* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Lect3
Survey
Document related concepts
Transcript
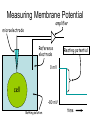
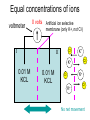
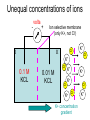
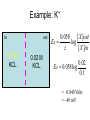
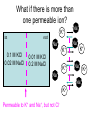
Announcements: • Last lecture 1. Organization of the nervous system 2. Introduction to the neuron • Today – electrical potential 1. 2. 3. 4. Generating membrane potential Nernst equation Goldman equation Maintaining ionic distributions Neural Signaling A Simple Circuit Within neurons electrical Between neurons chemical & electrical Bioelectric Potentials • Neurons have an electrical potential (voltage) across the cell membrane • The inside of the cell is more negative than the outside – called the Resting Membrane Potential Measuring Membrane Potential amplifier microelectrode Reference electrode Resting potential 0 mV cell -80 mV Bathing solution time Electrophysiology techniques Silver / Silver chloride wire electrode Amplifier output Reference electrode 3M KCl solution Glass micropipette Very tiny hole (<<0.1m) Resting Membrane Potential • How is it generated? 1. differential distribution of ions inside and outside the cell 2. Selective Permeability of the membrane to some ions • How does unequal concentration of ions give rise to membrane potential ? Equal concentrations of ions voltmeter 0 volts Artificial ion selective membrane (only K+, not Cl-) I Cl- II K+ Cl- K+ 0.01 M KCL 0.01 M KCL Cl- K+ K+ Cl- No net movement Unequal concentrations of ions - volts + Ion selective membrane (only K+, not Cl-) I Cl- II K+ Cl- K+ 0.1 M KCL 0.01 M KCL Cl- K+ K+ + K - Cl K+ ClCl- K+ concentration gradient K+ Initial K+ ClCl- K+ ClK+ New Equilibrium Cl- K+ K+ Cl- Cl- Cl- K+ Cl- Cl- K+ K+ CHEMICAL Cl- K+ Cl- Cl- K+ ClCl- +K+ + + K+ + + K + + K + Cl- Cl- Cl- Cl- CHEMICAL ELECTRICAL Unequal concentrations of ions • Initial diffusion of K+ down concentration gradient from I to II • This causes + charge to accumulate in II because + and - charges are separated – Remember that Cl- can’t cross the membrane ! • Therefore II becomes positive relative to I Equilibrium Potential • As II becomes +, movement of K+ is repelled • Every K+ near the membrane has two opposing forces acting on it: 1. Chemical gradient 2. Electrical gradient • These two forces exactly balance each other • Called the electrochemical equilibrium • • • The electrical potential that develops is called the equilibrium potential for the ion. Electrical potential at which there is no net movement of the ion Note: 1. only a very small number of ions actually contribute to the electrical potential 2. the overall concentrations of K and Cl in solution do not change. • To calculate the equilibrium potential of any ion (eg. K, Na, Ca,) at any concentration – we use the Nernst Equation: Nernst Equation Gas Constant Temp (K) Ion Concentration I RT [ X ]I Ex ln zF [ X ]II Equilibrium Potential of X ion (eg. K+) in Volts Valence of ion (-1, +1, +2) Ion Concentration II Faraday constant Nernst Equation • At 18C, for a monovalent ion, and converting to log10 ,the equation simplifies to: X 0.058 [X ]I I 0.058 EXEx z log log[ X ]II z X II • By convention electrical potential inside of cells is expressed relative to the outside of the cell 0.058 [ X ]outside Ex log z [ X ]inside Example: K+ in out EK 0.1 M KCL X I 0.058 log z X II 0.02 M KCL 0.058 [ X ]out EK log z [ X ]in 0.02 EK 0.058log 0.1 = -0.040 Volts = - 40 mV • Therefore, – initial movement of K+ down concentration gradient – When electrical potential of -40 mV develops, there will be no net movement of K+ – Thus K+ is in electrochemical equilibrium What if there is more than one permeable ion? in 0.1 M KCl 0.02 M NaCl out K+ Na+ 0.01 M KCl 0.2 M NaCl Na+ K+ K+ Na+ K+ Na+ Na+ K+ Permeable to K+ and Na+, but not Cl- Na+ K+ • To calculate the overall potential of multiple ions • use the Goldman Equation • Considers the permeability of ions and their concentrations Goldman equation Voltage PK[ K ]outside PNa[ Na]outside PCl[Cl ]inside Vm 0.058log PK[ K ]inside PNa[ Na]inside PCl[Cl ]outside Permeability Ion concentration Because Cl is negative Goldman equation • Example, typical mammalian cell: 1. Assume permeability for Na is 1/100 of permeability for K, and permeability of Cl is 0 2. Assume [K]in= 140, [K]out=5 [Na]in =10, [Na]out=120 1[5] 0.01[120] 0 Vm 0.058log 1[140] 0.01[10] 0 Vm 78mV Goldman equation • The resting membrane potential of most cells is predicted by the Goldman equation Summary & Key Concepts 1. Unequal distributions of an ion across a selective membrane • causes an electrochemical potential called the equilibrium potential 2. Two opposing forces act on ions at the membrane 1. A chemical force down the concentration gradient 2. An opposing electrical force Summary & Key Concepts 3. The equilibrium potential for an ion is described by the Nernst equation 4. Cell membranes are permeable to more than one ion 5. the membrane electrical potential is described by the Goldman equation So What??? • Everything the nervous system and muscles do depends on the resting membrane potential Sample question • If two concentrations of KCl solution across a membrane give an equilibrium potential for K+ of -60 mV, what will the equilibrium potential be if the concentrations on each side are reversed A. B. C. D. -120 mV 0 +60 mV -30 mV