* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Review - American Physiological Society
Survey
Document related concepts
Polyclonal B cell response wikipedia , lookup
Psychoneuroimmunology wikipedia , lookup
Cancer immunotherapy wikipedia , lookup
Innate immune system wikipedia , lookup
Adoptive cell transfer wikipedia , lookup
Hygiene hypothesis wikipedia , lookup
Immunosuppressive drug wikipedia , lookup
Management of multiple sclerosis wikipedia , lookup
Multiple sclerosis signs and symptoms wikipedia , lookup
Pathophysiology of multiple sclerosis wikipedia , lookup
Sjögren syndrome wikipedia , lookup
Transcript
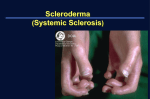
PHYSIOLOGY IN MEDICINE: A SERIES OF ARTICLES LINKING MEDICINE WITH SCIENCE Physiology in Medicine Dennis A. Ausiello, MD, Editor; Dale J. Benos, PhD, Deputy Editor; Francois Abboud, MD, Associate Editor; William Koopman, MD, Associate Editor Review Annals of Internal Medicine Paul Epstein, MD, Series Editor Following the Molecular Pathways toward an Understanding of the Pathogenesis of Systemic Sclerosis Sergio A. Jimenez, MD, and Chris T. Derk, MD Clinical Principles Physiologic Principles Systemic sclerosis is an autoimmune connective tissue disorder of unknown cause. The pathogenesis of systemic sclerosis is extremely complex; at present, no single unifying hypothesis explains all aspects. It is clinically heterogeneous, ranging from limited skin involvement to diffuse skin sclerosis with severe internal organ involvement. The Raynaud phenomenon, swelling of the extremities, and diffuse arthralgias often precede the onset of skin tightness and induration. Fundamental abnormalities in at least 3 types of cells are intimately involved in the development of the clinical and pathologic manifestations of the disease: 1) fibroblasts; 2) endothelial cells; and 3) cells of the immune system, in particular T and B lymphocytes. Autoantibodies, some with very high specificity, are present almost universally. The profound functional alterations in these cells result in the characteristic triad of pathologic changes: severe and often progressive cutaneous and visceral fibrosis; obliteration of the lumen of small arteries and arterioles; and humoral and cellular immunologic abnormalities, which include the production of autoantibodies, the chronic mononuclear cell infiltration of affected tissues, and the dysregulation of lymphokine and growth factor production. Mortality and morbidity are substantial and are directly related to the extent of the fibrotic and microvascular alterations. A crucial component in systemic sclerosis pathogenesis is the persistent and unregulated activation of genes encoding various collagens and other extracellular matrix proteins. A better understanding of the pathogenesis of this incurable disorder will help to design effective therapies in the future. Abnormal regulation of transcription of genes encoding various collagens is responsible for tissue and vascular fibrosis. Visceral involvement with fibrosis, microvascular alterations, and mononuclear cell infiltration of the gastrointestinal tract, lungs, heart, and kidneys is present to a variable extent in most patients. Alterations in the production of cytokines and growth factors, which are capable of modulating the function of fibroblasts and other target cells, appear to play a relevant role. Transforming growth factor- is one of the growth factors intimately involved in the fibrosis that accompanies systemic sclerosis. S ystemic sclerosis is a disease of unknown origin characterized by excessive deposition of collagen and other connective tissue macromolecules in skin and multiple internal organs, prominent and often severe alterations in the microvasculature, and humoral and cellular immunologic abnormalities (see Glossary). Systemic sclerosis is a complex and heterogeneous disease. Clinical forms range from limited skin involvement with minimal systemic alterations (limited cutaneous systemic sclerosis) to forms with diffuse skin sclerosis and severe internal organ disease (diffuse cutaneous systemic sclerosis) (1), and occasionally a fulminant course (fulminant systemic sclerosis) (2). The most apparent and almost universal clinical features of systemic sclerosis are related to the progressive Ann Intern Med. 2004;140:37-50. For author affiliations, see end of text. For definition of terms used, see Glossary. © 2004 American College of Physicians 37 Review Pathogenesis of Systemic Sclerosis Glossary Allele: One of 2 or more different genes containing specific inheritable characteristics that occupy corresponding positions (loci) on paired chromosomes. Allogenic: Having a different genetic constitution but belonging to the same species. Apoptosis: Disintegration of cells into membrane-bound particles that are then phagocytosed by other cells. Calcitonin gene-related peptide: A second product transcribed from the calcitonin gene. It is found in a number of tissues, including the nervous system. It can act as a vasodilator. Cellular immunity: Involves the production of lymphocytes by the thymus (T cells) in response to exposure to an antigen. Codon: A sequence of 3 nucleotides in a strand of DNA that provides the genetic code for a specific amino acid. COL1A1: Gene encoding the ␣1 chain of type I collagen. COL1A2: Gene encoding the ␣2 chain of type I collagen. Connective tissue growth factor (CTGF): Growth factor that plays a critical role in tissue fibrosis as well as angiogenesis, axial development of the musculoskeletal system, structural organization of connective tissue, and embryo implantation. Cytokines: Intercellular messenger proteins. Hormone-like products of many different cell types that are usually active within a small radius of the cells producing them. Dinucleotide: The product of cleaving a polynucleotide (nucleic acid composed of ⱖ2 nucleotides). Epitope: Any component of an antigen molecule that functions as an antigenic determinant by permitting the recognition and attachment of specific antibodies. Extracellular matrix: The ground substance occupying the space outside cells in a specific tissue. Exon: A portion of the DNA that is transcribed to RNA, which is then expressed. Fibroblasts: Mesenchymal cells responsible for the production of fibrous tissue. Fibronectin: A cell-binding glycoprotein. Growth factors: Polypeptides produced by various cells of the body that promote growth and development by directing cell division, maturation, and differentiation and by mediating maintenance and repair of tissues. Haplotypes: The genetic constitution