* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download s_block - ilc.edu.hk
Acid–base reaction wikipedia , lookup
Debye–Hückel equation wikipedia , lookup
History of chemistry wikipedia , lookup
Metastable inner-shell molecular state wikipedia , lookup
Chemical bond wikipedia , lookup
Electrolysis of water wikipedia , lookup
Metallic bonding wikipedia , lookup
Electronegativity wikipedia , lookup
Electrochemistry wikipedia , lookup
Hypervalent molecule wikipedia , lookup
Chemical element wikipedia , lookup
Nanofluidic circuitry wikipedia , lookup
Gas chromatography–mass spectrometry wikipedia , lookup
Electron configuration wikipedia , lookup
Rutherford backscattering spectrometry wikipedia , lookup
Abundance of the chemical elements wikipedia , lookup
Photosynthetic reaction centre wikipedia , lookup
Atomic theory wikipedia , lookup
Inorganic chemistry wikipedia , lookup
Coordination complex wikipedia , lookup
Stability constants of complexes wikipedia , lookup
Periodic table wikipedia , lookup
IUPAC nomenclature of inorganic chemistry 2005 wikipedia , lookup
Chemistry: A Volatile History wikipedia , lookup
Alkaline earth metal wikipedia , lookup
Evolution of metal ions in biological systems wikipedia , lookup
Metalloprotein wikipedia , lookup
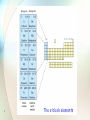
The s-Block Elements 1 The s-Block Elements • Elements of Groups IA* (the alkali metals) and IIA* (the alkaline earth metals) constitute the s-block elements their outermost shell electrons are in the s orbital *Note: In the following, Groups IA and IIA are abbreviated as Groups I and II respectively. 2 3 The s-block elements The s-Block Elements • Similarities 1. highly reactive metals 2. strong reducing agents 3. form ionic compounds with fixed oxidation states of +1 for Group I elements and +2 for Group II elements 4 Q.1 Group I Li Lithium Na Sodium K Potassium Rb Rubidium Cs Caesium *Fr Francium Electronic configuration [He] 2s1 [Ne] 3s1 [Ar] 4s1 [Kr] 5s1 [Xe] 6s1 [Rn] 7s1 Ca Calcium Sr Strontium Ba Barium *Ra Radium [Ar] 4s2 [Kr] 5s2 [Xe] 6s2 [Rn] 7s2 Group II Electronic configuration 5 Be Mg Beryllium Magnesium [He] 2s2 [Ne] 3s2 Group I elements • 6 Lithium Group I elements • 7 Sodium Group I elements • 8 Potassium Group I elements • 9 Rubidium Group I elements • 10 Francium - radioactive Group I elements • 11 Beryllium Group I elements • 12 Magnesium Group I elements • 13 Calcium Group I elements • 14 Strontium Group I elements • 15 Barium Group I elements • 16 Radium - radioactive Characteristic Properties of the s-Block Elements 17 Group I element Electronegativity value Group II element Electronegativity value Li 1.0 Be 1.5 Na 0.9 Mg 1.2 K 0.8 Ca 1.0 Rb 0.8 Sr 1.0 Cs 0.7 Ba 0.9 Fr – Ra – All have low electronegativity. electropositive 18 Group I element Electronegativity value Group II element Electronegativity value Li 1.0 Be 1.5 Na 0.9 Mg 1.2 K 0.8 Ca 1.0 Rb 0.8 Sr 1.0 Cs 0.7 Ba 0.9 Fr – Ra – EN down the group EN : Group II > Group I (∵ greater ENC) 19 Group I m.p.(C) b.p.(C) Group II m.p.(C) b.p.(C) Li 181 1342 Be 1287 2469 Na 98 883 Mg 650 1090 K 63 760 Ca 850 1492 Rb 39 688 Sr 770 1367 Cs 29 690 Ba 714 1637 Fr - - Ra - - Bonding Strength of metallic bond : Group II > Group I m.p./b.p. : Group II > Group I 20 Group I m.p.(C) b.p.(C) Group II m.p.(C) Li 181 1342 Be 1287 2469 Na 98 883 Mg 650 1090 K 63 760 Ca 850 1492 Rb 39 688 Sr 770 1367 Cs 29 690 Ba 714 1637 Fr - - Ra - - Hardness : Group I < Group II Na/K…can be easily cut with a knife 21 b.p.(C) Density Group I Structure Li b.c.c. 0.53 Be h.c.p. 1.86 Na b.c.c. 0.97 Mg h.c.p. 1.74 K b.c.c. 0.86 Ca f.c.c. 1.55 Rb b.c.c. 1.53 Sr f.c.c. 2.54 Cs b.c.c. 1.90 Ba b.c.c. 3.59 Fr - - Ra - - (g cm3) Group II Structure Density (g cm3) Structure Group I : b.c.c. Group II : f.c.c. or h.c.p. except Ba Density : Group II > Group I 22 Density Group I Structure Li b.c.c. 0.53 Be h.c.p. 1.86 Na b.c.c. 0.97 Mg h.c.p. 1.74 K b.c.c. 0.86 Ca f.c.c. 1.55 Rb b.c.c. 1.53 Sr f.c.c. 2.54 Cs b.c.c. 1.90 Ba b.c.c. 3.59 Fr - - Ra - - (g cm3) Group II Structure Density (g cm3) Structure Group I : b.c.c. Group II : f.c.c. or h.c.p. except Ba Density also depends on size and mass of the atoms 23 Group I E o (V) Group II E o (V) Li -3.04 Be -1.69 Na -2.72 Mg -2.37 K -2.92 Ca -2.87 Rb -2.99 Sr -2.89 Cs -3.02 Ba -2.90 Metallic charater (Reactivity) : High tendency to lose electrons as shown by –ve E Mn+(aq) + ne M(s) 24 Group I E o (V) Group II Li -3.04 Be -1.69 Na -2.72 Mg -2.37 K -2.92 Ca -2.87 Rb -2.99 Sr -2.89 Cs -3.02 Ba -2.90 Metallic charater (Reactivity) : - down the groups Group I > Group II 25 E o (V) sodium Sodium is stored under paraffin oil 26 caesium rubidium Caesium and rubidium are stored in vacuum-sealed ampoules 27 Formation of Basic Oxides 1. Group I Elements • 28 All alkali metals form more than one type of oxide on burning in air (except lithium) 1. Group I Elements • Three types of oxides: normal oxides peroxides superoxides Abundant supply O2– oxide ion 29 1 O2 2 O O22– 2O2– 2 peroxide ion superoxide ion 1. Group I Elements Type of oxide formed depends on 1. supply of oxygen 2. reaction temperature 30 3. charge density of M+ 1. Group