum cortisol value of 25 mg/dL (689 nmol/L) and plasma ACTH value of 30 pg/mL. A high-dose dexamethasone suppression test (2 mg dexamethasone orally every 6 hours for 8 doses with elevated plasma values of dexamethasone confirming absorption) document urinary free cortisol excretion of 50 mg/day, serum cortisol of 5 mg/dL (137 nmol/L), and ACTH of 5 pg/mL 6 hours after the last dose as well as a blood pressure of 120/75 mm Hg. Of the following, the MOST likely diagnosis for this patient is A. Bartter syndrome B. CYP11B1 deficiency C. pituitary adenoma D. polycystic ovarian syndrome E. primary glucocorticoid resistance Correct Answer: E The hypertensive hirsute adolescent described in the vignette presents with low aldosterone concentrations, metabolic alkalosis, and elevated serum cortisol values. Although CYP11B1 deficiency and Bartter syndrome could explain the abnormal electrolyte findings, the marked elevations of evening cortisol and 24-hour urine cortisol secretion are not consistent with these diagnoses or polycystic ovarian syndrome. A pituitary adenoma with mild or early Cushing syndrome could cause oversecretion of ACTH and adrenal steroids that fails to suppress with lowdose dexamethasone suppression, but minimal cortisol suppression with high-dose dexamethasone testing would be unlikely. Of note, this patient’s blood pressure decreased following dexamethasone administration. The excessive cortisol production and resistance to high-dose dexamethasone suppression testing could be due to ectopic ACTH production, but the absence of Cushing syndrome stigmata suggests the possibility of the rare inherited condition of familial glucocorticoid resistance with compensatory hyperactivity of the HPA axis. This lack of cortisol negative feedback leads to overproduction of CRH, arginine-vasopressin, and ACTH as well as adrenal glucocorticoids, mineralocorticoids, and androgens. Familial glucocorticoid resistance can be inherited as an autosomal recessive or dominant disorder attributed to mutations in the glucocorticoid receptor gene. Affected patients present with a broad range of symptoms related to their overactive HPA axis. Heterozygotes often have mild glucocorticoid resistance. Therapy for this heterogeneous condition aims to suppress the excess ACTH using mineralocorticoid-sparing potent synthetic glucocorticoids such as dexamethasone. Unlike peptide hormone receptors, which span the plasma membrane and bind ligand outside the cell, steroid hormone receptors are found in the cytosol and the nucleus (Figure). Adrenal steroids are lipophilic molecules that pass through the plasma membrane to enter the cell and bind with steroid receptors. These receptors have a hydrophobic ligand binding domain that is occupied by heat shock proteins when inactive. Steroid-specific receptor binding liberates the heat shock protein, thereby exposing a domain called the “hinge region,” which opens and extends the complex to enable passive translocation of the complex to the nucleus, where it binds DNA to stimulate transcription of messenger RNA and protein synthesis. Extensive characterization of the glucocorticoid receptor gene mutations in patients who have clinical and biochemical glucocorticoid resistance include findings of defective steroid receptor ligand binding, nuclear translocation, coactivator interactions, and/or transcription activity. Figure. Adrenal steroids are lipophilic molecules that passively enter the cell to bind to receptors (1), which are found in the cytosol and the nucleus. The steroid-receptor complex translocates to the nucleus (2), where it binds to a DNA regulatory site (3) to stimulate transcription of messenger RNA (4) and protein synthesis (5). Question: 8 Abbreviations: c-AMP=cyclic adenosine monophosphate, 1,25 (OH)2 vitamin D=1,25 dihydroxyvitamin D, PTH=parathyroid hormone A 2-year-old boy presents with lethargy, poor appetite, weight loss, and polyuria. He has not been as active as previously. He drinks 1 glass of milk a day and takes a chewable children’s multivitamin. He takes no other medications or supplements. Physical examination reveals a palpable abdominal mass. Laboratory evaluation documents a serum calcium of 14 mg/dL (3.5 mmol/L). Of the following, the MOST likely cause of the boy’s hypercalcemia is A. calcitriol B. cholecalciferol C. immobilization D. parathyroid hormone E. PTH-related peptide Correct Answer: E The abdominal mass described for the boy in the vignette likely is a solid tumor. Of the cases of hypercalcemia of malignancy associated with solid tumors, PTH-related peptide (PTHrP) secreted by the tumor is responsible in 80%. PTHrP causes bone resorption and subsequent hypercalcemia. Although PTHrP is the most common humoral cause of hypercalcemia of malignancy, numerous cytokines and other hormones have also been implicated, including interleukin (IL)-1, IL-6, TGF-α, tumor necrosis factor-α (TNF-α), macrophage inflammatory protein-1α (MIP-1α), calcitriol (1,25-(OH)2 vitamin D), and rarely, ectopic PTH. In addition, transforming growth factor-ß (TGF-ß) production from bone metastases upregulates PTHrP production. All of these humoral factors lead to calcium release from the bone. PTHrP and PTH also increase renal tubular calcium resorption. PTH secretion from a malignancy is extremely rare, so a parathyroid adenoma needs to be ruled out when the PTH value is elevated. Calcitriol also increases calcium absorption from the gut. Calcitriol is produced from macrophages in Hodgkin or non-Hodgkin lymphoma as well as sarcoid or granulomatous diseases. PTHrP is less frequently the cause of hypercalcemia in hematologic malignancies. In addition to secretion of humoral factors, malignancies can increase serum calcium by direct invasion and destruction of bone by the primary tumor or metastases (osteolytic hypercalcemia) as well as via acute immobilization, which causes increased bone resorption. This boy is less active but not immobilized. His only supplement is a chewable multivitamin, making vitamin D3 (cholecalciferol) toxicity unlikely. Hypercalcemia of malignancy is rare in children (<1% of cases) and occurs in association with leukemia, Hodgkin and non-Hodgkin lymphoma, rhabdoid tumors, rhabdomyosarcoma, hepatoblastoma, neuroblastoma, and Ewing sarcoma. PTHrP is related to, and has homology with, PTH. It is encoded by a single gene on the short arm of chromosome 12, and multiple isoforms are created by alternative splicing. Longer forms are secreted by malignancies and from keratinocytes as well as from mammillary epithelial cells during lactation. Mid-region PTHrP stimulates placental calcium transport and modulates renal bicarbonate handling. Carboxy-terminal fragments may inhibit osteoblast function and stimulate osteoblast proliferation. Both PTHRrP and PTH act through the PTH receptor-1 (PTHR1), a member of the 7-transmembrane-spanning family of G-protein-coupled receptors. The receptor couples to Gαs and signals via the c-AMP/protein kinase A pathway. It also couples to Gαq11 and signals via generation of inositol phosphates, diacylglycerols, and intracellular calcium. PTH binds more tightly to the PTHR1 and is more potent than PTHrP in increasing circulating calcium and 1,25-(OH)2 vitamin D concentrations. Serum concentrations of PTHrP are generally low, except when secreted by malignancies. PTHrP typically is synthesized locally and widely expressed during development. It is secreted by immature chondrocytes in response to Indian hedgehog, coordinates the rate of chondrocyte differentiation to maintain orderly growth of the long bones during development, and is required for formation of the mammary glands. The placenta produces PTHrP under regulation by the calcium-sensing receptor (CaSR), which promotes calcium transport from the mother to the fetus and is required to maintain normal fetal calcium concentrations. After embryogenesis, PTHrP is highly expressed by breast epithelial cells during lactation and secreted into the circulation to increase bone resorption and provide calcium in breast milk production. It also increases proliferation of chondrocytes, delays ossification, relaxes smooth muscle, has positive chronotropic and inotropic effects on cardiac muscle, may regulate proliferation of keratinocytes, stimulates resorption of overlying bone enabling tooth eruption, and is found in all cells in the pancreatic islets. April Question: 1 Abbreviations: CAH=congenital adrenal hyperplasia, DSD=disorder of sexual development, FSH=folliclestimulating hormone, LH=luteinizing hormone An 18-year-old military recruit presents to your clinic for evaluation after his army entrance screening physical examination. His history includes bilaterally undescended testes during infancy. The slender adolescent has a sparse beard and inconspicuous laryngeal prominence. He has normal-appearing male external genitalia with an 8cm stretched penile length and Tanner stage 4 pubic hair. A left 2-mL testis is palpable in the scrotum and a small soft, moveable approximately 2-mL mass is palpable in the right inguinal region. His older brothers and older sister are healthy. Laboratory results show testosterone of 117 ng/dL (4.06 nmol/L), FSH of 77 mIU/mL (77 IU/L), LH of 41 mIU/L (41 IU/L), and prolactin of 16 ng/mL. A karyotype is performed and is 46,XX. His estradiol measures 22 pg/mL (80.76 pmol/L) and progesterone measures 0.34 ng/mL (1.08 nmol/L). Ultrasonography does not show a uterus or ovaries. The right inguinal mass is calcified. You decide to pursue additional testing. Of the following, the MOST likely genetic finding is A. DAX-1 duplication B. SF-1 gene mutation C. SOX-9 deletion D. SRY translocation E. STaR protein deficiency Correct Answer: D Persons who have DSD have discrepancies between their external genitalia, gonadal, and/or chromosomal sex. The boy described in the vignette has a 46,XX testicular DSD with discordant sex chromosomes and genitalia. Evidence of functioning testicular tissue may be the result of a translocation of the SRY (sex-determining region Y) gene (MIM #480000). Individuals who have DAX-1 duplications (MIM #300018) have 46,XY complete gonadal dysgenesis and a female phenotype. Persons who are XX with SF-1 mutations (MIM #184757) may have ovarian insufficiency or early ovarian failure but do not have an intersex disorder. Although SRY box-related gene 9 (SOX9) duplications (MIM #608160) have been reported to cause 46,XX testicular DSDs with phenotypes similar to that of the boy in the vignette, deletions do not. Steroidogenic acute regulatory (STaR) protein deficiency (MIM #600617) is a cause of lipoid CAH, associated with undervirilization, not a 46,XX testicular DSD. 46,XX testicular DSDs are rare, with an incidence estimated at 1 in 20,000 to 25,000. Ninety percent of persons who have a 46,XX testicular DSD have a Y-to-X translocation involving the SRY gene found at Yp11.3. SRY is located adjacent to a chromosomal segment with a high degree of homology between the X and Y chromosomes, known as the pseudoautosomal region. Therefore, if unequal crossing over occurs during male meiosis, the SRY gene may be translocated to the distal short arm of the X chromosome. Diagnosis of an SRY gene translocation in a person who has a 46,XX testicular DSD is generally made by fluorescence in situ hybridization (FISH). SRY was first described in investigations of persons who had 46,XX testicular DSD with very small Y-to-X translocations. The SRY gene protein product was proven to be a testes-determining factor that was not only necessary but sufficient for testis development when transfection of a 14-kb genomic