of an individual with respect to 1 member of a pair of allele genes. HLAs: Human leukocyte antigens. The cell surface molecules encoded by genes in the MHC on chromosome 6. These molecules are divided into 2 classes (class I and class II) on the basis of their structure. Class I and II molecules attach and present antigens to CD8⫹ (cytotoxic and suppressor cells) T cells and CD4⫹ (helper) T cells, respectively. Humoral immunity: Immunologic system involved in the production of plasma lymphocytes (B cells) in response to antigen exposure with subsequent antibody production. Intercellular adhesion molecule-1 (ICAM-1): A glycoprotein that is expressed on activated endothelial cells because of stimulation by local production of cytokines, which in turn causes adherence of leukocytes. Interferons: A class of small glycoprotein cytokines produced by T cells, fibroblasts, and other cells in response to viral infection and other biological and synthetic stimuli. Their effects include inducing enzymes, suppressing cell proliferation, inhibiting viral proliferation, enhancing the phagocytic activity of macrophages, and augmenting the cytotoxic activity of T lymphocytes. Interleukins: A large family of hormone-like messenger proteins produced by immune cells that act on leukocytes and other cells. Intron: A portion of DNA that lies between two exons, is transcribed into RNA, but does not appear in mature messenger RNA because the intron is removed and the exons are spliced together, and so it is not expressed. Lymphokine: A hormone-like intercellular messenger protein produced by lymphocytes. Matrix metalloproteinases: A family of protein-hydrolyzing endopeptidases that hydrolyze extracellular proteins, especially collagens and elastin. MHC: Major histocompatibility complex. The pair of genes on human chromosome 6 that encode the cell surface molecules known as HLAs. Nucleotide: A combination of a nucleic acid (purine or pyrimidine), 1 sugar (ribose or deoxyribose), and a phosphoric group. Phenotype: The observable characteristics at the physical, morphologic, or biochemical level of an individual, which are determined by the genetic sequence of the individual. Platelet-derived growth factor (PDGF): A factor present in platelets that is mitogenic for cells at the site of a wound, causing proliferation of endothelial cells, fibroblasts, smooth-muscle cells, and glial cells. Polymerase: Any enzyme catalyzing a polymerization, as of nucleotides to polynucleotides. Polymorphism: The presence of alleles in a population at a frequency that is higher than expected and that cannot be explained by mutation, suggesting a positive selection mechanism. Prostacyclin: A prostanoid produced by endothelial cells that is a potent natural inhibitor of platelet aggregation and a powerful vasodilator. Protease: Descriptive term for proteolytic enzymes, both endopeptidases and exopeptidases, which hydrolize polypeptide chains. Sequence homology: The degree of similarity between the nucleotide sequences of strands of DNA. Smad: A family of messenger/transcription factor proteins that are involved in signal transduction by transforming growth factor-. Topoisomerases: A group of enzymes capable of converting 1 topologic version of DNA into another. They act by catalyzing the breakage and reformation of DNA phosphodiester linkages. Transcript: Same as messenger RNA. Transcription: Transfer of genetic code information from one kind of nucleic acid to another. Commonly used to refer to transfer of genetic information from DNA to RNA. Transcription factors: Protein factors that interact with promoters and upstream regulatory elements on the DNA of the protein that will be expressed. Transduction: Transfer of genetic material from one cell to another. Also, transmission of cell surface signals into the nucleus. Transforming growth factor- (TGF-): A regulatory cytokine that has multifunctional properties and can enhance or inhibit many cellular functions, including interfering with the production of other cytokines and enhancing collagen deposition. Tumor necrosis factor (TNF): A polypeptide cytokine produced by endotoxin-activated macrophages that has the ability to modulate adipocyte metabolism, lyse tumor cells in vitro, and induce hemorrhagic necrosis of certain transplantable tumors in vivo. 38 6 January 2004 Annals of Internal Medicine Volume 140 • Number 1 www.annals.org Pathogenesis of Systemic Sclerosis thickening and fibrosis of the skin (3). However, multiple internal organs are always involved in some degree even when it is not clinically apparent. The affected skin is tight, indurated, and firmly bound to the subcutaneous tissue. Hair follicles and sweat and sebaceous glands atrophy. The skin over the hands and face is most frequently involved; as the disease progresses, the sclerotic changes extend and may affect the entire body. In many cases, the cutaneous involvement is confined to the digits and the dorsum of the hands and feet (acrosclerosis), and progression of the sclerotic process is relatively slow. This form of disease was previously known as the CREST syndrome (Calcinosis, long-standing Raynaud phenomenon, Esophageal dysmotility, Sclerodactyly, and Telangiectases) (4). Skin ulcerations, usually localized to fingertips or knuckles, and peculiar pigmentary changes are frequent. Calcinosis is most commonly found in fingertips and periarticular tissues. The Raynaud phenomenon is the second most common manifestation of systemic sclerosis and is present in more than 85% of patients (5). It usually appears simultaneously with other manifestations but may antedate them by several years. Often, the initial vasoconstriction and blanching are followed by a dusky cyanosis and are accompanied by paresthesias and numbness. With return of blood flow, reactive erythema occurs. In some cases, larger blood vessels are affected and luminal narrowing and occlusion can result in ischemic necrosis. Musculoskeletal symptoms are often the initial manifestations and may vary from mild polyarthralgias to frank arthritis (6). Synovitis with a moderate effusion occurs occasionally; however, the synovial fluid is only mildly inflammatory. In more advanced stages, thickening and fibrosis of periarticular tissues result in severe flexion contractures and distal phalangeal resorption. Muscle involvement can reveal either an inflammatory myopathy or a more indolent noninflammatory form due to muscle infiltration with fibrotic tissue. The gastrointestinal tract is the most common internal organ system involved (7). Esophageal symptoms