I Elements • Lithium when it is burnt in air, it forms normal oxide only 2Li2O(s) 4Li(s) + O2(g) 180 C lithium oxide 31 1. Group I Elements • Sodium when it is burnt in an abundant supply of oxygen forms both the normal oxide and the peroxide 2Na2O(s) 4Na(s) + O2(g) 180 C sodium oxide 2Na2O(s) + O2(g) 2Na2O2(s) 300 C excess 32 sodium peroxide 1. Group I Elements • Potassium, rubidium and caesium form All three types of oxides when burnt in sufficient supply of oxygen 33 1. Group I Elements • Potassium: 4K(s) + O2(g) 2K2O(s) potassium oxide 2K2O(s) + O2(g) 2K2O2(s) potassium peroxide K2O2(s) + O2(g) 2KO2(s) potassium superoxide 34 1. Group I Elements • Rubidium: 4Rb(s) + O2(g) 2Rb2O(s) 2Rb2O(s) + O2(g) 2Rb2O2(s) Rb2O2(s) + O2(g) 2RbO2(s) 35 1. Group I Elements • Caesium: 4Cs(s) + O2(g) 2Cs2O(s) 2Cs2O(s) + O2(g) 2Cs2O2(s) Cs2O2(s) + O2(g) 2CsO2(s) 36 Oxides formed by Group I elements Group I element Normal oxide Peroxide Superoxide Li Li2O – – Na Na2O Na2O2 – K K2O K2O2 KO2 Rb Rb2O Rb2O2 RbO2 Cs Cs2O Cs2O2 CsO2 Cations with high charge densities (Li+ or Na+) tend to polarize the large electron clouds of peroxide ions and/or superoxide ions Making them decompose to give oxide ions 37 1. Group I Elements The electron cloud of the superoxide ion is greatly distorted by the small lithium ion 38 Oxides formed by Group I elements Group I element Normal oxide Peroxide Superoxide Li Li2O – – Na Na2O Na2O2 – K K2O K2O2 KO2 Rb Rb2O Rb2O2 RbO2 Cs Cs2O Cs2O2 CsO2 White solids Slightly coloured solids Highly coloured solids 39 KO2 used as oxygen generators and CO2 scrubbers in spacecrafts 4KO2 + 2H2O 4KOH + 3O2 2KOH + CO2 K2CO3 + H2O 40 2. Group II Elements • Beryllium, magnesium and calcium form normal oxides only on burning in air 2Be(s) + O2(g) 2BeO(s) 2Mg(s) + O2(g) 2MgO(s) 2Ca(s) + O2(g) 2CaO(s) 41 Q.2(a) Be2+, Mg2+ and Ba2+ have higher charge densities more polarizing distort the electron cloud of O22 O22 decomposes to give O2 42 Q.2(b) 2Sr(s) + O2(g) 2SrO(s) strontium oxide 2SrO(s) + O2(g) Sr(s) + O2(g) 43 2SrO2(s) strontium peroxide SrO2(s) Q.2(b) 2Ba(s) + O2(g) 2BaO(s) barium oxide 2BaO(s) + O2(g) 500C 700C 2BaO2(s) barium peroxide Ba(s) + O2(g) 44 BaO2(s) Oxides formed by Group II elements Group II Normal oxide element Peroxide Superoxide Be BeO – – Mg MgO – – Ca CaO – – Sr SrO SrO2 – Ba BaO BaO2 – KO2 superoxide 45 Oxides formed by Group II elements Group II Normal oxide element Peroxide Superoxide Be BeO – – Mg MgO – – Ca CaO – – Sr SrO SrO2 – Ba BaO BaO2 – All these oxides are basic in nature (except beryllium oxide which is amphoteric) 46 Formation of hydroxides 1. Group I hydroxides 2Li(s) + 2H2O(l) 2LiOH(aq) + H2(g) 2Na(s) + 2H2O(l) 2NaOH(aq) + H2(g) 2K(s) + 2H2O(l) 2KOH(aq) + H2(g) 2Rb(s) + 2H2O(l) 2RbOH(aq) + H2(g) 2Cs(s) + 2H2O(l) 2CsOH(aq) + H2(g) 47 Formation of hydroxides 1. Group I hydroxides For normal oxides, M2O(s) + H2O(l) 2MOH(aq) For peroxides, M2O2(s) + 2H2O(l) 2MOH(aq) + H2O2(aq) For superoxides, 2MO2(s) + 2H2O(l) 2MOH(aq) + H2O2(aq) + O2(g) 48 Formation of hydroxides 2. Group II hydroxides Ca(s) + 2H2O(l) Ca(OH)2(aq) + H2(g) Sr(s) + 2H2O(l) Sr(OH)2(aq) + H2(g) Ba(s) + 2H2O(l) Ba(OH)2(aq) + H2(g) Mg reacts with steam but not water. Mg(s) + H2O(g) MgO(s) + H2(g) Be does not react with water and steam. 49 Formation of hydroxides 2. Group II hydroxides CaO(s) + H2O(l) Ca(OH)2(aq) SrO(s) + H2O(l) Sr(OH)2(aq) BaO(s) + H2O(l) Ba(OH)2(aq) MgO(s) + H2O(l) slightly soluble Mg(OH)2(aq) BeO(s) + H2O(l) No reaction 50 Ionic Bonding with Fixed Oxidation State in their Compounds Predominantly ionic Group I : +1 ∵ Low 1st I.E. but very high 2nd I.E. Group II : +2 ∵ Low 1st and 2nd I.E. but very high 3rd I.E. 51 Chemical formulae of some Group I compounds and the oxidation states of Group I elements in the compounds Group I element Oxide Hydride Chloride Oxidation state of Group I element in the compound Li Li2O LiH LiCl +1 Na Na2O2 NaH NaCl +1 K KO2 KH KCl +1 Rb RbO2 RbH RbCl +1 Cs CsO2 CsH CsCl +1 52 Chemical formulae of some Group II compounds and the oxidation states of Group II elements in the compounds Group II element Oxide Hydride Chloride Oxidation state of Group II element in the compound Be BeO BeH2 BeCl2 +2 Mg MgO MgH2 MgCl2 +2 Ca CaO CaH2 CaCl2 +2 Sr SrO SrH2 SrCl2 +2 Ba BaO BaH2 BaCl2 +2 53 Weak Tendency to Form Complexes A complex is formed when a central metal atom or ion is surrounded by other molecules or ions (called ligands) which form dative covalent bonds with the central metal atom or ion using their lone pair. 54 Weak Tendency to Form Complexes Unlike transition metals, all s-block metals (except Be) show little tendency to form complexes 55 Weak Tendency to Form Complexes Reasons : 1. Absence of low-lying vacant d-orbtals to accept lone pairs from ligands. For Na+, 1s2, 2s2, 2p6, 3s, 3p, 3d High-lying relative to 2p For Fe2+, 1s2, 2s2, 2p6, 3s2, 3p3, 3d6 Low-lying relative to 3p 56 Weak Tendency to Form Complexes Reasons : 2. s-block cations (M+, M2+) have relatively low charge densities less polarizing and less able to accept lone pairs from ligands. 