fragment containing SRY into 46,XX mouse embryos resulted in transgenic 46,XX mice with testes and male sex differentiation. SRY is an intronless gene found only in mammals that encodes a DNA-binding protein belonging to a family of high-mobility group (HMG) proteins. SRY gene transcripts are expressed briefly in cells from the coelomic epithelium that become Sertoli cells and direct mesonephric cell migration. The highly conserved SRY protein HMG box domain binds to DNA in a sequence-specific manner (A/TAACAAT), resulting in DNA bending. The chromatin change affects downstream target gene transcription. The SOX-9 gene is the most likely target of SRY. In mice, SRY is known to upregulate SOX-9 and foster its continued expression in Sertoli cells. When SRY is absent, SOX-9 is suppressed and ovaries develop. Deletions (sometimes due to translocations) or point mutations of the SRY gene can result in 46,XY complete gonadal dysgenesis (Swyer syndrome) and account for about 15% to 20% of such cases. Most such mutations are found in the DNA-binding region (the HMG box) of the protein, although mutations that affect DNA bending and SRY protein nuclear localization have also been implicated. Question: 2 Abbreviations: ALT=alanine aminotransferase, BMI=body mass index, FFA=free fatty acid, HbA1c=glycolated hemoglobin, HDL=high-density lipoprotein, LDL=low-density lipoprotein, TG=triglyceride, T2D=type 2 diabetes A 16-year-old African American boy is referred to your clinic by his primary care provider after measuring a fasting glucose of 105 mg/dL (5.83 mmol/L). The boy reports that his mother and grandmother both have diabetes. He does not have polyuria or polydipsia. His BMI is 36, and he has evidence of acanthosis nigricans. You repeat a fasting glucose test, which measures 103 mg/dL (5.72 mmol/L). His fasting lipid panel reveals a total cholesterol of 185 mg/dL (4.79 mmol/L), HDL cholesterol of 29 mg/dL (0.75 mmol/L), LDL cholesterol of 116 mg/dL (3.00 mmol/L), and TG of 190 mg/dL (2.15 mmol/L). His HbA1c is 6.1%. His ALT measures 52 IU/L. Of the following, the metabolic alteration that is MOST responsible for the pathogenesis of this boy’s presentation is A. decreased interleukin-1 B. decreased leptin C. decreased tumor necrosis factor-α D. increased adiponectin E. increased FFAs Correct Answer: E Insulin resistance (IR) is the fundamental metabolic alteration implicated in the development of several chronic medical problems, including T2D, fatty liver disease, hypertension, and cardiovascular disease. IR refers to the inhibition of normal physiologic pathways stimulated by insulin, including cellular disposal of glucose, lipogenesis, and glycogenolysis. Obesity, particularly visceral adiposity, is the primary risk factor for development of IR. Compared to the nonobese population, obese individuals have elevated markers of inflammation, atherogenic profiles of adipokines, and increased FFA values. Peripheral and hepatic IR stem from these adipocyte-driven derangements and their effect on hepatic and skeletal muscle glucose and lipid metabolism. The boy described in the vignette is typical of a commonly encountered referral to the pediatric endocrinologist’s practice. Obesity rates have risen dramatically over the past 25 years worldwide. Mirroring the rise in obesity has been an increase in metabolic derangements, including T2D, nonalcoholic fatty liver disease, and dyslipidemia, previously rarely observed in the pediatric population. This adolescent has an obese BMI and acanthosis nigricans, an external finding suggesting IR. His laboratory findings are consistent with impaired fasting glucose and dyslipidemia (decreased HDL cholesterol, elevated TG values), common to individuals residing within the disease spectrum spanning IR to frank T2D. He would be considered high risk for progression to T2D, based on his ethnic background, family history, BMI, signs of IR, and laboratory results in the prediabetes spectrum. The development of IR and progression to T2D involve a complex interplay of genetic and environmental components. However, one of the primary metabolic derangements that contributes to this pathogenesis is increased amounts of adipocyte-derived FFAs. Increased numbers and size of adipocytes, particularly visceral adipocytes, result in increased circulating FFAs. FFAs impede normal insulin actions of lipolysis inhibition, glycogenolysis, gluconeogenesis, and stimulation of glucose uptake in peripheral tissues. Chronically elevated FFAs can lead to T2D by interfering with normal insulin signaling, thereby contributing to hepatic and peripheral insulin resistance, and by directly inhibiting insulin secretion from the ß-cell. The mechanisms that underlie the FFA-driven pathologic changes are not completely understood but likely involve: Increased intracellular TG content that leads to decreased phosphorylation of insulin receptor substrate-1 (IRS) and normal signaling along the PI-3 kinases pathway, resulting in impaired glucose uptake Increasing endoplasmic reticulum stress and generation of reactive oxygen species Activation of proinflammatory pathways, further interfering with insulin signaling at the level of the receptor and IRS Binding to toll-like receptor 4 on adipocyte-located macrophages, generating increased nuclear factor-kB, which can enter the nucleus and turn on genes that promote an inflammatory response Inhibition of the insulin-stimulated production of endothelial nitric oxide, a potent vasodilator In experimental models, lowering of FFA concentrations reverses these changes associated with IR. Interleukin-1 and tumor necrosis factor-α are both proinflammatory cytokines that are increased in visceral adipose tissue and play a role in the development of IR. Leptin concentrations are increased in obese individuals, although its actions in improving endothelial function and insulin sensitivity are reduced due to a relative state of leptin resistance. Lastly, adiponectin, an adipocyte-derived hormone with important roles in improving insulin sensitivity and decreasing glucose and TG concentrations, is decreased in obese individuals, especially those who have IR. Individuals who have IR must have an exaggerated, compensatory ß-cell response to ensure that glucose is effectively driven into peripheral tissues to maintain a euglycemic state. When this compensatory ß-cell response is impaired, peripheral and hepatic uptake of glucose is impaired, blood glucose concentrations remain elevated, and symptoms of diabetes mellitus ensue. Lipotoxicity and glucotoxicity are terms used to describe the deleterious, toxic effects of excess lipid and/or glucose on progressive ß-cell failure. Pathologically, these metabolic derangements have an almost synergistic role in their deleterious effects on the ß-cell. This has led to the term glucolipotoxicity to describe the relationship between chronic hyperglycemia leading to alteration in intracellular lipid partitioning away from oxidation and toward lipid synthesis and hyperlipidemia. Within the ß-cell, increased intracellular glucose and FFA metabolism results in elevated malonyl-CoA that, in turn, inhibits carnitine-palmitoyl transferase-1 and subsequently activates the esterification pathway. Shifting to this pathway results in accumulation of lipid-derived signaling molecules that adversely affect ß-cell function. Lipotoxicity and glucotoxicity also contribute to endoplasmic reticulum stress, another proposed mechanism for inducing ß-cell apoptosis. It is, therefore, probable that glucotoxicity and lipotoxicity both contribute to the progressive deterioration in ß-cell function that occurs in T2D. Question: 3 Abbreviations: ELISA=enzyme-linked immunosorbent assay, HIV=human immunodeficiency virus, OI=osteogenesis imperfecta You are caring for the sixth child of consanguineous parents who has severe OI. The infant has 4 healthy siblings and an older brother who was diagnosed with severe OI in infancy. Testing for Col1A1 and Col1A2 mutations yields negative results. You suspect that the baby might have a recessive form of OI. You send a blood specimen to a research laboratory where scientists find a deletion in exon 4 of the cartilage-associated protein (CRTAP) gene. The scientists ask you for a skin biopsy to assay the baby’s skin fibroblasts. Of the following, the technique that is MOST likely to determine if this mutation alters the amount of CRTAP protein produced is A. fluorescence in situ hybridization B. Northern blot C. polymerase chain reaction D. restriction fragment length polymorphism analysis E. Western blot Correct Answer: E A variety of laboratory techniques are used to identify chromosomal abnormalities and characterize their effects. Of the options given, only Western blots detect and quantitate specific proteins in a tissue or blood sample. The other techniques are useful for detecting heritable abnormalities but do not help with quantifying protein amounts. Northern blots examine RNA. Fluorescence in situ hybridization (FISH), polymerase chain reaction (PCR), and restriction fragment length polymorphism (RFLP) analyses are all studies of DNA. FISH requires matching of a labeled DNA probe to the patient’s genomic DNA to look for the presence or absence of particular genomic DNA sequences. PCR is a rapid in vitro method of amplifying a particular sequence of DNA. After the DNA is amplified, mutation searches can be conducted using a variety of techniques, including DNA sequencing. RFLPs are differences in the length of fragments of DNA, which are detected by enzymatically cleaving DNA and using Southern blotting to separate the DNA fragments. Western blots are used to identify proteins, quantify them, and determine their molecular weight. The blot is so named due to its evolution from Southern blots (named after their inventor Edward Southern), which are used to separate DNA fragments and detect specific sequences. Northern blots detect RNAs, quantify them, and determine their size. To perform a Western blot, protein is obtained either from whole tissue or tissue culture (Fig 1A). Whole tissue is broken down using either homogenization or sonication methods. Cells are lysed using detergents, salts, and/or buffers that may contain proteases and/or phosphatase inhibitors to prevent protein degradation (Fig 1B). The concentration of protein in the sample is determined using a spectrophotometer. The proteins in the extract are separated by size, usually by using a gel containing polyacrylamide and the anionic detergent SDS (Fig 1C). SDS denatures the proteins and gives them negative charges. When voltage is applied to the gel in either 1 or 2 directions (electrophoresis), proteins of different sizes and charges migrate at different rates, separating into bands. The gel should contain a lane with a molecular weight marker to allow determination of the molecular weight of the targeted protein. The gel should also have a lane with a positive internal control of known protein concentration and molecular weight, which will be used later to determine if the primary antibody is effective. Once electrophoresis has been performed, separated proteins are transferred from the gel to a membrane (the blot), usually made of nitrocellulose (Fig 1D). The transfer is made via an electric current that pulls the proteins into the paper. The blot is “blocked” from nonspecific binding using a dilute protein solution. After blocking, the blot is incubated with a primary antibody to the protein of interest (Fig 2A). It is then washed, reblocked, and incubated with a secondary reporter antibody. This antibody can have a