resulting from reduced lower esophageal sphincter pressure and dysmotility of the lower two thirds of the esophagus include reflux, heartburn, and dysphagia to solid foods. In severe cases, chronic esophagitis can lead to stricture and odynophagia. Poor gastric emptying and small intestine involvement may cause abdominal distention, bloating, nausea, and pain, and, in many instances, bacterial overgrowth with secondary malabsorption, diarrhea, and weight loss. Pulmonary involvement has emerged as potentially the most serious visceral lesion in systemic sclerosis. Because no reliable ameliorating treatment is available for such involvement, it frequently leads to severe respiratory disability and often death (8). The most prominent symptoms are tachypnea and exertional dyspnea, secondary to either pulmonary fibrosis or pulmonary hypertension. Many patients remain asymptomatic despite evidence of fibrotic involvewww.annals.org Review ment of the lung parenchyma. In some cases, fulminant pulmonary hypertension may lead to rapid death (9). Cardiac involvement is not uncommon (9, 10). Chest pain, arrhythmias, various degrees of heart block, and myocardiopathy with left ventricular or biventricular failure may result from myocardial fibrosis. These alterations occur almost exclusively in patients with diffuse systemic sclerosis. Cor pulmonale can develop in patients with pulmonary hypertension. Pericardial involvement is usually asymptomatic; however, pericardial effusions are found by echocardiography or autopsy examination in approximately one half of patients. Until recently, renal disease was the most rapidly fatal visceral involvement of systemic sclerosis. It is known as scleroderma renal crisis and is typically characterized by the abrupt onset of malignant hypertension and rapidly progressive renal insufficiency (11). It is often heralded by severe headache; visual symptoms from hypertensive retinopathy; seizures and other central nervous system symptoms; or myocardial ischemia, infarction, or left ventricular failure. Prompt aggressive treatment now can usually reverse this process, which otherwise is fatal. Functional thyroid abnormalities, including elevated levels of antithyroid autoantibodies and clinical and subclinical hypothyroidism, are common (12). Impotence caused by erectile failure may be an early feature of systemic sclerosis (13), and some degree of erectile dysfunction ultimately develops in many affected patients. Patients may develop the sicca syndrome (keratoconjunctivitis sicca and xerostomia) caused by fibrosis and lymphocytic infiltration of the salivary and lacrimal glands (14). Unusual clinical manifestations include urinary bladder involvement, pneumatosis cystoides intestinalis with pneumoperitoneum, trigeminal neuralgia, and hoarseness from vocal chord involvement. The pathologic changes in systemic sclerosis encompass a spectrum reflecting variable stages of development and progression of 3 major processes in the affected tissues: 1) severe tissue fibrosis with exaggerated deposition of collagen and other connective tissue components in the extracellular matrix (see Glossary); 2) chronic inflammation, occurring predominantly in the early stages of disease and characterized by infiltration with mononuclear cells, mostly of the macrophage and T-cell lineages; and 3) microvascular disease, characterized by intimal proliferation, concentric subendothelial deposition of collagen and mucinous material, and narrowing and thrombosis of the vessel lumen. Progression of the vascular and fibrotic changes and a decrease in the inflammatory component lead to end-stage fibrosis and atrophy of the affected organs. The histopathologic findings in the skin include marked thickening of the dermis with massive accumulation of dense collagen, causing epidermal atrophy, flattening of the rete pegs, and replacement of sebaceous and sweat glands as well as hair follicles. A prominent inflammatory infiltrate is often present at the dermal–adipose 6 January 2004 Annals of Internal Medicine Volume 140 • Number 1 39 Review Pathogenesis of Systemic Sclerosis Figure 1. General overview of the pathogenesis of systemic sclerosis. The illustrations on the bottom row show examples of, from left to right, the fibrotic process (biopsy of skin), microvascular alterations in pulmonary arterioles, autoantibodies detected by immunofluorescence, and mononuclear inflammatory cell infiltrates in affected skin. tissue interphase, especially in early lesions (15, 16). The small vessels of the lower dermis show fibrous thickening but no evidence of vasculitis. In the lungs, the architecture of the alveolocapillary membrane and the parenchymal interstitium are markedly disrupted by fibrosis and severe mononuclear-cell infiltration. Prominent vascular abnormalities with intimal proliferation causing narrowing or complete obliteration of small vessels are frequent. The renal lesions display severe narrowing and obliteration of the medium-size arterioles because of subintimal accumulation of loose connective tissue and intimal and perivascular fibrosis. The glomeruli frequently appear ischemic, and there is no evidence of glomerulitis. Severe interstitial, perivascular, and periglomerular fibrosis may be present in cases of long duration. Other affected organs display variable degrees of fibrosis, mononuclear-cell infiltration, and microvascular obliteration and fibrosis. PATHOGENESIS The pathogenesis of systemic sclerosis is extremely complex. At present, no single unifying hypothesis explains all aspects of its pathogenesis. However, fundamental abnormalities in at least 3 types of cells are intimately involved in the development of the clinical and pathologic manifestations of the disease: 1) fibroblasts (see Glossary); 2) endothelial cells; and 3) cells of the immune system, 40 6 January 2004 Annals of Internal Medicine Volume 140 • Number 1 particularly T and B lymphocytes. The profound functional alterations in these cells result in the characteristic triad of pathologic changes in systemic sclerosis: severe and often progressive cutaneous and visceral fibrosis; obliteration of the lumen of small arteries and arterioles; and humoral and cellular immunologic abnormalities, which include the production of numerous autoantibodies (some very highly specific for the disease), the chronic mononuclear-cell infiltration of affected tissues, and the dysregulation of lymphokine and growth factor production (see Glossary). These dysfunctional cellular processes and their resulting effects on the affected