57 A complex ion, [Co(NH3)6]3+ NH 3 NH 3 H3N Co3+ NH 3 H3N All six bonds are strong dative covalent bonds NH 3 A hydrated ion, Na+(aq) OH 2 H2O Na + H2O OH 2 58 OH 2 Dipole-ion attraction OH 2 Weaker than dative bond Weak Tendency to Form Complexes Owing to its high charge density, Be2+ can form complexes 59 [Be(H2O)4]2+(aq) + H2O(l) [Be(H2O)3(OH)]+(aq) + H2O(l) [Be(H2O)2 (OH)2](s) + H2O(l) [Be(H2O)(OH)3 ] (aq) + H2O(l) [Be(H2O)3(OH)]+(aq) + H3O+(aq) [Be(H2O)2(OH)2](s) + H3O+(aq) [Be(H2O)(OH)3](aq) + H3O+(aq) [Be(OH)4]2(aq) + H3O+(aq) Overall reaction : (1) + (2) + (3) + (4) [Be(H2O)4]2+(aq) + 4H2O(l) 60 [Be(OH)4]2(aq) + 4H3O+(aq) [Be(H2O)4]2+(aq) + H2O(l) [Be(H2O)3(OH)]+(aq) + H2O(l) [Be(H2O)2 (OH)2](s) + H2O(l) [Be(H2O)(OH)3 ] (aq) + H2O(l) [Be(H2O)3(OH)]+(aq) + H3O+(aq) (1) [Be(H2O)2(OH)2](s) + H3O+(aq) (2) [Be(H2O)(OH)3](aq) + H3O+(aq) (3) [Be(OH)4]2(aq) + H3O+(aq) (4) Overall reaction : (1) + (2) + (3) + (4) [Be(H2O)4]2+(aq) + 4H2O(l) [Be(OH)4]2(aq) + 4H3O+(aq) pH equilibrium positions shifts to the right 61 [Be(H2O)4]2+(aq) + H2O(l) [Be(H2O)3(OH)]+(aq) + H2O(l) [Be(H2O)2 (OH)2](s) + H2O(l) [Be(H2O)(OH)3 ] (aq) + H2O(l) [Be(H2O)3(OH)]+(aq) + H3O+(aq) (1) [Be(H2O)2(OH)2](s) + H3O+(aq) (2) [Be(H2O)(OH)3](aq) + H3O+(aq) (3) [Be(OH)4]2(aq) + H3O+(aq) (4) (1) + (2) [Be(H2O)4]2+(aq) + 2H2O(l) [Be(H2O)2(OH)2](s) + 2H3O+(aq) + 2OH(aq) [Be(H2O)4]2+(aq) + 2OH(aq) + 2OH(aq) [Be(H2O)2(OH)2](s) + 2H2O Or simply, Be2+(aq) + 2OH(aq) 62 Be(OH)2(s) Characteristic Flame Colours of Salts Most s-block elements and their compounds give a characteristic flame colour in the flame test Group I element Li Na K Rb Cs 63 Flame colour Crimson Golden yellow Lilac Bluish red Blue Group II element Be Mg Ca Sr Ba Flame colour Bright white Brick red Blood red Apple green Mechanism : 1. In the hotter part of the flame, Na(g) heat Ground state [Ne] 3s1 Na(g)* [Ne] 3p1 2. In the cooler part of the flame, Na(g)* [Ne] 3p1 64 cool Na(g) + golden yellow light [Ne] 3s1 Visible region Mechanism : For salts of s-block elements, the metal ions of the salts are first converted to metal atoms Na2CO3(s) Na+Cl 65 heat Na(g) heat Na(g)* cool Conc. HCl Na+Cl (more volatile) Na(g) + Cl(g) Na(g)* Na(g) + golden yellow light Q.3 heat Na+(g) [He] 2s2 2p6 Na+(g)* Na+(g)* [He] 2s2 2p5 3s1 cool [He] 2s2 2p5 3s1 Na+(g) + uv light [He] 2s2 2p6 3p visible 3s uv 2p 66 Li Na K Ca Pt or nichrome(an alloy of Ni and Cr) is suitable for making the wire because 1.They have no reaction with conc. HCl 2.They do not impart visible light when heated 67 Variation in Physical Properties of s-block Elements 1. Atomic Radius and Ionic Radius 2. Ionization Enthalpies 3. Hydration Enthalpies 4. Melting Points 68 1. Atomic Radius and Ionic Radius Group I element Atomic radius (nm) Group II element Atomic radius (nm) Li 0.152 Be 0.112 Na 0.186 Mg 0.160 K 0.231 Ca 0.197 Rb 0.244 Sr 0.215 Cs 0.262 Ba 0.217 Fr 0.270 Ra 0.220 down the groups ∵ the outermost electrons are further away from the nuclei 69 1. Atomic Radius and Ionic Radius Group I element Atomic radius (nm) Group II element Atomic radius (nm) Li 0.152 Be 0.112 Na 0.186 Mg 0.160 K 0.231 Ca 0.197 Rb 0.244 Sr 0.215 Cs 0.262 Ba 0.217 Fr 0.270 Ra 0.220 Group II < Group I ∵ ENC from left to right across the periods 70 71 On moving down the groups, first sharply (e.g. from Li to K) then slowly (e.g. from K to Fr) 1. There is a sharp in NC from 19K to 37Rb Outermost e is drawn closer to the nucleus 72 73 2. The inner d-electrons (of Rb, Cs, Sr, Ba) have poor shielding effect on the outermost electrons transition contraction 2. Ionization Enthalpy Group I element 1st IE 2nd IE Group II element 1st IE 2nd IE 3rd IE Li 519 7 300 Be 900 1 760 14 800 Na 494 4 560 Mg 736 1 450 7 740 K 418 3 070 Ca 590 1 150 4 940 Rb 402 2 370 Sr 548 1 060 4 120 Cs 376 2 420 Ba 502 966 3 390 Fr 381 – Ra 510 979 – Both atomic radius and ENC down the groups Atomic radius is more important IE down the groups 74 2. Ionization Enthalpy Group I element 1st IE 2nd IE Group II element 1st IE 2nd IE 3rd IE Li 519 7 300 Be 900 1 760 14 800 Na 494 4 560 Mg 736 1 450 7 740 K 418 3 070 Ca 590 1 150 4 940 Rb 402 2 370 Sr 548 1 060 4 120 Cs 376 2 420 Ba 502 966 3 390 Fr 381 – Ra 510 979 – For Group I elements, 2nd IE >> 1st IE because 1.the outer s-electron is well shielded by inner shell electrons 75 2. Ionization Enthalpy Group I element 1st IE 2nd IE Group II element 1st IE 2nd IE 3rd IE Li 519 7 300 Be 900 1 760 14 800 Na 494 4 560 Mg 736 1 450 7 740 K 418 3 070 Ca 590 1 150 4 940 Rb 402 2 370 Sr 548 1 060 4 120 Cs 376 2 420 Ba 502 966 3 390 Fr 381 – Ra 510 979 – For Group I elements, 2nd IE >> 1st IE because 2. the 2nd electron is closer to the nucleus and is poorly shielded by other electrons in the 76 same shell which is completely filled. 