colorimetric, radioactive, or fluorescent tag (Fig 2B). A readout can be obtained for relative quantitation of the protein of interest by comparing the reporter signal intensity (Fig 2C) with that of a known control or different time point. Often this is obtained using a densitometric analysis. Sometimes relative levels of protein expression are determined by comparing the ratio of the intensity of the queried band to that of a reference band (such as ß-actin or glyceraldehyde-3-phosphate dehydrogenase). In addition to providing semiquantitative protein analyses, Western blots also can be used to determine whether a screening immunometric test result is true-positive or false-positive. For example, persons who have positive initial screen results by ELISA for HIV infection should have a Western blot performed. In these cases, stock HIV proteins are separated on a gel, blotted onto a filter paper membrane, and the serum from the patient is added. If antibodies are present in the patient’s serum, they bind to the viral proteins and can be seen after a labeled antihuman immunoglobulin G antibody is added. Question: 4 A neurosurgical colleague asks for your help treating a 12-year-old boy who is in the pediatric intensive care unit after sustaining a closed-head injury 5 days ago while riding an all-terrain vehicle. Initially after his injury, his serum sodium concentrations were normal, but over the last 2 days, they have been decreasing, and now his sodium measures 128 mEq/L (128 mmol/L) despite receiving lactated Ringer solution at maintenance IV rates. On physical examination, the boy’s heart rate is 79 beats/min, blood pressure is 117/78 mm Hg, and central venous pressure is 3 mm Hg. Of the following, the hormone that is MOST likely causing the persistent hyponatremia is A. aldosterone B. angiotensin I C. angiotensin II D. antidiuretic hormone E. atrial natriuretic peptide Correct Answer: E Hyponatremia is a well-known complication of cerebral injury from infection, trauma, hemorrhage, or stroke. In this context, hyponatremia has a variety of causes, including volume expansion due to inappropriate ADH secretion. Although excess ADH secretion leading to intravascular volume expansion is the most common cause of hyponatremia in this setting, the central venous pressure described for the boy in the vignette is low, indicating intravascular volume depletion. While excess ADH secretion may occur in cases of severe volume depletion (from vomiting, diarrhea, bleeding, or sweat losses), due to loss of the inhibitory effect of hyponatremia on ADH release, this child had no initial volume loss. Hyponatremia in the context of brain injury can also be associated with cerebral salt wasting due to excessive production of atrial natriuretic protein (ANP). For the child in the vignette, the low circulating sodium value, the timing of the salt wasting (occurring >3 days after initial injury), and the low central venous pressure should raise suspicion of cerebral salt wasting, which can be associated with high circulating concentrations of ANP. Aldosterone works to conserve sodium (and secrete potassium) and, therefore, would not be implicated as a cause of hyponatremia. Angiotensin II works to stimulate aldosterone secretion. Angiotensin I has no biological activity until cleaved by angiotensin-converting enzyme and exists solely as an angiotensin II precursor. Of note, hyponatremia in a clinically ill child should also raise concerns about adrenal insufficiency that may result in decreased free water clearance. The natriuretic peptide family consists of structurally related peptides derived from prohormones from different genes (Figure). ANP is secreted from granules found in muscle cells in the atria (and to a lesser extent in the ventricles). ANP is derived from a large precursor peptide, but when released from the heart, it circulates as a 28-amino acid peptide, with a characteristic 17-amino acid ring formed by a cysteine disulfide bond. In the kidney, the ANP prehormone is alternatively processed, leading to a 32-amino acid peptide, urodilatin, that acts in a paracrine manner to regulate sodium transport. Brain (b-type) natriuretic peptide (BNP) was named based upon its original isolation from porcine brain. BNP is released primarily from the heart ventricles. It circulates as a 32-amino acid protein, also with a 17-amino acid ring. A third natriuretic protein, C-type natriuretic peptide (so named because it was discovered third) circulates at very low concentrations and is believed to act primarily in an autocrine/paracrine manner in the brain, pituitary, kidneys, and vascular endothelium. Recently, a fourth natriuretic protein, dendroaspis natriuretic peptide (DNP), has been discovered in the venom of the Green Mamba snake. The primary stimulus for ANP and BNP secretion is increased cardiac wall pressure. However, a variety of other factors, including T4, prostaglandins, glucocorticoids, angiotensin II, and cytokines, have been shown to increase ANP and BNP synthesis in cell culture. Studies have shown that ANP is released in any hypervolemic state, including heart failure, aldosterone excess, renal failure, and ingestion of salt. As in the vignette, ANP concentrations have been reported to be elevated in certain cases of cerebral injury, including adults who have cerebral hemorrhages. The cause of this elevation remains unclear because cases of cerebral salt wasting are characterized by low atrial pressure, and although ANP-containing neurons have been found in the brain, low concentrations of this hormone in brain tissue make it unlikely that brain-derived ANP is responsible for the renal salt wasting. Evidence to date suggests that the CNS can modulate cardiac ANP release. Once released, both ANP and BNP act through a common G-protein-coupled receptor, guanylyl cyclase-A. As circulating endocrine hormones, ANP and BNP act to decrease blood pressure and enhance natriuresis. The primary effect of ANP appears to be on vascular dilation; urodilatin and BNP appear to be more important natriuretic hormones. In the vasculature, both ANP and BNP increase capillary permeability, leading to fluid extravasation and decrease in blood pressure, and relax smooth muscle in arterioles and venules. ANP and BNP also inhibit the pressor effects of angiotensin II and catecholamines. In the kidneys, they increase glomerular filtration rate and decrease renal tubular sodium reabsorption. They inhibit renin secretion and reduce aldosterone secretion. ANP and BNP may also play key roles in regulation of fatty acid metabolism and body weight, with beneficial effects in fat cells on lipolysis, adiponectin secretion, and adipocyte size and beneficial effects in muscle on PPARg, mitochondria, and muscle fat oxidation. In addition to their endocrine effects, both ANP and BNP act in a paracrine manner to decrease cardiac fibrosis and hypertrophy. Targeting this peptide system has been attempted in the treatment of congestive heart failure. Recombinant versions of ANP (carperitide, available only in Japan) and BNP (nesiritide) have been marketed for use in persons who have CHF due to their natriuretic effects. A second approach is to inhibit the breakdown of endogenous natriuretic peptides by limiting their physiologic degradation by ectodermal neutral endopeptidase 24.11 (NEP), and NEP inhibitors were once being actively developed as therapies for hypertension and heart failure. DNP may be less susceptible to enzymatic degradation and useful pharmacologically. Measurements of these natriuretic peptides and inactive amino-terminal fragments of their prohormones have been used to enhance diagnosis and risk stratification for persons who have acute and chronic heart failure. Question: 5 Abbreviations: ALT=alanine transferase, BMI=body mass index, HbA1c=glycolated hemoglobin, T 4=thyroxine, T2D=type 2 diabetes A 16-year-old girl presents to the emergency department with a severe headache that awoke her from sleep this morning. Her mother describes the girl’s sleep as restless, with loud snoring. The girl reports nausea, vomiting, and recent blurred vision associated with the headache. She is dizzy and complains of some neck and shoulder pain and a new finding of double vision when looking to the right but not to the left. She has been previously healthy, with an evaluation for possible diabetes 2 years ago. At that time, she had normal findings on an oral glucose tolerance test, HbA1c, assessment of thyroid function, lipid profile, and electrolytes evaluation. She has irregular menses and her last period was 6 weeks ago. She denies use of tobacco, prescribed medications, or other drugs. On physical examination, the girl, who is uncomfortable due to her severe headache and nausea, is at more than the 97th percentile for weight and the 25th percentile for height and her BMI is greater than the 99th percentile. Her heart and respiratory rates are normal, and her blood pressure measures 130/75 mm Hg. She has photophobia, with limited funduscopic examination showing papilledema, no goiter, and normal cardiovascular and lung findings. Her abdomen is obese, with mild right upper quadrant tenderness. She has no hyperreflexia, nerve palsies, significant striae, or dysmorphic findings. Immediate laboratory testing shows a normal complete blood count with differential count, normal chemistry panel, slight elevation of ALT, and normal free T 4 value. Of the following, the test result that would be MOST helpful in determining the cause of this girl’s symptoms is A. elevated thyroid peroxidase antibodies B. increased opening pressure on lumbar puncture C. normal QRS complex on electrocardiography D. polysomnography apnea/hypopnea index of 4 E. TSH of 6 mIU/L Correct The girl described in the vignette has symptoms consistent with pseudotumor cerebri or idiopathic intracranial hypertension associated with obesity. Of note, pseudotumor cerebri can be attributed to an extensive list of other causes, including abnormal thyroid function. Neither a mild increase in TSH to 6 mIU/L nor the common finding of elevated thyroid peroxidase antibodies in the presence of normal free T4 concentrations is diagnostic of hypothyroidism. Obesity-related headaches can also be associated with hypertension or sleep apnea, although a normal QRS complex suggests the absence of left ventricular hypertrophy, and a polysomnography apnea/hypopnea index of 4 reflects a normal sleep study result. Pseudotumor cerebri is a diagnosis of exclusion. Obesity-related pseudotumor cerebri is more likely to occur in postpubertal obese adolescents who have symptoms of elevated intracranial pressure (ICP) and papilledema with no other identifiable cause of increased ICP or underlying neurologic disease. Initial symptoms may include headaches that are typically worse in the early morning or with Valsalva maneuver, visual symptoms, and facial nerve palsies. Clinical examination findings of papilledema often are associated with neuroimaging reports of flattened posterior sclera or optic nerve distention, but the cerebral ventricles are normal or small. Obesity places children at risk for numerous conditions. For example, obesity is a risk factor for metabolic insulin resistance syndrome, including hypertension, dyslipidemia, fatty liver disease, dysglycemia, sleep disturbance, and gonadal dysfunction. Hypertension is defined as blood pressure at or above the 95th percentile for sex, age, and height on 3 or more occasions, and is likely underrecognized. Insulin resistance and obesity are risk factors for T2D. The SEARCH for Diabetes in Youth Study estimates that