tissues are illustrated in Figure 1. At present, it is not clear which of these alterations is of primary importance or how they interrelate to cause the progressive fibrotic process in systemic sclerosis. However, a crucial component in the pathogenesis of the disorder is the persistent and unregulated activation of genes encoding various collagens and other extracellular matrix proteins in systemic sclerosis fibroblasts. This is the most important difference between normal fibroblasts, which promote normal wound healing, and systemic sclerosis fibroblasts, which demonstrate an uncontrolled production and tissue deposition of collagen resulting in pathologic organ fibrosis. This paper reviews some of the numerous components of the complex puzzle of systemic sclerosis pathogenesis, as illustrated in Figure 1, and atwww.annals.org Pathogenesis of Systemic Sclerosis tempts to form hypotheses that may provide frameworks for the development of novel and effective therapies. Because we were not able to provide a comprehensive discussion of the abundant literature published, we refer readers to recent reviews for a more complete and detailed description. Possible Causative Agents The cause of systemic sclerosis has remained elusive despite intense investigations. Although the disease is not inherited in a classical Mendelian pattern, there is strong evidence that genetic factors contribute to its development and clinical manifestations, as discussed in more detail shortly. However, it has become apparent that environmental agents play a crucial and more important role than genetic influences. One study reported a remarkably low concordance in the development of systemic sclerosis among homozygous twins, indicating that the heritability component of the disease was very low and that the most important factor was of an environmental or acquired origin (17). Many infectious, chemical, and physical agents have been postulated as being involved in the cause of the disease. The hypothesis that infectious agents may cause systemic sclerosis has been studied extensively. Some researchers have suggested that the production of specific autoantibodies in systemic sclerosis is the result of an antigen-driven response caused by “molecular mimicry.” The concept of “molecular mimicry” proposes that antibodies against self-antigens are produced because these antigens contain epitopes (see Glossary) that share structural similarities with viral or bacterial proteins. In the immunopathogenesis of systemic sclerosis, herpesviruses, retroviruses, and human cytomegalovirus infections, among others, have been suggested as possible causative agents. Evidence supporting the role of retroviruses includes the demonstration of sequence homologies (see Glossary) between certain retroviral proteins and the topoisomerase I antigen (see Glossary), which is the target of anti–Scl-70 antibodies in patients with systemic sclerosis (18). In addition, it has been shown that the induced expression of retroviral proteins in normal human dermal fibroblasts results in the acquisition of a systemic sclerosis–like phenotype (see Glossary) in the production of extracellular matrix proteins (18). Furthermore, antibodies to retroviral proteins have been detected in serum specimens from patients with systemic sclerosis (19). Another hypothesis has suggested that human cytomegalovirus may be involved in the initial events of systemic sclerosis. This hypothesis is supported by the observations of a higher prevalence of IgA antihuman cytomegalovirus antibodies in patients with systemic sclerosis, which are capable of inducing apoptosis (see Glossary) in human endothelial cells; the increased prevalence of anticytomegalovirus IgA antibodies in patients positive for Scl-70 autoantibodies; and the severe fibroproliferative vascular changes and the increased occurrence of antinuclear antibodies with an immunofluoreswww.annals.org Review cence pattern similar to that present in serum specimens from patients with systemic sclerosis in human cytomegalovirus infections (20, 21). Despite intensive study, however, there is no definitive evidence to conclude that systemic sclerosis has a viral origin. Environmental agents have also been implicated in the development of systemic sclerosis (22, 23). Silica and metal dust exposure had been shown in case studies to be related to systemic sclerosis (22–24), although some studies have failed to confirm an association. On the other hand, organic solvent exposure may eventually be proven to be an important environmental factor in triggering this disease. Indeed, persons exposed to vinyl chloride have an increased risk for skin thickening, the Raynaud phenomenon, and digital ulcers (23), and recent epidemiologic studies have found a higher frequency of organic solvent exposure in patients with systemic sclerosis than in normal controls (24). Several other environmental exposures have been associated with the development of systemic sclerosis, including certain pesticides, hair dyes, and fuel-derived or industrial fumes (22, 23). Role of Genetic Factors The contribution of genetic factors in the development and expression of systemic sclerosis is strongly supported by the observation of familial clustering of the disease, the high frequency of autoimmune disorders and autoantibodies in family members of patients with systemic sclerosis, differences in prevalence and clinical manifestations among different ethnic groups, and the increased prevalence of certain HLAs and MHC alleles (see Glossary) among different ethnic groups and among patients with different clinical subsets of the disease or with different patterns of autoantibodies (25). Strong evidence indicates that genetic factors largely determine the production of specific autoantibodies in systemic sclerosis. Although the concordance of systemic sclerosis among identical twins is only 4.2% and is not significantly different from the concordance of disease in dizygotic twins (5.9%), the concordance for the presence of specific autoantibodies is substantially higher (17). These observations indicate that inherited genetic factors are important for the production of autoantibodies but are not sufficient for development of disease. Prevalence of the disease varies by geographic region and by ethnic background; the prevalence in the United States is 242 to 286 cases per million persons in the population. However, in Native American persons of the Choctaw tribe in Oklahoma, one of the best-studied groups for the role of genetic factors in systemic sclerosis, the