2. Ionization Enthalpy Group I element 1st IE 2nd IE Group II element 1st IE 2nd IE 3rd IE Li 519 7 300 Be 900 1 760 14 800 Na 494 4 560 Mg 736 1 450 7 740 K 418 3 070 Ca 590 1 150 4 940 Rb 402 2 370 Sr 548 1 060 4 120 Cs 376 2 420 Ba 502 966 3 390 Fr 381 – Ra 510 979 – For Group II elements, 3rd IE >> 2nd IE Similar reasons can be applied 77 Variations in the first and second ionization enthalpies of Group I elements 78 Variations in the first, second and third ionization enthalpies of Group II elements 79 2. Ionization Enthalpy Group I element 1st IE 2nd IE Group II element 1st IE 2nd IE 3rd IE Li 519 7 300 Be 900 1 760 14 800 Na 494 4 560 Mg 736 1 450 7 740 K 418 3 070 Ca 590 1 150 4 940 Rb 402 2 370 Sr 548 1 060 4 120 Cs 376 2 420 Ba 502 966 3 390 Fr 381 – Ra 510 979 – Group II > Group I ∵ The outer s-electrons of Group II atoms are closer to the nucleus and experience higher ENC 80 3. Hydration enthalpy Hydration enthalpy (Hhyd) is the amount of energy released when one mole of aqueous ions is formed from its gaseous ions. M+(g) + aq M+(aq) H = Hhyd M2+(g) + aq M2+(aq) H = Hhyd always has a negative value 81 Group I ion Hydration enthalpy (kJ mol–1) Group II ion Hydration enthalpy (kJ mol–1) Li+ –519 Be 2+ –2 450 Na+ –406 Mg2+ –1 920 K+ –322 Ca2+ –1 650 Rb+ –301 Sr2+ –1 480 Cs+ –276 Ba2+ –1 360 Fr+ – Ra2+ – down the groups ∵ charge density of metal ions down the groups attraction between ions and water molecules + 82 Group I ion Hydration enthalpy (kJ mol–1) Group II ion Hydration enthalpy (kJ mol–1) Li+ –519 Be 2+ –2 450 Na+ –406 Mg2+ –1 920 K+ –322 Ca2+ –1 650 Rb+ –301 Sr2+ –1 480 Cs+ –276 Ba2+ –1 360 Fr+ – Ra2+ – Group II > Group I ∵ Group II ions have higher charge and small size higher charge density stronger ion-dipole interaction 83 Variations in hydration enthalpy of the ions of Groups I and II elements 84 4. Melting Point The melting points of s-block elements depend on the metallic bond strength which in turn depends on 1. charge density of cations 2. number of valence electrons participating in the sea of electrons 3. packing efficiency of the crystal lattices 85 Group I element Melting Point (C) Group II element Melting Point (C) Li 180 Be 1280 Na 97.8 Mg 650 K 63.7 Ca 850 Rb 38.9 Sr 768 Cs 28.7 Ba 714 Fr 24 Ra 697 1. down the groups ∵ ionic radii down the groups charge density interaction between ions and electron sea 86 Group I element Melting Point (C) Group II element Melting Point (C) Li 180 Be 1280 Na 97.8 Mg 650 K 63.7 Ca 850 Rb 38.9 Sr 768 Cs 28.7 Ba 714 Fr 24 Ra 697 2. Group II > Group I ∵ (a)Group II cations have higher charge density (b) More valence electrons are involved in the sea of electrons (c)Packing efficiency : Group II > Group I 87 Reason not known !! 88 Variation in Chemical Properties s-Block elements have strong reducing power ∵ low ionization enthalpies low atomization enthalpies 89 M+(g) ~Ea Ionization enthalpy M(g) M(s) Atomization enthalpy Hydration enthalpy ΔH0 reaction 0 M+(aq) M(s) M+(aq) + e 90 H < 0 M+(g) ~Ea Ionization enthalpy M(g) M(s) Atomization enthalpy Hydration enthalpy ΔH0 reaction 0 M+(aq) Reactivity : Na > Ca (depends on Ea) 91 Position in e.c.s. : Ca > Na (depends on Ho or Eo Variation in Chemical Properties The reactivity of s-block elements down the groups ∵ both I.E. and A.E. down the groups Ea down the groups Reaction rate down the groups 92 Variation in Chemical Properties Reactivity : Group I > Group II ∵ both I.E. and A.E. across the periods Ea across the periods Reaction rate across the periods 93 1. Reactions with hydrogen Group I 2M(s) + H2(g) Group II M(s) + H2(g) 94 300C – 500C 600C – 700C 2MH(s) MH2(s) 1. Reactions with hydrogen 4LiH + AlCl3 Dry ether LiAlH4 + 3LiCl Reducing agent in organic syntheses 95 2. Reactions with Oxygen Most s-block elements show a silvery white lustre when they are freshly cut they tarnish rapidly upon exposure to the atmosphere 96 they react with oxygen in the air to form an oxide layer Sodium shows a silvery white lustre when freshly cut 97 Group I (p.2) Group II 2M(s) + O2(g) M(s) + O2(g) 98 heat heat 2MO(s) MO2(s) 3. Reactions with Chlorine Group I 2M(s) + Cl2(g) Group II M(s) + Cl2(g) 99 heat heat 2MCl(s) MCl2(s) 4. Reactions with water or steam Group I 2M(s) + H2O(l) Group II M(s) + 2H2O(l) heat heat 2MOH(aq) + H2(g) M(OH)2(aq) + H2(g) Mg reacts with steam but not water Mg(s) + H2O(g) 100 heat MgO(s) + H2(g) Be has no reaction with either water or steam Variation in chemical properties of the compounds of s-block elements Reactions of oxides Reactions of hydrides Reactions of chlorides 101 Reactions of oxides 1. Reactions with water Group I M2O(s) + H2O(l) 2MOH(aq) M2O2(s) + 2H2O(l) 2MOH(aq) + H2O2(aq) 2MO2(s) + 2H2O(l) 2MOH(aq) + H2O2(aq) + O2(g) 102 Na2O2 is used in qualitative analysis of Cr3+ green 2Cr(OH)3(s) + 3Na2O2(s) 2Na2CrO4(aq) + 2NaOH(aq) + 2H2O(l) yellow 103 Reactions of oxides Group II CaO(s) + H2O(l) Ca(OH)2(aq) SrO(s) + H2O(l) Sr(OH)2(aq) BaO(s) + H2O(l) Ba(OH)2(aq) MgO(s) + H2O(l) slightly soluble Mg(OH)2(aq) BeO(s) + H2O(l) No reaction 104 increasing basicity Reactions of oxides 2. Reactions with acids Group I M2O(s) + 2HCl(aq) 2MCl(aq) + H2O(l) M2O2(s) + 2HCl(aq) 2MCl(aq) + H2O2(aq) 2MO2(s) + 2HCl(aq) 2MCl(aq) + H2O2(aq) + O2(g) Group II More vigorous than those with water MO(s) + 2HCl(aq) MCl2(aq) + H2O(l) 105 Reactions of oxides 3. Reactions with alkalis Reaction with water instead except BeO BeO(s) + 2OH(aq) + H2O(l) Be(OH)42(aq) amphoteric 106 Reactions of hydrides MOH(aq) + H2(g) MH(s) MCl(aq) + H2(g) H (a strong base) tends to react with protonic reagents to release H2 Reactivity down the groups 107 Reactions of chlorides Group I No significant reactions with water, acids or alkalis Group II Do not undergo significant hydrolysis except BeCl2 and MgCl2 More favoured in alkaline solutions BeCl2(aq) + 2H2O(l) Be(OH)2(aq) + 2HCl(aq) MgCl2(aq) + H2O(l) Mg(OH)Cl(aq) + HCl(aq) Basic salt 108 Relative Thermal Stability of the Carbonates and Hydroxides of s-Block Elements Thermal stability refers to the resistance of a compound to undergo decomposition on heating. 