T2D accounts for 15% of all diabetes cases among non-Latino white adolescents ages 10 years or older in the United States, and it is a more predominant cause for T2D among high-risk racial/ethnic groups. Pulmonary complications of obesity include an increased risk for childhood asthma. Obstructive sleep apnea may occur in up to one third of severely overweight youth, and sleep-disordered breathing is even more common. A polysomnography apnea/hypopnea index or respiratory distress index above 10 suggests an elevated rate of abnormal breathing events; such data can be used to quantify sleep apnea symptoms. Sleep apnea is associated with excessive daytime fatigue, decreased alertness, and elevated daytime blood pressure. Increased left ventricular hypertrophy can develop if sleep apnea is not treated. Early screening for dyslipidemia is recommended for all obese children because elevated lipid concentrations may be associated with nonalcoholic fatty liver disease and pancreatitis. Obesity is also a risk factor for cholelithiasis, although cholesterol gallbladder stones develop independently of serum lipid concentrations. Other obesity-related complications include progressive tibia vara or Blount disease with an abnormal gait. Obesity increases the risk of a slipped capital femoral epiphysis, which typically occurs near the age of skeletal maturity. Question: 6 Abbreviations: HbA1c=glycolated hemoglobin, TG=triglyceride, T1D=type 1 diabetes, T2D=type 2 diabetes You are asked to present an overview of current knowledge regarding the most effective medications used to treat pediatric hypertriglyceridemia in children who have T2D. You review the literature and find 2 recent meta-analyses that were calculated using different assumptions. The first study included outcomes of hyperlipidemia therapy reported for all prospective studies in the United States over the past 5 years regardless of size or type of diabetes (T1D and T2D) in children ages 10 through 16 years. The second study included cross-sectional, crossover, and prospective studies of therapy specific to hypertriglyceridemia in children younger than 18 years of age who had only T2D. You appreciate that the process of undertaking a systematic review and a meta-analysis involves making many decisions. Of the following, the statistical analysis that would be MOST helpful in understanding and determining the underlying differences leading to different conclusions between the 2 meta-analyses is A. adjusted odds ratio B. ANOVA testing C. chi-square testing D. Fisher’s exact t-test E. sensitivity analysis Correct Answer: E The 2 different meta-analysis data sets described in the vignette were included for meta-analysis with a priori inclusion criteria to assess treatment outcomes for hyperlipidemia or hypertriglyceridemia in children who have diabetes. Differences between the hyperlipidemia definition criteria, age of the patients, and their types of diabetes as well as study design criteria could affect the conclusions of each meta-analysis. However, both reviews are relevant to the clinical question. A sensitivity analysis would allow determination of how the different entry criteria and eligible studies affected the results. Sensitivity analysis is a statistical approach used to compare the results of 2 or more meta-analyses that differ in their assumptions for inclusion. A sensitivity analysis addresses the question of whether the results would change if there were a change in the entry criteria of the meta-analyses being compared and reviewed. This approach might require exclusion of studies with doubtful eligibility, poor quality results, small study size, or significant missing data. Additionally, an original incomplete search of the literature may have biased the findings of 1 of the meta-analyses. Sensitivity analysis includes recalculating the meta-analysis results using only selected subgroups of the data or inclusion of only cohort or prospective study trials to minimize result bias. In this case, elimination of the T1D studies might be considered to determine whether this alters the outcomes of the first meta-analysis. The strengths of sensitivity analysis relate to the ability to compare complex study results that may have included entry criteria considered problematic for their criteria choice or methodology. Such analysis often allows identification of the most significant studies to the outcome of the meta-analysis. This approach provides a statistical method to resolve complex differences between meta-analysis outcomes. The Cochrane reviews are a valuable resource for published sensitivity analysis results. Sensitivity analysis is also often used to make costeffectiveness decisions. The analysis frequently is presented as a chart ranking the factors identified from the most important to the least important in the model and can be used for evidence-based medicine decisions related to a specific outcome. A sensitivity analysis is only as good as the data placed into it. Disadvantages of sensitivity analysis include the potential for bias as studies are reselected for analysis. Assessing the interrelationships between each of the variables selected may not be realistic, and results may be misleading. Sensitivity analysis methodology typically requires familiarity with appropriate software and computational assistance to graph and present the outcomes appropriately to address the significance of findings expressed as a calculated correlation coefficient. Adjusted odds ratio, ANOVA testing, chi-square testing, and Fisher’s exact t-test are used to assess individual study data. They are not appropriate approaches for comparing 2 meta-analyses. The adjusted odds ratio statistic is used to compare data regarding outcomes or exposure of interest while taking into account the impact of other risk factors included in the multivariable model. An example of an adjusted odds ratio study is a comparison of the improvement in hypertriglyceridemia between 2 randomized groups of children who have T2D and received 2 different therapies for elevated TG concentrations. An analysis of variance (ANOVA) test provides a statistical method of determining which effects are significant in a normally distributed data set for randomized treatment groups. For example, ANOVA testing might find that the HbA1c rather than the hypertriglyceridemia therapy explains the major effect of lowering TG values in a randomized study of children who have T2D. Similarly, the chi-square test and the Fisher’s exact t-test are among the most widely used statistical approaches for 2 data sets to determine whether an observed difference in the proportions is unlikely to be explained by chance alone and, thus, is a statistically significant result. Question: 7 Abbreviations: DXA=dual-energy x-ray absorptiometry, 25-OH vitamin D=25 hydroxyvitamin D, OI=osteogenesis imperfecta, PTH=parathyroid hormone An 11-year-old boy presents with the acute onset of severe lower back pain after doing a somersault. He has had bilateral heel pain for the past 6 months that he says is exacerbated by exercise. He had reactive airway disease diagnosed at 8 years. He takes albuterol via nebulizer 3 times daily as well as budesonide oral inhalation 2 puffs twice daily during the winter. Since receiving the diagnosis, he has been prescribed 3 1-week courses of oral prednisone (20 mg daily x 7 days). The last course was 4 months ago. One year ago, he tore his left hamstring muscle and was on crutches for 5 weeks, followed by physical therapy for 2 weeks. He has no additional past medical or surgical history of note. There is no family history of recurrent fracture. Physical examination reveals a healthy-appearing boy with white sclerae, percussion tenderness over his lower thoracic vertebrae, and tenderness over his heels bilaterally without swelling or redness. He has no joint hyperextensibility or bruising. Radiography of the spine documents a compression fracture of his 11th thoracic vertebra. Complete blood count, electrolytes, liver function tests, calcium, phosphate, alkaline phosphatase, 25-OH vitamin D, and PTH results are within normal parameters. DXA scan of his lumbar vertebrae shows a bone mineral density z-score of -4.1. Of the following, the MOST likely diagnosis is A. idiopathic juvenile osteoporosis B. juvenile idiopathic arthritis C. osteogenesis imperfecta D. osteoporosis due to glucocorticoid therapy E. osteoporosis due to reduced mobility Correct Answer: A The boy described in the vignette most likely has idiopathic juvenile osteoporosis (IJO), a rare bone condition that affects children and adolescents. It is believed to be due to impaired bone remodeling that results in decreased amounts of cancellous bone. Affected patients generally present with vertebral compression fractures and metaphyseal fractures of long bones that lead to bone pain and impaired mobility. Some cases of IJO appear to be caused by mutations in the low-density lipoprotein receptor-related protein 5 (LRP5) gene. Spontaneous resolution has been reported in some individuals; others have progressed to severe disability. Before considering the diagnosis of IJO, clinicians should exclude secondary causes of osteoporosis that generally fall into the categories of reduced mobility, inflammatory diseases, systemic glucocorticoids, and disordered puberty and low body weight. This boy’s 5-week history of using crutches would not be expected to have any significant effect on his bone density. Reduced mobility that has been associated with osteoporosis includes conditions such as cerebral palsy, severe head injury, muscular dystrophy, and spinal muscular atrophy. Increased inflammatory cytokines can impair bone turnover and result in osteoporosis in children who have inflammatory conditions such as juvenile idiopathic arthritis (JIA) and Crohn disease. However, this boy’s isolated heel pain as well as lack of swelling and redness is more likely due to Sever disease (apophysitis of the calcaneus) than JIA. Another very important cause of secondary osteoporosis is glucocorticoid excess. Glucocorticoids reduce intestinal calcium absorption and impair bone metabolism, resulting in decreased bone formation and increased bone resorption. However, the winter use of inhaled budesonide and few short 1-week courses of oral prednisone would not be expected to cause significant change in his bone density. OI is an important cause of primary osteoporosis that is due to defective synthesis of type I collagen (either amount or quality). The estimated incidence of OI is 1 in 10,000 to 20,000 births. There are several types of OI, but the most common form, type 1, presents with recurrent fractures in adolescents, blue sclera, joint hypermobility, easy bruising, and brittle teeth. The absence of any of these features makes OI unlikely for this boy. When considering treatment of osteoporosis, it is important to exclude or address any conditions that may be causing or exacerbating the condition. Initial focus should be on optimizing nutrition and reducing inflammation. Physical therapy, using crutches, avoiding unsafe weightbearing activities, and other supportive care should be initiated. Weightbearing activity should be encouraged, if possible. For patients who have limited mobility, physical therapy and/or use of vibrating platforms may be helpful. If these measures fail to improve or prevent further bone loss and fracture, medical therapy should be considered. Although none of the drugs used to treat osteoporosis in adults have been approved by the United States Food and Drug Administration for use in pediatric