prevalence is 469 cases per million persons in the population (26). The differences of MHC and HLA allele expression are evident when comparing the different haplotypes (see Glossary) identified as being linked to disease expression, in particular to the pattern of autoantibody response, among ethnic groups (27). Disease expression 6 January 2004 Annals of Internal Medicine Volume 140 • Number 1 41 Review Pathogenesis of Systemic Sclerosis also appears to differ among ethnic groups. African-American persons are more likely to have anti-topoisomerase I antibodies and more severe visceral manifestations, including a higher frequency of pulmonary fibrosis. In contrast, anticentromere antibodies are more common in white persons, who are also more likely to have limited disease with less severe systemic manifestations (27). In addition to these genetic characteristics, which may predispose persons to systemic sclerosis, other genetic factors may influence the expression of the disease. Recent studies have examined mutations or polymorphisms (see Glossary) in relevant genes that may result in their increased expression. For example, in patients with systemic sclerosis, the upstream region of COL1A2 (see Glossary), the region of the gene preceding the initiation of gene transcription, harbors numerous dinucleotide repeats that may increase gene activity (see Glossary) (28). Similarly, 2 single nucleotide polymorphisms at codon 10 of the transforming growth factor (TGF)-1 gene were found in statistically significant higher frequency in patients with systemic sclerosis (see Glossary) (29). This suggests a genetic predisposition to higher production of TGF-1 (29). Furthermore, patients who had the TGF-1 polymorphism had a higher frequency of pulmonary fibrosis and graft fibrosis following lung transplantation (30). As discussed later in more detail, TGF-1 is a potent profibrotic cytokine (see Glossary) secreted from activated macrophages and T lymphocytes that may play a central role in the pathogenesis of tissue fibrosis in systemic sclerosis. Another genetic polymorphism that may influence expression of systemic sclerosis has been described in the gene encoding fibrillin-1, a large extracellular matrix protein that is a structural component of connective tissue microfibrils. The polymorphism is located in the upstream region (vide supra) of the gene, and its presence is strongly associated with the occurrence of systemic sclerosis in Native American persons from the Choctaw tribe and in Japanese persons (31). These collective studies on the influence of genetic polymorphisms on the development or expression of systemic sclerosis, however, must be performed in larger cohorts to corroborate their relevance. Microchimerism Microchimerism is a novel and provocative hypothesis of the cause of systemic sclerosis (32–34). The hypothesis suggests that, during pregnancy, allogenic (see Glossary) fetal or maternal cells cross the placenta in bidirectional traffic and persist in the circulation and tissues of the mother or child, respectively, as a result of HLA II (DRB1) compatibility between the mother and the fetus. These engrafted foreign cells may become activated by a second event and may mount a graft-versus-host reaction, which manifests as systemic sclerosis. The remarkable similarities in clinical, histopathologic, and serologic features between graft-versus-host disease and systemic sclerosis, including esophageal, lung, and skin involvement; lymphocytic infil42 6 January 2004 Annals of Internal Medicine Volume 140 • Number 1 tration and fibrosis of affected organs; and production of autoantibodies, strongly support this hypothesis. Researchers suggested this theory when fetal cells were found in the circulation of normal women several decades after the birth of their children and fetal DNA and fetal cells were subsequently identified in affected skin samples from women with systemic sclerosis who had previously been pregnant with a male fetus (32). Microchimeric cells of maternal origin have also been identified in the circulation of offspring (35). These findings might explain the occurrence of systemic sclerosis in nulliparous women and men. Although some studies found that the frequency of male chromosome sequences in the skin of women with systemic sclerosis who had male offspring was similar to that of normal controls, quantification of microchimeric fetal cells showed a significant difference in the amount of male DNA in affected skin between the 2 groups (36). This indicates that it is the quantity and not only the mere presence of fetal cells that might contribute to the pathogenesis of systemic sclerosis. A more recent study confirmed the quantitative difference in the number of microchimeric cells between patients with systemic sclerosis and healthy individuals and also showed that immunologic stimulation expected to trigger antigen-specific T cells caused a potent amplification of the microchimeric cells in patients with systemic sclerosis but not in controls (37). Despite these observations, at present, the role of microchimerism in the pathogenesis of systemic sclerosis remains undetermined. Humoral Immune System Alterations The presence of specific autoantibodies is one of the most common manifestations of systemic sclerosis. More than 90% of patients with systemic sclerosis harbor antinuclear antibodies in their serum. Numerous autoantibodies, some of which are extremely specific for systemic sclerosis, have been described in patients with the disease; other autoantibodies are associated with different clinical manifestations (38). Anti–Scl-70 antibodies have been shown to react with DNA topoisomerase I and are almost exclusively present in serum specimens from patients with the diffuse form of systemic sclerosis, although only about 30% to 40% of these patients harbor these autoantibodies. Anticentromere antibodies are present in 80% to 90% of patients with the limited form of systemic sclerosis but are found in fewer than 10% of patients with diffuse systemic sclerosis. These 2 autoantibodies rarely coexist in the same patient. Other autoantibodies are less common in patients with systemic sclerosis. They include anti-RNA polymerases I and III antibodies in patients with rapidly progressive disease and severe internal organ involvement; antifibrillarin antibodies, which are commonly found in diffuse systemic sclerosis; and anti–PM-Scl antibodies, which are usually found in patients with systemic sclerosis who develop an inflammatory myopathy. Although autoantibodies are www.annals.org