109 Thermal decomposition reactions Metal carbonates M2CO3(s) heat M2O(s) + CO2 MCO3(s) heat MO(s) + CO2 Metal hydroxides 110 2MOH(s) heat M2O(s) + H2O(g) M(OH)2(s) heat MO(s) + H2O Relative thermal stability can be measured in two ways 1. By comparing the decomposition temperatures A higher decomposition temperature a greater thermal stability 111 Metal carbonate Decomposition temperature /C BeCO3 MgCO3 CaCO3 SrCO3 BaCO3 ~100 540 900 1290 1360 Decomposition temperature is the temperature at which the pressure of CO2 in equilibrium with the solid carbonate reaches 1 atm in a closed system. Below the DT, some CO2 can still be detected but the pressure is less than 1 atm 112 1. The Carbonates • Example: 100 C BeCO3(s) BeO(s) + CO2(g) 540 C MgCO3(s) MgO(s) + CO2(g) 900 C CaCO3(s) CaO(s) + CO2(g) 1290 C SrCO3(s) SrO(s) + CO2(g) 1360 C BaCO3(s) BaO(s) + CO2(g) 113 Relative thermal stability can be measured in two ways 2. By comparing the standard enthalpy changes of thermal decomposition reactions M(OH)2(s) MO(s) + H2O(g) H > 0 A more positive H value a thermally more stable compound 114 Metal hydroxide Ho / kJ mol1 Be(OH)2 Mg(OH)2 Ca(OH)2 Sr(OH)2 Ba(OH)2 +54 +81 +109 +127 +146 Trends : 1. down the groups 2. Group I > Group II 3. Li resembles Mg more than the other group 1 elements (diagonal relationship, pp.14-15) 115 2. The Hydroxides Be(OH)2(s) BeO(s) + H2O(g) H = +54 kJ mol–1 Mg(OH)2(s) MgO(s) + H2O(g) H = +81 kJ mol–1 Ca(OH)2(s) CaO(s) + H2O(g) H = +109 kJ mol–1 Sr(OH)2(s) SrO(s) + H2O(g) H = +127 kJ mol–1 116 Ba(OH)2(s) BaO(s) + H2O(g) H = +146 kJ mol–1 Factors affecting thermal stability of carbonates and hydroxides 1. Polarizing power of cation 2. Polarizability of polyatomic anion 3. Lattice enthalpy of metal oxide produced 117 Interpretation of trends in thermal stability of carbonates and hydroxides 1. Group I > Group II (a) M2+ ions have higher charge densities than M+ ions M2+ ions are more polarizing than M+ ions Can polarize more the electron cloud of polyatomic anions 118 polarization M2+ O O heat C MO + CO2 O polarization M2+ 119 heat O H O H MO + H2O Polarizability as the size of anion 120 Thermal decomposition Polyatomic ion 121 When a compound with large anions undergoes thermal decomposition, a compound with small anions will be formed since small anions are less easily polarized 122 more stable compound with stronger bond Simple ion 123 M2+ S2 Simple ion 124 polarization M S Stronger ionic bond with covalent character Interpretation of trends in thermal stability of carbonates and hydroxides 1. Group I > Group II (b) M2+ ions have higher charge densities than M+ ions Lattice enthalpy : MO > M2O Energetic stability : MO > M2O 125 CaCO3(s) Na2CO3(s) more favourable heat less favourable heat CaO(s) + CO2(g) more stable Na2O(s) + CO2(g) less stable Thermal stability of carbonates : Group I > Group II 126 Interpretation of trends in thermal stability of carbonates and hydroxides 2. Thermal stability down the groups ∵ size of cations down the groups ∴ (a) charge density/polarizing power of cation down the groups (b) lattice enthalpies of MO/M2O down the groups 127 MgCO3(s) more favourable more polarized BaCO3(s) less polarized heat less favourable heat MgO(s) + CO2(g) more stable BaO(s) + CO2(g) less stable Thermal stability of carbonates : down the groups 128 Effect of sizes of the cations on thermal stability of the carbonates and hydroxides of both Groups I and II metals 129 Interpretation of trends in thermal stability of carbonates and hydroxides 3. Li compounds resemble Mg compounds (diagonal relationship) Charge density/polarizing power : Li+ Mg2+ 130 Interpretation of trends in thermal stability of carbonates and hydroxides 4. Thermal stability of nitrates follows similar patterns (Optional) 2MNO3(s) 2M(NO3)2(s) 131 heat heat 2MNO2 + O2 2MO + 4NO2 + O2 Relative Solubility of the Sulphates(VI) and Hydroxides of s-Block Elements In general, Group I >> Group II 132 Q.4 Compounds Mg(OH)2 Ca(OH)2 Sr(OH)2 Ba(OH)2 Solubility / mol per 100 of water 0.02 103 1.5 103 3.4 103 15 103 Solubility / mol per Compounds 100 of water MgSO4 1800 104 CaSO4 11 104 SrSO4 0.71 104 BaSO4 0.009 104 133 In general, Size and/or charge of the anion Polarizability of anion Covalent character Solubility in water Solubility / mol per Compounds 100 of water Mg(OH)2 0.02 103 Ca(OH)2 1.5 103 Sr(OH)2 3.4 103 Ba(OH)2 15 103 down the group Solubility / mol per 100 of water 1800 104 11 104 0.71 104 0.009 104 down the group Compounds MgSO4 CaSO4 SrSO4 BaSO4 134 1. Processes involved in Dissolution and their Energetics • Two processes are 1. the breakdown of the ionic lattice 2. the subsequent stabilization of the ions by water molecules (this process is called hydration) 135 