patients, bisphosphonates are considered a reasonable choice for children who have moderate-to-severe OI (>2 fractures in 1 year or vertebral compression fractures). For osteoporosis due to other causes, bisphosphonates may be used on a compassionate basis to treat low-trauma fractures of the spine or extremities. Optimal intake of calcium and adequate vitamin D stores are important to optimize bone mineral accretion in all children. The current recommended daily intake of calcium is: 700 mg for children ages 1 to 3 years 1,000 mg for children ages 4 to 8 years 1,300 mg for children and adolescents ages 9 to 18 years of age The Table lists the calcium content of many calcium-rich food sources. Question: 8 Abbreviations: cAMP=cyclic adenosine monophosphate, 1,25 (OH)2 vitamin D=1,25 dihydroxyvitamin D, PTH=parathyroid hormone You are consulted by the neurology team about a 3-year-old child who has intractable epilepsy, pancreatitis, and hypocalcemia. He was diagnosed with epilepsy 1 year ago. He is currently taking valproate, but seizures have not fully responded to medication. He began a traditional ketogenic diet 3 months ago, resulting in a dramatic reduction in seizure frequency. He was otherwise healthy until 3 days ago, when he developed severe abdominal pain and stopped eating. At the emergency department yesterday, results of his physical examination and serum laboratory tests confirmed a diagnosis of pancreatitis. He was admitted to the hospital for monitoring and further laboratory testing. The neurology team is asking you about a critically low serum ionized calcium value. Of the following, the MOST likely other expected serum laboratory findings for this patient are A . B . Magnesium Potassium PTH 1-25 (OH)2 Vitamin D High High High High Low Low High Normal C Low High High Low Low Low Low Low Normal High Normal High . D . E . Correct Answer: D The child described in the vignette has hypocalcemia resulting from hypomagnesemia. Hypomagnesemia, hypokalemia, low PTH, and low calcitriol would be expected because of use of the ketogenic diet and the occurrence of acute pancreatitis. Hypermagnesemia may cause hypocalcemia but is not expected with pancreatitis or use of the ketogenic diet. The ketogenic diet, whose use dates back to the 1920s, is effective treatment for many forms of intractable epilepsy. It consists of a high-fat, normal-protein, and low-carbohydrate diet, with the traditional ketogenic diet comprising 4 parts fat to 1 part protein and carbohydrate (ie, 4:1 lipid-to-nonlipid ratio). Complications from the diet include hypomagnesemia and pancreatitis. Isolated hypomagnesemia is usually mild and likely results from decreased gastrointestinal absorption. Pancreatitis is more common in patients also taking valproate and may lead to severe hypomagnesemia. Magnesium is the second most abundant intracellular cation. It is predominantly stored in bone, with only 1% to 2% of total body stores in the extracellular space. Magnesium is absorbed through intestinal channels in a vitamin D-independent process and is reabsorbed in the kidneys in the thick ascending loop of Henle and in the proximal tubules. Normal serum values range from 1.5 to 2 mEq/L (1.5 to 2 mmol/L), although serum measurements may not adequately reflect total body stores. Hypomagnesemia may result from decreased dietary magnesium intake, increased gastrointestinal or renal losses, or increased shift from the extracellular space into bone or intracellular space. Affected patients are not usually symptomatic until serum magnesium concentrations fall below 1.0 mEq/L (1.0 mmol/L). Hypomagnesemia is often associated with hypokalemia and hypocalcemia. The hypokalemia results from impaired renal tubular reabsorption of potassium and may be severe. Hypocalcemia may also be severe and likely results from the suppressive effects of low magnesium on PTH secretion, PTH action, and calcitriol synthesis. Sustained correction of potassium and calcium is not possible until the magnesium concentrations have been normalized. Impaired PTH secretion is primarily responsible for the hypocalcemia seen with magnesium deficiency. When serum magnesium concentrations are normal or high, magnesium affects PTH secretion in a similar manner to calcium. It binds to the extracellular calcium-sensing receptor (CASR), leading to suppression of PTH release through the generation of inositol phosphates and the inhibition of cAMP. Paradoxically, sustained or severe hypomagnesemia also leads to suppressed PTH secretion through augmented CASR signaling within the parathyroid cell. This appears to be mediated through increased activity of Gsa subunits of heterotrimeric Gproteins. Hypomagnesemia may also lead to reduced PTH response in the kidneys and bone, impaired calcitriol synthesis, and increased calcitriol resistance. These effects are not primarily responsible for the hypocalcemia but may exacerbate the severity of the condition. May Question: 1 Abbreviations: FGF=fibroblast growth factor, PTH=parathyroid hormone, 1,25 (OH)2 vitamin D=1,25 dihydroxyvitamin D, 25-OH vitamin D=25 hydroxyvitamin D You are seeing a toddler in clinic. Her father is a second-year medical student who has X-linked hypophosphatemic rickets. His daughter’s linear growth has recently slowed, and she is manifesting mild frontal bossing and some metaphyseal widening and tibial bowing. Her father is curious about the endocrine abnormalities that are present in his daughter. Of the following, the hormone that is MOST likely to have an inappropriately low value in the girl is A. calcitonin B. FGF-23 C. 1,25 (OH)2 vitamin D D. PTH-related protein E. 25-OH vitamin D Correct Answer: C X-linked hypophosphatemic rickets (XLH) is the most common inherited disorder of renal phosphate wasting. It is caused by inactivating mutations in the PHEX gene that are inherited in an X-linked dominant pattern. Decreased PHEX expression leads to increased circulating FGF-23 concentrations. FGF-23 acts in conjunction with a coreceptor Klotho at the proximal renal tubule to decrease proximal tubule renal phosphate reabsorption. FGF-23 also suppresses production of 1,25 (OH)2 vitamin D, leading to an inappropriately low serum value for that hormone. Standard therapy for XLH includes frequent dosing of phosphate and pharmacologic doses of 1,25 (OH) 2 vitamin D. 1,25 (OH)2 vitamin D is used as therapy, in part, because it helps to increase blood calcium concentrations by promoting absorption of dietary calcium, increasing renal calcium reabsorption, and stimulating calcium release from bone. Although the other hormones listed all are factors that affect mineral homeostasis, none is inappropriately low in persons who have XLH. 25-OH vitamin D is the primary “storage” form of vitamin D and would likely be normal in the girl described in the vignette. Calcitonin is primarily manufactured by thyroid parafollicular cells (C-cells). It acts to decrease calcium concentrations and may play a role in regulating serum magnesium, but its concentrations are not altered in XLH, and it does not a play a large role in any aspect of calcium homeostasis in humans; neither excess nor deficiency is associated with calcium abnormalities. PTH-related protein (PTHrP) has close homology with PTH and is produced as a paracrine/autocrine factor by a wide variety of fetal and adult tissues. Its most important functions are to regulate endochondral bone formation and breast development/lactation. PTHrP concentrations are usually undetectable (<2 pmol/L) and are not changed by XLH. Vitamin D is obtained from dietary sources or synthesized in the skin after ultraviolet radiation of 7dehydrocholesterol (Figure). Fat-soluble, dietary plant-derived vitamin D2 (ergocalciferol) and animal-derived vitamin D3 (cholecalciferol) are absorbed in the duodenum and jejunum. Once absorbed, vitamin D is transported into the circulation bound to D-binding protein. In the liver, vitamin D is converted by vitamin D-25-hydroxylase to 25-OH vitamin D. 25-OH vitamin D concentrations in the serum reflect vitamin D nutritional stores. Although 25OH vitamin D can bind to the vitamin D receptor and circulates in concentrations 1,000 times greater than those of 1,25 (OH)2 vitamin D, 25-OH vitamin D is primarily regarded as a prohormone to the active 1,25 (OH)2 vitamin D because it has much lower affinity for the vitamin D receptor than 1,25 (OH)2 vitamin D. 25-OH vitamin D undergoes 1α-hydroxylation in the kidney by the enzyme 1α-hydroxylase to 1,25 (OH)2 vitamin D, a circulating hormone and the primary active metabolite. Some 1,25 (OH)2 vitamin D is also produced in extrarenal sites, where it stimulates the vitamin D receptor in an intracrine or paracrine fashion. 1,25 (OH)2 vitamin D production is highly regulated, primarily by circulating PTH (stimulatory) and FGF-23 (inhibitory). Dietary phosphate restriction can increase serum 1,25 (OH)2 vitamin D concentrations by decreasing FGF-23, thus increasing renal 1α-hydroxylase expression and activity. PTH is a potent stimulator of 1α-hydroxylase gene expression. In conditions characterized by high PTH concentrations (such as vitamin D deficiency), 1,25 (OH)2 vitamin D concentrations can be high, even in the face of very low or even clinically undetectable amounts of the substrate 25-OH vitamin D. This is at least partly because decreased phosphate concentrations lead to decreased circulating FGF-23, relaxing FGF-23 inhibition of renal 1α-hydroxylase. In conditions of high FGF-23 such as XLH, 1,25 (OH)2 vitamin D is generally inappropriately suppressed. 1,25 (OH)2 vitamin D binds to the vitamin D receptor (VDR), a member of the steroid receptor superfamily. Once 1,25 (OH)2 vitamin D binds, the VDR associates with the retinoid X receptor (RXR). This heterodimerization is required for migration of the 1,25 (OH)2 vitamin D-VDR-RXR complex from the cytoplasm to the nucleus. In the nucleus, the complex regulates gene transcription by interacting with specific vitamin D response elements in vitamin D-responsive genes. 1,25 (OH)2 vitamin D regulates the production of multiple proteins involved in increasing intestinal and renal calcium transport, including calbindins, transient receptor potential cation channel subfamily V members (TRPVs), plasma membrane calcium pumps, and sodium-calcium exchangers. 