Pathogenesis of Systemic Sclerosis common in systemic sclerosis, they are not directly involved in the clinical manifestations of the disease. However, owing to their high frequency and their specificity for certain clinical disease subsets, their presence is very helpful in establishing the diagnosis and predicting a probable pattern of organ involvement, severity, and disease progression (38). Tissue Inflammation: An Early Event in the Cascade? Some of the earliest evidence suggesting that chronic and persistent inflammation may play a role in the pathogenesis of systemic sclerosis was provided by the demonstration of lymphocytic infiltrates in affected skin from patients with disease of recent onset (15, 16). Subsequently, researchers found that the extent of lymphocytic infiltration correlated with the severity and the progression of skin sclerosis (39). The mononuclear cells within the skin infiltrates are predominantly CD4⫹ T cells and express the activation marker class II MHC antigen DR (40, 41). Subsequent expansion of these cells within the tissue appears to be oligoclonal, as shown in studies of T-cell receptor transcripts (see Glossary) in the skin of patients with systemic sclerosis (42). These results suggest an antigen-driven response, although at present there is no information on the putative antigen or antigens that may be involved. The expanded populations of inflammatory cells in affected tissues release cytokines and growth factors that initiate or perpetuate the fibrotic process as well as the endothelial and vascular alterations. Indeed, clones of T cells established from infiltrating lymphocytes isolated from affected skin of patients with systemic sclerosis produce cytokines that can stimulate fibroblast collagen production (43). The mechanisms responsible for the migration of distinct populations of T cells from peripheral blood to skin and other affected tissues in patients with systemic sclerosis are not completely understood. However, it is likely that their retention and accumulation in such tissues result from specific interactions between certain T-cell subsets and fibroblast membrane proteins or extracellular matrix, which are mediated by integrins and cell adhesion molecules such as the adhesive protein intercellular adhesion molecule-1 (see Glossary) (44). The inflammatory changes in affected tissues are also reflected in quantitative and functional alterations in peripheral blood cells (45). For example, the proportions and absolute numbers of total CD4⫹ CD45RA⫹ (suppressorinducer) T cells and CD8⫹/CDIIb⫹ suppressor T cells are decreased. Thus, the balance between immunoregulatory T-cell populations appears to be substantially impaired. Peripheral blood T cells in systemic sclerosis manifest evidence of previous activation, indicated by the spontaneous expression of high affinity interleukin-2 receptor (IL-2R) on their membranes (see Glossary). Several observations further support the evidence for in vivo T-cell activation in systemic sclerosis. First, serum specimens from patients with systemic sclerosis contain approximately 3-fold higher www.annals.org Review levels of soluble IL-2R than specimens from normal controls. In addition, mononuclear cells obtained from bronchoalveolar lavage fluid in patients with systemic sclerosis display ongoing activation. Finally, elevated levels of serum interleukin-2 and increased production of this cytokine by T cells in vitro have also been demonstrated in patients with systemic sclerosis. Abnormalities in numerous other cytokines have also been described (45). However, whether the alterations in the proportions of regulatory T-cell subsets and the various cytokines are secondary to disease activity or reflect a more fundamental pathogenic event is unknown. Role of Transforming Growth Factor-, Connective Tissue Growth Factor, and Smad Proteins At present, it is unknown whether the exaggerated connective tissue production by systemic sclerosis fibroblasts is a response to an unknown injury, thus representing an abnormal regulation of a physiologic process, or whether the primary event is an alteration in the regulation of expression of the relevant matrix protein genes. It has been postulated that alterations in the regulation of extracellular matrix gene expression may be induced by cytokines and growth factors released from the tissue-infiltrating inflammatory cells. Numerous alterations in the expression of cytokines and growth factors with potent effects on fibroblast collagen synthesis, endothelial cell functions, and T-cell responses have been demonstrated in patients with systemic sclerosis (45). Many studies have examined the effects of various products of inflammatory cells, including TGF-, connective tissue growth factor, platelet-derived growth factor, interleukin-1, interleukin-2, interleukin-4, interferons, and tumor necrosis factor, on many different aspects of fibroblast biology (see Glossary) (46). These proteins exert stimulatory or inhibitory effects on fibroblast proliferation and collagen production and sometimes exert bifunctional effects, depending on their concentrations and on the context of the other cytokines and growth factors present simultaneously in the pericellular environment. One of the growth factors that appears to play a crucial role in the fibrosis that accompanies systemic sclerosis is TGF-. The 3 functionally and structurally similar human isoforms of TGF- play important roles in embryonic development, in immune responses, and in the regulation of tissue repair following injury (47). One of the most important effects of TGF- is the stimulation of extracellular matrix synthesis by stimulating the production of various collagens and other matrix proteins, including fibronectin (see Glossary) (48). Small amounts of TGF- appear to sensitize fibroblasts and maintain them in a persistently activated state involving an autocrine signaling mechanism that causes further production of TGF-. Systemic sclerosis fibroblasts also seem to express increased levels of TGF- receptors on their surface. This might account for the increased TGF-–induced signaling and the resulting 6 January 2004 Annals of Internal Medicine Volume 140 • Number 1 43 Review Pathogenesis of Systemic Sclerosis Figure 2. Schematic diagram of the transforming growth factor- (TGF-) and Smad pathways involved in stimulation of collagen gene expression. R ⫽ receptor. increased collagen production by these fibroblasts (49). In addition to its stimulatory effects on extracellular matrix synthesis, TGF- also decreases the