1. the breakdown of the ionic lattice NaCl(s) Na+(g) + Cl(g) H1 = (lattice enthalpy) > 0 2. the subsequent stabilization of the ions by water molecules (this process is called hydration) Na+(g) + Cl(g) + aq Na+(aq) + Cl(aq) H2 = (hydration enthalpy) < 0 136 Hsolution NaCl(s) Na +(aq) + Cl-(aq) Na+(g) + Cl -(g) ΔH o solution ΔH o hydration ΔH o lattice = (-772 +776) kJ mol1 137 = +4 kJ mol1 o ΔH If solution 0 , we expect the solids to dissolve in water o Solubility as ΔHsolution becomes more –ve (less +ve) o Solids (e.g. NaCl) with small +ve ΔHsolution values are also soluble in water if the dissolution involves an increase in the entropy of the system. ΔG o solution 138 ΔH o solution TS o solution ΔG o solution ΔG o solution ΔH o solution 0 TS o solution Spontaneous dissolution TS o is always positive solution Dissolution with slightly positive can be spontaneous 139 H o solution Trends and Interpretations 1. The solubility of Group(II) sulphate decreases down the group On moving down the group, cationic radius(r+) both H However, 140 o L and H H o hydration become less -ve o L less rapidly than H o hydration Trends and Interpretations rSO 2 r 4 ΔH o L 1 rSO 2 r constant 4 ΔH o solution less –ve down the group 141 ΔH o hydration less –ve down the group ΔH o lattice +ve constant Solubility down the group Trends and Interpretations rSO 2 r 4 ΔH o L 1 rSO 2 r 4 ΔH o solution less –ve down the group 142 constant (-ve) ΔH o hydration more rapidly down the group (+ve) ΔH o lattice less rapidly down the group Solubility down the group Trends and Interpretations 2. The solubility of Group(II) hydroxides increases down the group On moving down the group, cationic radius(r+) both H However, 143 o L and H H o hydration become less -ve o o Hhydration L more rapidly than Trends and Interpretations ΔH o solution (-ve) ΔH less +ve more –vedown down the group o hydration less rapidly down the group (+ve) ΔH o lattice more rapidly down the group Solubility down the group 144 For s-block compounds with small anions (e.g. OH, F), solubility in water down the group For s-block compounds with large anions (e.g. SO42, CO32-), solubility in water down the group For s-block compounds with medium size anions (e.g. Br), solubility in water exhibits irregular pattern down the group 145 Solubility / mol per Compounds 100 of water 5.5 101 MgBr2 CaBr2 6.3 101 SrBr2 4.3 101 BaBr2 3.3 101 Irregular Solublily : First and then ΔH o solution (-ve) ΔH o hydration Then less -ve First +ve Then more down the group rapidly 146 (+ve) ΔH o lattice First more rapidly Group II compounds with doubly-charged anions (MX) are less soluble than those with singly-charged anions (MY2) Reasons : 1. HL of MX > HL of MY2 2. HL is the major factor affecting solubility Hsolution of MX is more positive Solubility : MX < MY2 147 Solubility : Group I > Group II Reasons : For a given anions, both HL and Hhydration become more –ve from Group I to Group II However, HL is the major factor affecting solubility Hsolution : Group I is less positve than Group II Solubility : Group I > Group II 148 Diagonal relationship 149 Reaction Other Group I elements Lithium Magnesium Combination with O2 Peroxides and superoxides Li2O (normal oxide) MgO (normal oxide) Combination with N2 No reaction Li3N Mg3N2 Action of heat on carbonate No reaction (thermally stable) Decomposes to give Li2O and CO2 Decomposes to give MgO and CO2 Action of heat on hydroxide No reaction (thermally stable) Decomposes to give Li2O and H2O Decomposes to give MgO and H2O Action of heat on nitrate Decomposes to give MNO2 and O2 Decomposes to give Li2O, NO2 and O2 Decomposes to give MgO, NO2 and O2 Hydrogen carbonates Exist as solids Only exist in solution Solubility of salts in water Most salts are more soluble than those of Li, Mg. Fluoride, hydroxide, carbonate, phosphate, ethanedioate are sparingly soluble. Solubility of salts in organic solvents. Halides only slightly soluble in organic solvents Halides (with covalent character) dissolve in organic solvents 150 The END 151 40.1 Characteristic Properties of the s-Block Elements (SB p.40) Metals are sometimes referred to as electropositive elements. Why? Answer They have low electronegativity values. Back 152 40.1 Characteristic Properties of the s-Block Elements (SB p.46) s-Block compounds give a characteristic flame colour in the flame test. Based on this, can you give one use of s-block compounds? Answer s-Block compounds can be used in fireworks. Back 153 40.1 Characteristic Properties of the s-Block Elements (SB p.48) (a) Which ion has a greater ionic radius, potassium ion or calcium ion? Explain your answer. Answer (a) Potassium ion (0.133 nm) has a greater ionic radius than calcium ion (0.099 nm) . In fact, potassium ion and calcium ion are isoelectronic and have the same number of electron shells. However, calcium ion has one more proton than potassium ion, the electron cloud of calcium ion will experience greater attractive forces from the nucleus. This leads to a smaller ionic radius of calcium ion. 154 40.1 Characteristic Properties of the s-Block Elements (SB p.48) (b) Explain why Group I elements show a fixed