1,25 (OH)2 vitamin D also stimulates bone resorption by increasing osteoclast numbers and the active transport of phosphate across the intestinal mucosa. Within target tissues, 24-hydroxylase (CYP24) attenuates 1,25 (OH)2 vitamin D signaling by hydroxylating 1,25 (OH)2 vitamin D to the short-lived metabolite 1,24,25(OH)3 vitamin D. Question: 2 You are evaluating a 16-year-old girl for primary amenorrhea. She is otherwise healthy and has no significant past medical history. Her weight and height are each at the 50th percentile, and she has Tanner stage 5 breast and pubic hair development. Genitourinary examination reveals a small vaginal pouch without clitoromegaly or labial fusion. You plan to pursue a more thorough evaluation of this patient, including a family history, laboratory studies, and radiologic assessment. Of the following, the MOST likely finding of these studies is A. absence of fallopian tubes B. history of maternal in utero exposure to diethylstilbestrol (DES) C. history of maternal müllerian aplasia D. missense mutation in the androgen receptor gene E. urologic anomaly Correct Answer: E The girl described in this vignette has clinical findings consistent with müllerian aplasia or vaginal agenesis. Müllerian aplasia results from incomplete müllerian duct migration, fusion, or canalization. This results in underdevelopment of the uterus, cervix, and vagina. The karyotype is 46,XX, and genetic transmission is believed to be multifactorial. Müllerian aplasia is usually associated with the Mayer-Rokitansky-Küster-Hauser syndrome (MRKH), which is characterized by congenital partial or total absence of the vagina and uterus. Seventy-five percent of patients have no vaginal opening, with the remaining 25% having a small vaginal pouch. In approximately 10% of patients, functional uterine tissue is present and may lead to cyclic or chronic abdominopelvic pain. Ovaries and fallopian tubes are usually present, and the karyotype is normal. MRKH is believed to be a sporadic occurrence and not a heritable condition. Girls who have müllerian aplasia often have extragenital abnormalities. Approximately 25% to 50% of patients have urologic anomalies, including unilateral renal agenesis, pelvic or horseshoe kidneys, or abnormalities of the collecting system. Skeletal anomalies have been found in approximately 10% of patients, with absence of vertebrae and spinal fusion described. Androgen insensitivity syndrome (AIS) results from mild, partial, or complete unresponsiveness of the androgen receptor due to a missense mutation of the androgen receptor gene. The karyotype in AIS is 46,XY, and the gonads are testicles that are often undescended. Complete AIS should be suspected in a phenotypic female who has normal breast development, minimal axillary or pubic hair, and primary amenorrhea. In partial AIS, the phenotype depends on the degree of responsiveness of the external genitalia to androgens. The typical phenotype is a male patient who has micropenis, hypospadias, and a bifid scrotum that may contain gonads. In the more severe presentation, the phenotype may be female, with clitoromegaly, posterior labial fusion, and decreased-tonormal axillary and pubic hair. DES is a nonsteroidal synthetic estrogen that was used to prevent pregnancy loss from 1940 to the 1970s. It was administered to at least 2 million women in the United States before it was discontinued due to its lack of efficacy and concerns about its safety. The adverse effects of in utero DES exposure in women included vaginal clear cell adenocarcinoma, preterm delivery, pregnancy loss, and infertility due to structural abnormalities of the reproductive tract. Mice studies have shown that structural anomalies result from DES-induced disruption of müllerian epithelium differentiation. In utero DES exposure in men was linked to genitourinary anomalies and possibly an increased risk of infertility and testicular cancer. Studies in mice have also shown that adverse effects of DES on the reproductive tract involve persistent changes in gene expression, which has prompted concern about human offspring of women exposed in utero to DES. The United States National Cancer Institute has tracked these third-generation offspring through the 1990s and into the 21st century. The most frequently reported birth defects in third-generation sons and daughters are cardiac and skeletal anomalies. Sons also have an increased risk of penile and testicular anomalies; daughters did not have an increased risk of genitourinary problems. There appears to be no increased risk of cancer in males or females. Question: 3 Abbreviations DKA=diabetic ketoacidosis, GH=growth hormone, IGF-1=insulinlike growth factor-1, IV=intravenous, T1D=type 1 diabetes, T2D=type 2 diabetes A 14-year-old girl who had T1D diagnosed 3 days ago is complaining of generalized swelling. Over the past 3 weeks, she had marked polyuria and polydipsia and a 15-lb weight loss. She presented to the emergency department with vomiting and dehydration and was found to be in DKA. She was admitted to the intensive care unit for IV insulin therapy and IV fluid hydration and was transferred to your service yesterday. You subsequently initiated a basal-bolus injection regimen. On physical examination, the very thin girl has periorbital edema and generalized facial swelling as well as pitting edema in her lower extremities. Chest radiography confirms the presence of bilateral pleural effusions. Of the following, the MOST likely cause for her clinical findings is A. genetic mutation also resulting in hepatic dysfunction B. increased renal excretion of albumin C. inhibition of endothelial nitric oxide synthase D. stimulation of natriuresis in the distal renal tubule E. stimulation of renal Na+/K+-ATPase activity Correct Answer: E The girl described in this vignette has recently diagnosed T1D and generalized edema after starting insulin therapy that is most likely caused by stimulation of Na+/K+-ATPase activity in the renal tubules. Edema that develops after starting insulin therapy in a severely insulin deficient patient is an underrecognized and underreported, but usually benign complication of such therapy. It typically presents in a newly diagnosed patient after an episode of DKA, although those who have established diabetes and poor glycemic control can have a similar reaction after improving consistency with insulin administration. Intensive fluid administration in a catabolic state can lead to leaking of fluids into subcutaneous tissues, an effect that is exacerbated by the increased vascular permeability seen with chronic hyperglycemia. Renal excretion of albumin is usually not increased, although serum albumin concentrations may be slightly reduced through transcapillary leakage. Insulin therapy may lead to generalized edema through its vasodilatory actions, its stimulatory effects on renal sodium absorption, and its inhibitory effects on glucagon action in the renal distal tubule. Insulin promotes vasodilation by increasing endothelial nitric oxide synthase production. Insulin promotes renal sodium reabsorption by stimulating the Na+/K+-ATPase and the Na+/H+ exchanger in the renal proximal tubules. Glucagon usually stimulates natriuresis in the distal tubule. Insulin suppresses glucagon secretion that, in turn, leads to reduced natriuresis and may exacerbate the swelling. There is a rare mitochondrial mutation (3243 mitochondrial tRNA) associated with peripheral edema and hepatic dysfunction. Most diabetic patients who have this mutation do not have T1D, and edema is localized to the lower extremities. Insulin therapy may also cause localized skin reactions, including lipohypertrophy, lipoatrophy, and insulin allergy. Lipohypertrophy is the most common reaction and consists of raised subcutaneous fat deposits at the site of injections. This usually results from improper rotation of injection sites and resolves with avoidance of these sites. Lipoatrophy and insulin allergies are much less common after the transition was made from animal insulin to recombinant human insulin. Insulin allergy is rare and usually results from the protamine component in NPH; most allergic reactions after injections are in response to the preservatives and plastics used in insulin delivery. Poor glycemic control in a person who has established diabetes often results in weight loss and possibly delayed puberty and poor linear growth. Any slowing of growth is typically most pronounced during the pubertal years unless the patient also has untreated hypothyroidism or celiac disease. Mechanisms for diminished growth include malnutrition, chronic acidosis, increased glucocorticoid production, end-organ unresponsiveness to GH, and reduced delivery of IGF-1 to tissues. GH binding protein (BP), IGF-1, and IGFBP-3 concentrations are often low in the face of elevated GH values, supporting a role for unresponsiveness to GH. IGFBP-1 concentrations are often high in pubertal children who have poor glycemic control, resulting in sequestering of free IGF-1 and decreased delivery to tissues. This is not seen in prepubertal children. Mauriac syndrome describes children who have poor glycemic control, severe growth failure, and hepatosplenomegaly due to excess hepatic glycogen deposition and steatosis. The mechanism is poorly understood but may reflect hypercortisolemia from recurrent hyperglycemia and ketosis. Fortunately, Mauriac syndrome is very rare with modern diabetes care. You are reviewing a journal article with a junior colleague in which the authors evaluated a newly developed diagnostic test for hypothyroidism. The results include a receiver operating characteristic analysis (Figure 1). Of the following, the MOST correct statement for you to make in discussion with your colleague is that A. cutoff A gives the highest sensitivity and lowest specificity B. cutoff B gives the highest false-positive rate C. cutoff C gives the highest specificity D. sensitivity is plotted on the x-axis E. specificity is plotted on the x-axis Correct Both the clinical sensitivity and specificity of a diagnostic test are determined using established cutoff levels as dividing lines between positive and negative. Receiver operating characteristic (ROC) curve analysis is used to determine the ideal cutoff value that maximizes both the sensitivity and specificity of a particular test as applied to a specific disease and population. In ROC analysis, both the experimental and a “gold standard” test are performed in the patient cohort. Using each result of the experimental test as a potential cutoff, the sensitivity and specificity are calculated, and the results are plotted on a graph with sensitivity (true positives) on the y-axis and the falsepositive rate (1 – specificity) on the x-axis. Figure 2 illustrates a ROC curve analysis of a new test for hypothyroidism versus a “gold standard” measure. Using each result of the new test as a potential threshold between positive and negative, the sensitivity and specificity are calculated and a ROC curve is prepared. The point on the curve closet to the upper left (Point A) generally indicates the optimal compromise between high sensitivity and a low false-positive rate. Point B indicates a nearby cutoff value with maximum sensitivity. While Point B has a relatively low false-positive rate, Point C indicates a cutoff value with the lowest false-positive rate and, thus, the maximum specificity. Inherent in the determination of clinical validity is the assumption that true positives and true negatives can be clearly distinguished. This requires application of a widely accepted reference or gold standard test to separate the 2 groups. Long-term follow-up may also provide a tool to determine if disease is actually present. When evaluating sensitivity or specificity of assays for a particular disease, it is important to consider how the gold standard is applied. Ideally, the reference test should be obtained independently, with the researchers blinded to the results of the experimental test. The same reference test should be used for all study participants as opposed to those with positive tests being subsequently studied with one standard and those with negative results being studied with another standard. If multiple standards are used, they should be equally rigorous to avoid bias in the calculation of sensitivity and specificity. Furthermore, proper evaluation of the clinical sensitivity and specificity of assays should be performed on a patient population whose disease status is not known a priori. In situations where the clinical impact of a false-negative result far outweighs the impact of a false-positive result (such as newborn screening), a point on the curve may be chosen for the assay cutoff that maximizes sensitivity, such as Point B in Figure 2. The reverse might be true for confirmatory tests, in which a false-positive result would unnecessarily commit the patient to