production of collagen-degrading metalloproteinases and stimulates the production of protease (see Glossary) inhibitors, such as tissue inhibitor of metalloproteinases-1, which prevent breakdown of the extracellular matrix (46). Recent studies have provided a clearer picture of the intricate pathways that mediate the stimulation of collagen gene expression by TGF- (50, 51). These include the binding of TGF- to specific cell surface receptors and the transduction (see Glossary) of the binding signal into the nucleus to influence the activity and expression of TGF–responsive genes, such as those encoding extracellular matrix proteins. Briefly, TGF- is secreted from activated lymphocytes or monocytes in an inactive form and requires complex events to become activated. Once active TGF- comes into contact with a given target cell, it binds to a specific TGF-II receptor on the target cell surface, which in turn recruits and phosphorylates a TGF-I receptor to form an active receptor complex. Subsequent signaling to the nucleus then occurs through the Smad family of proteins (see Glossary) (50, 51). Smad 2 or Smad 3 binds to the TGF- receptor complex and then becomes phosphorylated and able to form a complex with Smad 4, a cytoplasmic protein involved in the translocation of the Smad complex into the nucleus. Smad 7 is an inhibitory Smad that can bind to the TGF- receptor complex and prevent 44 6 January 2004 Annals of Internal Medicine Volume 140 • Number 1 Smad 2 or Smad 3 phosphorylation. It appears that both interferon-␥ and tumor necrosis factor-␣, 2 potent collagen synthesis inhibitory cytokines, stimulate increased expression of Smad 7. This results in decreased signaling of TGF-–induced collagen gene expression. Following nuclear translocation, the stimulatory Smad complexes bind to specific promoter sites in target genes and activate their expression. The pathways involved in the activation of collagen genes following TGF- binding to target cell surface receptors are shown in Figure 2. The potential involvement of Smad proteins in the pathogenesis of systemic sclerosis has been reviewed elsewhere (52). Furthermore, a recent study described substantially reduced levels of Smad 7 in systemic sclerosis (53). The reduction of Smad 7 inhibitory effects may be responsible for an exaggerated and unregulated TGF- signaling cascade resulting in excessive extracellular matrix tissue accumulation. Connective tissue growth factor also appears to play a crucial role in fibrosis (54). It participates in angiogenesis, axial development of the musculoskeletal system, structural organization of connective tissues, and embryo implantation. Transforming growth factor- causes potent stimulation of connective tissue growth factor synthesis in fibroblasts, vascular smooth-muscle cells, and endothelial cells. Connective tissue growth factor also seems to be involved in an autocrine loop stimulating its own production and, thus, maintaining a continuous or prolonged cycle of excessive fibrosis. Although studies on the role of connective www.annals.org Pathogenesis of Systemic Sclerosis tissue growth factor in the pathogenesis of fibrotic diseases are just emerging, it is likely that this growth factor may become recognized as an important mediator in the tissue accumulation of extracellular matrix in systemic sclerosis. Tissue and Vascular Fibrosis: The Final Step in the Pathogenic Pathway The most prominent clinical manifestations of systemic sclerosis are caused by the exaggerated accumulation of collagen and other connective tissue components in the affected organs. The excessive collagen deposition in systemic sclerosis is due to overproduction of this protein by fibroblasts. Regardless of the etiologic event, the resulting alterations in the biosynthetic phenotype of collagen-producing cells are crucial in the pathogenesis of systemic sclerosis. Indeed, it is the persistent activation of the genes encoding various collagens in systemic sclerosis fibroblasts that distinguishes controlled repair, such as that occurring during normal wound healing, from the uncontrolled fibrosis that is the hallmark of systemic sclerosis. Despite recent advances in the understanding of the regulation of collagen gene expression under normal conditions or under the effects of various cytokines and growth factors, the intimate mechanisms responsible for the pathologic increase in the expression of collagen genes in systemic sclerosis remain obscure. The exaggerated production of extracellular matrix macromolecules by systemic sclerosis fibroblasts largely results from increased transcription rates of their corresponding genes (55, 56). The most important molecule in the fibrotic processes of systemic sclerosis is type I collagen, the prototype of the interstitial collagens, which is largely responsible for the functional failure of the affected organs. Normal fibroblasts are capable of regulating collagen production according to the requirements of the situation during development, differentiation, and repair. As for most eukaryotic genes, the regulation of transcription rates of collagen genes involves precise interactions between specific nucleotide sequences present in the promoters and, often, in their first introns (see Glossary) and various transcription factors (see Glossary) capable of recognizing and binding specifically to these sequences. The entire complement of regulatory elements in collagen genes is not yet known, and the number of identified transcription factors that interact with these elements continues to increase. Numerous transcription factors capable of regulating the expression of collagen genes have been identified (57). One of the most extensively studied transcription factors is Sp1. Sp1 recognizes a specific sequence in the promoters of various Sp1-responsive genes. Sp1 appears to play a pivotal function in the increased expression of COL1A1 (see Glossary) in systemic sclerosis. Increased Sp1 binding activity has been demonstrated in systemic sclerosis fibroblast nuclear extracts in comparison with nuclear extracts from normal cells (58), and increased Sp1 phosphorylation associated with increased expression of www.annals.org Review type I collagen genes has also been found in these cells (59). Another study showed that activated hepatic stellate cells, which are the primary cells responsible for increased type I collagen production during liver fibrosis, also display increased Sp1 binding activity (60). Binding of Sp1 to its recognition sites has also been implicated in the regulation of expression