oxidation state of +1 in their compounds in terms of ionization enthalpies. Answer 155 40.1 Characteristic Properties of the s-Block Elements (SB p.48) (b) Group I elements form ions with an oxidation state of +1 only. It is because they have only one outermost shell electron. Once this outermost shell electron is removed, a stable fully-filled electronic configuration is obtained. Therefore, the first ionization enthalpies of Group I elements are low. The second ionization involves the removal of an electron from an inner electron shell. Once this electron is removed, the stable electronic configuration will be disrupted. Therefore, their second ionization enthalpies are very high. As a result, Group I elements form predominantly ionic compounds with non-metals by losing their single outermost shell electron, and they form ions having a fixed oxidation state of +1. 156 40.1 Characteristic Properties of the s-Block Elements (SB p.48) (c) Ions of Group I and Group II elements have a very low tendency to form complexes. Give one reason to explain your answer. Answer (c) As ions of Group I and Group II elements do not have low-lying vacant orbitals available for forming dative covalent bonds with the lone pair electrons of surrounding ligands, they rarely form complexes. 157 40.1 Characteristic Properties of the s-Block Elements (SB p.48) (d) Give one test which would enable you to distinguish a sodium compound from a potassium compound. Answer (d) Sodium compounds and potassium compounds can be distinguished by conducting a flame test. In the flame test, sodium compounds give a golden yellow flame, while potassium compounds give a lilac flame. Back 158 40.1 Characteristic Properties of the s-Block Elements (SB p.48) What is a dative covalent bond? How is it formed? Answer A dative covalent bond is a covalent bond in which the shared pair of electrons is supplied by only one of the bonded atoms. A dative covalent bond is formed by the overlapping of an empty orbital of an atom with an orbital occupied by a lone pair of electrons of another atom. Back 159 40.2 Variation in Properties of the s-Block Elements (SB p.56) (a) (i) List the factors that affect the value of the ionization enthalpy of an atom. Answer (a) (i) 160 There are four main factors affecting the magnitude of the ionization enthalpy of an atom. They are the electronic configuration of an atom, the nuclear charge, the screening effect, and the atomic radius. 40.2 Variation in Properties of the s-Block Elements (SB p.56) (a) (ii) Why is ionization enthalpy of an atom always positive? Answer (a) (ii) 161 Ionization enthalpy of an atom always has a positive value because energy is required to overcome the attractive forces between the nucleus and the electron to be removed. 40.2 Variation in Properties of the s-Block Elements (SB p.56) (a) (iii) Describe the general trend of the first and second ionization enthalpies down Group I of the Periodic Table. Answer 162 40.2 Variation in Properties of the s-Block Elements (SB p.56) (a) (iii) The first ionization enthalpies of Group I elements are relatively low. The outermost s electron is located in a new electron shell. The attractive force between this s electron and the nucleus is relatively weak. Also, this s electron is effectively shielded from the attraction of the nucleus by the fully-filled inner electron shells. Once this electron is removed, a stable octet or duplet electronic configuration is obtained. Consequently, this s electron is relatively easy to be removed, and hence the first ionization enthalpies of Group I elements are relatively low. However, the second ionization of Group I elements involves the loss of an inner shell electron which is closer to the nucleus. The removal of this electron disrupts the stable electronic configuration. Therefore, the second ionization enthalpies of Group I elements are extremely high. 163 40.2 Variation in Properties of the s-Block Elements (SB p.56) (b) (i) List the factors that affect the value of the hydration enthalpy of an ion. Answer (b) (i) 164 The value of the hydration enthalpy of an ion depends on the size and the charge of the ion. 40.2 Variation in Properties of the s-Block Elements (SB p.56) (b) (ii) Why does hydration enthalpy of an ion always have a negative value? Answer (b) (ii) 165 Hydration enthalpy of an ion always has a negative value because it is the amount of energy released resulting from the attraction between the ion and water molecules. 