a high-risk or long-term course of treatment. The ideal ROC curve is nearly square, with sensitivity close to 1 and false-positive rate (1 – specificity) close to 0. The area under the curve (AUC) represents the accuracy of the test, and a test with an AUC of 1 performs perfectly. A poorly performing test has a flat ROC curve, close to the line of unity, indicating that the test is only marginally better than chance. An AUC of 0.5 represents a worthless test. ROC analysis allows comparison of 2 different diagnostic tests with a gold standard. Each diagnostic test is compared to the gold standard, and the 2 resulting ROC curves are plotted on the same graph. Determination of AUC for each data set demonstrates the better of the 2 tests (Figure 3 ). Question: 5 A pathology resident contacts you to help him understand the results of some practice immunochemistry he has been doing. He has several slide-mounted tissue specimens, including pancreas, liver, gall bladder, and ileum, and has been using an antiglucagon antibody. He reports that he found glucagon immunoreactivity not only in the pancreas but also in the ileum. The other tissues (liver and gallbladder) have no staining. He asks you for your opinion on this finding. Of the following, your BEST response is to tell him A. to repeat the staining because his finding is unexpected B. that antiglucagon antibodies are often nonspecific C. that ectopic pancreatic tissue is commonly found in the ileum D. that the staining is likely of that of glucagon-like peptide E. to find out if the patient from whom the specimen is derived had a history of hyper- or hypoglycemia Correct Answer: D The proglucagon gene (gcg) gene encodes for not only glucagon but also 2 glucagon-like peptide hormones: glucagon-like peptide-1 (GLP-1) and GLP-2 (Figure). Posttranslational processing of proglucagon differs in the pancreas and in the gut. In pancreatic A cells, proglucagon is cleaved to produce glucagon and the major proglucagon fragment, which are secreted together (Figure). The primary action of glucagon is to stimulate hepatic glucose release. In contrast, in the enteroendocrine L-cells, proglucagon is cleaved to produce GLP-1 and -2, which are formed and secreted separately. The intestinal L-cells are found in greatest abundance in the distal ileum and colon, and, therefore, the finding of glucagon immunoreactivity in the ileum is expected. Given that this is an expected finding, the patient from whom this sample came would not be expected to have abnormal glucose regulation. The specificity of the immunochemistry findings is shown by the absence of staining in the liver and gallbladder. Ectopic pancreatic tissue is an unlikely explanation for the glucagon immunoreactivity in the ileum. However, ectopic pancreatic tissue may be found in a number of sites, as a result of small amounts of pancreas tissue being displaced during embryonic development. The common sites of ectopic pancreatic tissue are the stomach, duodenum, and jejunum; it can rarely be seen in a Meckel diverticulum. GLP-1 is released in response to meal intake and is extremely rapidly metabolized and inactivated by the enzyme dipeptidyl peptidase IV (DPP-4). The primary actions of GLP-1 are to: 1. Stimulate insulin secretion (ie, act as an incretin hormone) 2. Inhibit glucagon secretion, thereby contributing to limitation of postprandial glucose excursions. 3. Inhibit gastrointestinal motility and secretion. 4. Play a role in the regulation of appetite and food intake. Because of these properties, GLP-1 agonists are used for the treatment of type 2 diabetes. Question: 6 Abbreviations: GH=growth hormone, GHRH=growth hormone-releasing hormone, IGF-1=insulinlike growth factor-1, IGFBP3=insulinlike growth factor-binding protein-3 You have been asked to evaluate a patient for short stature. On physical examination, you note a striking clinical finding (Figure). Of the following, the test results that are MOST consistent with the cause for this patient’s short stature are A. IGF-1 IGFBP-3 Peak GH to Arginine and ITT * Decreased Decreased <5 ng/mL B. Decreased Decreased >10 ng/mL C. Decreased Normal >10 ng/mL D. Increased Increased <5 ng/mL E. Normal Normal >10 ng/mL Correct Answer: A The boy described in the vignette has a single central incisor, specifically, a single median maxillary central incisor (SMMCI), which is a midfacial defect. Midfacial defects are associated with GH deficiency as well as hypopituitarism. Other midfacial abnormalities include colobomas of the eye, midface hypoplasia, cleft lip and palate, and median cleft face. GH deficiency (GHD) cannot be diagnosed by a single GH sample because values are often low due to sampling between the normal pulses of GH secretion. Screening for GHD should include measurement of IGF-1, the major GH-dependent peptide, because its serum concentration is relatively stable throughout the day. IGF-1 values vary with sex, age, pubertal maturation, nutritional status, and ethnicity. In addition to GHD, IGF-1 concentrations may be reduced in delayed puberty, malnutrition, hypothyroidism, hepatic disease, and diabetes mellitus, and intraindividual variability can range from 3% to 36%. Another screening tool is IGFBP-3, the major serum carrier of IGF peptides. IGFBP-3 is also GH-dependent but is less affected by age and nutrition. Provocative tests for GH release can also be used to diagnose GHD, although they are expensive, uncomfortable, and associated with some risks. GH concentrations are measured over a period of time after administration of agents known to stimulate GH secretion. Tests available in the United States include insulin-induced hypoglycemia (insulin tolerance test), arginine, glucagon, clonidine, and propranolol. The definition of a subnormal response is somewhat arbitrary. Initially, the threshold for a normal response was set at 5 ng/mL. Over time, the threshold has increased, but with increasing cut-offs, more children who have normal GH concentrations are identified as having GHD. The current widely used cut-off is 10 ng/mL. Lower GH values (2- to 3-fold) are obtained in the current monoclonal assays compared to the older polyclonal assays. Due to the heterogeneity of the analyte, the availability of different preparations for calibration, and the interference from matrix components such as GHbinding protein, variability between assay results exceeds 100%. In addition, the reliability of the GH peak measured by comparing the results of 2 tests using different stimuli, or even the same stimulus on a different day, is poor. Peak GH response is blunted in obese patients and is low in adolescents who have delayed puberty and are still prepubertal. Accordingly, some clinicians advocate priming with estrogen or androgen for a few days before GH stimulation testing in these patients. Examining the options provided, a peak GH response of less than 5 ng/mL to 2 provocative stimuli in a nonobese patient is consistent with GHD, as are low IGF-1 and IGFBP-3 values. Low IGF-1 and IGFBP-3 values with high stimulated GH suggest GH resistance. Low IGF-1, normal IGFBP-3, and normal stimulated GH results can be seen in undernutrition. Elevations in all 3 test results are consistent with GH excess. Normal study results are most consistent with a healthy child. SMMCI is found primarily in association with holoprosencephaly (HPE). Sporadically, it can occur in VACTERL (vertebral anomalies, anal atresia, cardiac malformations, tracheoesophageal fistula, renal anomalies, and limb anomalies), CHARGE (coloboma, heart anomaly, choanal atresia, retardation, and genital and ear anomalies), and velocardiofacial syndromes as well as SMMCI syndrome (an association of short stature, choanal atresia/midnasal stenosis, and SMMCI). The most frequent genetic cause for SMMCI is a mutation in sonic hedgehog (SHH), as well as mutations in other genes involved in the hedgehog signaling pathway (SIX3, TGIF, GLI2, and PTCH) and associated with holoprosencephaly. SMMCI also can involve a mutation in SALL4, which has no association with the hedgehog signaling pathway or holoprosencephaly. A few mutations in SHH and one in hedgehog interacting protein (HHIP) have been found in patients who have hypopituitarism. GHD can be congenital or acquired (Table). Other genetic causes of GHD include mutations in the GH-releasing hormone (GHRH) receptor or GH gene. In addition, mutations can occur in other transcription factors and signaling molecules involved in the development of the hypothalamus and pituitary that usually result in combined pituitary hormone deficiencies: pituitary primordium factors such as HESX1, OTX2, RIEG (PITX2), LHX3, LHX4, and SOX3; ventral diencephalon factors such as fibroblast growth factor 8 (FGF8) and GLI2; and pituitary differentiation factors such as Prophet of Pit-1 (PROP1) and POU1F1 (Pit-1). No mutations in GHRH have been found to date. Acquired GHD is due to injuries of the hypothalamus and pituitary. Such injuries can result from trauma, tumors, or surgery involving the hypothalamus and/or pituitary; inflammation/infections; infiltrative diseases of the hypothalamus or pituitary stalk; cranial irradiation; or autoimmune diseases. Treatment with recombinant human GH (rhGH) should be initiated for patients who have GHD as soon as possible after diagnosis. There is a lack of consensus as to rhGH dosage, but typical dosing is 25 to 50 mcg/kg per day. Some advocate doubling the dose during puberty to mimic the adolescent growth spurt. Some clinicians also routinely monitor IGF-1 concentrations during therapy. rhGH is administered subcutaneously in the evening to mimic normal physiology. Daily dosing is more effective than the older 3 times weekly dosing. Question: 7 Abbreviations ADH=antidiuretic hormone, BUN=blood urea nitrogen, CNS=central nervous system, IM=intramuscular You are called to see a 13-year-old boy who was brought to the emergency department with signs of dehydration and lethargy. He has a history of craniopharyngioma resection 2 years ago. He is currently taking levothyroxine, hydrocortisone, growth hormone, and desmopressin acetate (DDAVP). His uncle reports that he forgot to give the boy his DDAVP medicine last night after spending most of the day at the baseball game. He reports that the boy was not very hungry last night and did not eat or drink much. He heard the boy wake up twice at night to go to the bathroom to urinate. The uncle checked in with him both times, but the boy reported he was fine and went back to bed. This morning, when the uncle had difficulty arousing the boy, he administered IM hydrocortisone. On physical examination, the boy has a blood pressure of 102/56 mm Hg and heart rate of 124 beats/min. Initial laboratory evaluation reveals: Serum sodium, 172 mEq/L (172 mmol/L) Potassium, 4.2 mEq/L (4.2 mmol/L) Chloride, 131 mEq/L (131 mmol/L) Carbon dioxide, 19 mEq/L (19 mmol/L) BUN, 34 mg/dL (12.1 mmol/L) Creatinine, 1.3 mg/dL (114.9 mmol/L) Glucose, 104 mg/dL (5.8 mmol/L) Of the following, the MOST likely cause of this patient’s current state of hypernatremia and dehydration is an abnormality or defect in A. angiotensin II conversion B. aquaporin channel insertion into the basolateral membrane C. organum vasculosum lamina terminalis D. supraoptic and paraventricular nuclei E. vasopressin binding to its V2 receptor Correct Answer: C The boy described in the vignette presents with hypernatremic dehydration and has a past history of craniopharyngioma and a medication list suggesting he has panhypopituitarism. His DDAVP was not administered adequately, leaving him at risk for increased urinary free water losses. Although he had access to water to drink, he has not been drinking, despite an increase in urine output. This suggests an adipsic state or primary problem with osmoregulation, an abnormality that can be observed at the time of diagnosis or following surgery for suprasellar/hypothalamic tumors. The organum vasculosum lamina terminalis (OVLT) is the primary center for central osmoregulation. A lesion to the OVLT can result in loss of normal thirst and severe hypernatremia in the setting of concurrent diabetes insipidus (DI) if adequate water is not ingested. For patients who have central DI with intact osmoregulation, even without DDAVP, hypertonicity stimulates sufficient thirst to maintain plasma osmolality close to normal despite increased free water losses. Therefore, a lesion to the supraoptic and paraventricular nuclei, both responsible for vasopressin synthesis, would result in central DI, not loss of thirst. Mutations in the gene for the vasopressin 2 receptor or to the aquaporin 2 channel result in nephrogenic diabetes, which is not consistent with this clinical scenario. Osmoregulation is intact in nephrogenic DI, and affected patients experience thirst and drink water in states of hyperosmolality. Angiotensin II has been proposed as having a stimulatory effect on the sensation of thirst, but a definitive role in this process remains unclear. In this clinical setting, angiotensin II generation would be normal, but it would not have its proposed effect in the OVLT to stimulate thirst. Osmoregulation is a critical adaptive mechanism to maintain tonicity and cell volume within the brain. Although glucose and urea nitrogen exert a small effect, plasma osmolality is primarily dependent on 2 variables: sodium and water intake or excretion (Figure 1). Increases in serum osmolality trigger specific mechanisms to counter any further increases and maintain the osmolality in a set range of 282 to 295 mOsm/kg under most physiologic conditions. This is accomplished through a series of osmosensors that influence the secretion of vasopressin (ADH) and stimulate thirst. When plasma osmolality falls below this range, both vasopressin secretion and thirst are inhibited. Osmoreceptors are housed in neurons within the central and peripheral nervous system. In the periphery, these neurons are found along the proximal gastrointestinal tract and its vasculature (oropharynx, splanchnic vasculature, liver, portal vein). These peripheral osmoreceptors sense alterations in the ingested osmotic load and transmit a signal via the vagus nerve and spinal pathways to the CNS, thereby influencing vasopressin secretion and thirst perception. Receptors in the oropharynx are sensitive to both the volume and coldness of ingested fluid. Central osmoregulation occurs within the OVLT, a circumventricular organ in the anterior ventral region of the third ventricle and, importantly, outside the blood-brain barrier. This area receives input and transmits signals to other parts of the brain that are involved in osmoregulation. Additional sites of osmosensation and regulation likely reside in the subfornical organ. Vasopressin is synthesized within the supraventricular and supraoptic nuclei of the hypothalamus. The activity of these neurons is related primarily to plasma tonicity and, to a lesser extent, plasma volume. At plasma osmolality of approximately 282 to 285 mOsm/kg, vasopressin is released in escalating concentrations as plasma osmolality increases (Figure 2). Its maximum effect on urine concentration, however, peaks at a plasma osmolality of approximately 300 mOsm/kg. The supraventricular and supraoptic neurons receive afferent signals originating from peripheral osmoreceptors and other regions of the brain to affect vasopressin synthesis and transfer to the posterior pituitary gland. Angiotensin II indirectly stimulates vasopressin release through the OVLT and subfornical regions of the hypothalamus. The sensation of thirst is triggered in humans at an osmotic threshold of approximately 295 mOsm/kg. Notably, this is approximately 10 mOsm/kg higher than the threshold for vasopressin release, allowing the kidneys to retain ingested water (Figure 2). The mechanism and neural pathways for the sensation for thirst are still being elucidated. However, it is now established that alteration of tonicity outside the set point stimulates mechanical changes in cell volume of osmosensitive cells within the OVLT that, in turn, changes the rate of depolarization or hyperpolarization of the cell membrane, depending upon whether hypertonicity or hypotonicity, respectively, is present. The sensation of thirst is subsequently transmitted and activated in multiple regions within the cerebral cortex. Data from both animal models and imaging studies from humans demonstrate that the anterior cingulate cortex and insular cortex are the predominant areas. Lesions to this cortical region, therefore, impair the sense of thirst but may not completely eliminate drinking because additional subcortical areas are likely involved in the habitual behavior of water drinking. Disorders of thirst regulation can be broadly separated into polydipsic and adipsic categories. A spectrum of osmoregulatory abnormalities have been described in individuals who have adipsia. Primary hypernatremia or type A adipsia occurs in individuals who have maintained osmoregulatory capacity but at set points much higher than normal. Both thirst and vasopressin release occur at a plasma osmolality that is typically greater than 300 mOsm/kg. Although extremely rare, children who have this disorder often present in infancy or early childhood with chronic dehydration and hypernatremia. Structural abnormalities may be identified in midline CNS structures in the region of the hypothalamus. An underlying cause is not always identified in such patients, but a variant of transient receptor potential vanilloid-responsive 1 channel may be responsible. Tumors, granulomatous disease, and hemorrhage either occurring in the area of the OVLT or iatrogenically following surgery are additional causes for inappropriate adipsia, often with frank central DI if the supraoptic and supraventicular nuclei or posterior pituitary are involved. Polydipsia can be psychogenic, related to compulsive water drinking behavior, or occur in the context of an underlying CNS abnormality. Patients who have compulsive or psychogenic polydipisa may develop some impairment in the renal response to vasopressin due to the dilutional effect of chronic hyponatremia but otherwise maintain normal osmoregulation and vasopressin release. Some studies have suggested a lower osmotic threshold for thirst stimulation and inadequate satiation following water drinking in affected patients. Twenty percent of individuals who have schizophrenia have symptoms of primary polydipsia. Altered osmoregulation resulting in adipisa is observed with intracranial tumors, particularly craniopharyngiomas, but polydipsia can also be seen following removal of these tumors, accompanied by symptoms of polyphagia. Question: 8 Abbreviations: MEN=multiple endocrine neoplasia A pediatrician calls you with the concern that a 4-year-old girl’s flushed cheeks are a symptom of a carcinoid tumor because the child’s father has MEN1 syndrome. Of the following, the MOST likely presentation of carcinoid tumor is A. asymptomatic B. bronchospasm C. flushing D. hypoglycemia E. secretory diarrhea Correct Carcinoid tumors are estimated to occur in 10% of patients who have MEN1. These neuroendocrine tumors arise from enterochromaffin cells located in the bronchi, gastrointestinal tract, pancreas, or thymus. They have neurosecretory granules that can contain vasoactive amines such as 5-hydroxytryptamine (5-HT or serotonin), peptides, tachykinins, and prostaglandins, but fewer than 10% of carcinoids are secretory. Therefore, carcinoid tumors are most commonly silent and patients are generally asymptomatic. Most carcinoid tumors contain the enzyme tryptophan hydroxylase, which converts tryptophan to 5hydroxytryptophan (5-HTP). Most ileal carcinoid tumors also contain aromatic l-amino acid decarboxylase, which converts 5-HTP to 5-HT (serotonin). Serotonin is then released into the circulation. Gastric carcinoids often lack aromatic l-amino acid decarboxylase and, thus, secrete 5-HTP. Serotonin is oxidized to 5hydroxyindoleacetaldehyde, which is rapidly converted to 5-hydroxyindoleacetic acid (5-HIAA) by aldehyde dehydrogenase. 5-HIAA is excreted into the urine. Normally, the products of the carcinoid are metabolized by the liver; when they escape into the circulation, carcinoid syndrome occurs. Patients who have midgut carcinoids (small intestine, appendix, proximal large bowel) develop carcinoid syndrome only with hepatic metastases; foregut carcinoids (lungs, bronchi, stomach) can develop carcinoid syndrome without hepatic metastases because their secretory products enter the systemic circulation directly. Hindgut carcinoids (distal large bowel, rectum) seldom secrete these products. Clinical signs of the carcinoid syndrome include: Cutaneous flushing, typically of the head, neck, and upper trunk, probably mediated by tachykinins Bronchoconstriction Secretory diarrhea due to increased intestinal motility, likely mediated by serotonin Venous telangiectasia Pellagra (dermatitis, diarrhea, and dementia) due to dietary tryptophan shuttled to production of 5-HTP and 5-HT and away from niacin Valvular heart disease (usually right-sided) due to fibrotic plaques Intestinal obstruction from the primary tumor or from a desmoplastic reaction in the surrounding mesentery Hypoglycemia is not a manifestation of carcinoid syndrome but does occur with insulinomas. Carcinoid syndrome is characterized by increased excretion of urinary 5-HIAA. Urinary 5-HIAA values of greater than 25 mg/day are diagnostic of carcinoid syndrome; values of 9 to 25 mg/day may be seen with carcinoid syndrome, nontropical sprue, vomiting, or acute intestinal obstruction. However, ingestion of serotonin-containing foods (avocados, bananas, eggplant, kiwi fruit, nuts [hickory nuts, pecans, walnuts], pineapple, plums, tomato products) and some medications (acetaminophen, antihistamines, antihypertensives, antipsychotics, caffeine, cough suppressants, diazepam, muscle relaxants, nicotine, warfarin) can give false-positive results. Plasma chromogranin A assessment is a sensitive test (values are often elevated in those who have carcinoid tumor, including those who do not have carcinoid syndrome). However, the assessment is not specific because plasma chromogranin A can be elevated in a variety of neuroendocrine tumors as well as in patients who have essential hypertension, heart failure, renal failure, and atrophic gastritis and in those using protein pump inhibitors. Carcinoid tumors are not one of the primary manifestations of MEN1 (Table). Most patients who have MEN1 present in the third to fifth decades, most commonly with hypercalcemia due to hyperparathyroidism from adenomas/hyperplasia in multiple parathyroid glands. Extrapancreatic islet cell tumors are the second most common finding (60% to 80% of patients), with about one half gastrinomas and about one third insulinomas. Approximately 15% to 50% of patients who have MEN1 develop pituitary tumors, with the most common type being a prolactinoma. Patients who have MEN1 have heterozygous germline mutations in the MEN1 gene, which is located on chromosome 11. The MEN1 gene encodes menin, a “tumor suppressor.” In the tumors, there is loss of heterozygosity: the normal MEN1 gene is lost, leaving only the mutant MEN1 gene.