40.2 Variation in Properties of the s-Block Elements (SB p.56) (b) (iii) Describe the general trend of the hydration enthalpy down Group II of the Periodic Table. Answer (b) (iii) Going down Group II, the hydration enthalpy of the ions decreases (becomes less negative). Since the ions get larger in size on moving down the group, the charge density of the ions falls. As a result, the electrostatic attraction between the ions and water molecules becomes weaker, and the hydration enthalpy becomes less negative down the group. Back 166 40.2 Variation in Properties of the s-Block Elements (SB p.57) The burning of lithium, sodium and potassium in oxygen gives different types of oxides. Why do the metals behave differently? Answer 167 40.2 Variation in Properties of the s-Block Elements (SB p.57) On burning in air, lithium forms only lithium oxide, and it does not form the peroxide or superoxide. This is because the size of lithium ion is very small, leading to its high polarizing power. When a peroxide ion or superoxide ion approaches a lithium ion, the electron cloud of the peroxide ion or superoxide ion (large in size) would be greatly distorted by the lithium ion. The greater the distortion of the electron cloud, the lower the stability of the compound. That is why lithium peroxide and lithium superoxide do not exist. Sodium ion has a larger size than lithium ion. Its lower polarizing power allows it to form the peroxide when sodium is burnt in air. Potassium ion has a much larger size, so it has relatively low polarizing power. The electron cloud of the peroxide ion or superoxide ion would not be seriously distorted by potassium ion. This allows the peroxide ions or superoxide ions to pack around potassium ion with a higher stability. As a result, potassium is able to form stable peroxide or superoxide on burning in air. Back 168 40.2 Variation in Properties of the s-Block Elements (SB p.58) (a) Suggest a reason why the reaction of lithium with water is less vigorous than those of sodium and potassium. Answer (a) The reactivity of Group I metals with water is related to the relative ease of the metal atoms to lose the outermost shell electron. Going down the group, as the atomic size increases, the outermost shell electron becomes easier to be removed. Therefore, the reactivity of Group I metals towards water increases down the group. Lithium reacts with water vigorously. Sodium reacts with water violently and moves on the water surface with a hissing sound. 169 40.2 Variation in Properties of the s-Block Elements (SB p.58) (b) Which element is the strongest reducing agent, calcium, strontium or barium? Answer (b) Barium is the strongest reducing agent. It is because the reducing power of an element is related to the ease of the atom to lose the outermost shell electron. Since barium has larger atomic sizes, its outermost shell electrons are less firmly held by the nucleus. Therefore, barium has a higher tendency to lose its outermost shell electrons than both calcium and strontium. Back 170 40.3 Variation in Properties of the Compounds of the s-Block Elements (SB p.64) The value of Hsoln of a solid does not indicate whether the solid is soluble in water or not. So how can we predict the solubility of a solid in water? Answer Generally speaking, for a solid to be soluble in water, its enthalpy change of solution has to be a negative or a small positive value. Back 171 40.3 Variation in Properties of the Compounds of the s-Block Elements (SB p.65) (a) Give balanced chemical equations for the following reactions: (i) Thermal decomposition of barium carbonate (ii) Reaction between sodium peroxide and water (iii) Reaction between calcium oxide and dilute hydrochloric acid Answer (a) (i) BaCO3(s) BaO(s) + CO2(g) (ii) Na2O2(s) + 2H2O(l) 2NaOH(aq) + H2O2(aq) (iii) CaO(s) + 2HCl(aq) CaCl2(aq) + H2O(l) 172 40.3 Variation in Properties of the Compounds of the s-Block Elements (SB p.65) (b) Suggest a reason why barium sulphate(VI) is insoluble in water, while potassium sulphate(VI) is soluble in water although they have cations of similar sizes and the same anion. (The ionic radii of potassium ion and barium ion are 0.133 nm and 0.135 nm respectively.) Answer 173 40.3 Variation in Properties of the Compounds of the s-Block Elements (SB p.65) (b) When an ionic solid dissolves in water, two processes are taking place. They are the breakdown of the ionic lattice and the subsequent stabilization of the ions by water molecules. The enthalpy change involved in the whole dissolution process is known as the enthalpy change of solution, Hsoln, which is equal to Hsoln = Hhyd – Hlattice. For an ionic compound to be soluble in water, the enthalpy change of solution has to be a negative or a small positive value. The reason why barium sulphate(VI) is insoluble in water while potassium sulphate(VI) is soluble in water is that potassium ion has a smaller charge than barium ion. The Hlattice of potassium sulphate(VI) is smaller in magnitude (less negative) than that of barium sulphate(VI). As a result, the enthalpy change of solution of potassium sulphate(VI) is more negative, and hence it is soluble in water while barium sulphate(VI) is not. 174 40.3 Variation in Properties of the Compounds of the s-Block Elements (SB p.65) (c) Compare the solubility of calcium sulphate(VI) and barium sulphate(VI) in water. Explain your answer. Answer (c) Calcium sulphate(VI) is expected to be more soluble than barium sulphate(VI). It is because calcium ion has a smaller size than barium ion. This causes the Hhyd of calcium sulphate(VI) to be more negative than that of barium sulphate(VI). As a result, the Hsoln of calcium sulphate(VI) becomes more negative than that of barium sulphate(VI), and hence calcium sulphate(VI) is more soluble in water than barium sulphate(VI). Back 175