of type I collagen genes under the influence of TGF-. Furthermore, numerous studies have shown that specific inhibition of Sp1 binding to its cognate elements within collagen genes caused potent and selective inhibition of collagen production in normal fibroblasts and fibroblasts cultured from patients with systemic sclerosis (61, 62). Another transcription factor that appears to play an important role in collagen gene regulation is the CCAATbinding factor (CBF). Both CBF and Sp1 transcription factors are involved in a protein–protein interaction in the collagen gene promoter, and both appear to be crucial regulatory factors in the expression of collagen genes. The role of CBF in the regulation of collagen gene expression in systemic sclerosis has also been examined, and it has been shown that CBF binding activity is increased in systemic sclerosis fibroblasts (63). In contrast with the stimulatory effects of Sp1 and CBF, the transcription factor c-Krox mediates tissue-specific inhibition of collagen gene expression (64), although its role in pathologic fibrosis has not been investigated. Endothelial Cells Vascular dysfunction is one of the earliest alterations of systemic sclerosis, and it has been suggested that it may represent the initiating event in its pathogenesis (65). Severe alterations in small blood vessels of skin and internal organs, including fibrosis and perivascular cellular infiltration with activated T cells, are almost always present in systemic sclerosis. Cytokines such as TGF- are secreted by these activated lymphocytes and in turn injure the endothelial cells, inducing their expression of MHC class I and II antigens and adhesion ligand intercellular adhesion molecule-1. Transforming growth factor- causes an upregulation of connective tissue growth factor, which also results in increased production of extracellular matrix components as well as an upregulation of platelet-derived growth factor. Elevated expression of platelet-derived growth factor causes increased endothelial cell proliferation and a downregulation of vascular endothelial growth factor, the endogenous growth factor that promotes neovascularization. Chemoattraction of fibroblasts into the vessel wall also occurs as a result of the effects of the cytokines released locally, leading to increased collagen synthesis and deposition in the vessel wall. Transforming growth factor- may also induce transdifferentiation of vessel wall fibroblasts into myofibroblasts, which are phenotypically distinct cells with contractile properties and increased extracellular matrix biosynthetic ability. The endothelial injury initiated by the release of cyto6 January 2004 Annals of Internal Medicine Volume 140 • Number 1 45 Review Pathogenesis of Systemic Sclerosis Figure 3. Diagram of the pathogenesis of systemic sclerosis according to the hypothesis of microchimerism. CTGF ⫽ connective tissue growth factor; IL ⫽ interleukin; NO ⫽ nitric oxide; PDGF ⫽ platelet-derived growth factor; TGF- ⫽ transforming growth factor-; TNF-␣ ⫽ tumor necrosis factor-␣. kines causes the subendothelium to become exposed to circulating platelets, which eventually adhere to it and initiate fibrin deposition and intravascular thrombus formation. It has also been suggested that endothelial cell damage in systemic sclerosis may be due to a direct effect of cytotoxic factors for endothelium, since serum specimens from patients with systemic sclerosis contain cytotoxic activity directed against endothelial cells (66). Other proposed mechanisms of endothelial cell injury in systemic sclerosis are those mediated by proteolytic activities present in serum (67) or by serum IgG antibodies that result in antibody-dependent cell-mediated cytotoxicity (68). These antibodies occasionally coexist with Clq-binding immune complexes and anticardiolipin antibodies, both of which are associated with thrombosis. Vasodilation, which is controlled by both endothelial and nonendothelial (neural) mechanisms, also appears to be impaired in patients with systemic sclerosis. Endothelial cells normally secrete vasoactive substances that control 46 6 January 2004 Annals of Internal Medicine Volume 140 • Number 1 vascular tone, such as the potent vasodilators prostacyclin, nitric oxide, and calcitonin gene–related peptide (see Glossary) (69). On the other hand, they also produce endothelin-1, a 21-amino acid polypeptide that has potent vasoconstrictor activities and stimulates extracellular matrix production and deposition in the vessel wall (70). Patients with systemic sclerosis appear to have a relative deficiency of vasodilators and a remarkable increase in levels of endothelin-1. This imbalance in turn causes further vascular hypoxia, which leads to increased collagen gene expression as well as endothelial injury, thus maintaining the vicious cycle of endothelial injury and fibrosis. DISCUSSION Although the exact mechanisms involved in the pathogenesis of systemic sclerosis are not known, there is sufficient evidence to allow cogent hypotheses about the possible pathway the disease takes from its causation to organ www.annals.org Pathogenesis of Systemic Sclerosis Review Figure 4. Diagram of the pathogenesis of systemic sclerosis according to the hypothesis of an environmental agent causing initial tissue inflammation. CTGF ⫽ connective tissue growth factor; HCMV ⫽ human cytomegalovirus; NO ⫽ nitric oxide; PDGF ⫽ platelet-derived growth factor; TGF ⫽ transforming growth factor-. fibrosis and resulting clinical manifestations. At present, there is no single unifying concept but rather several possible pathways, as shown in Figures 3, 4, and 5. The novel hypothesis of a graft-versus-host reaction secondary to engrafted microchimeric cells is illustrated in Figure 3. In this pathway, it is necessary to postulate an environmental agent (for example, physical, chemical, or infectious) acting as a required second event that will trigger this reaction. In contrast, in Figure 4, it is hypothesized that the environmental agent or agents is the inciting factor that acts on a genetically predisposed host and results in the subsequent recruitment and homing of macrophages and T cells to the affected tissues. The inflammatory cells would undergo sewww.annals.org lective proliferation and expansion, perhaps because of an antigen-driven response, and then release cytokines and growth factors that initiate the process of tissue and vascular fibrosis. Finally, in Figure 5, it is postulated that the environmental factor or factors, most likely of an infectious origin acting in a genetically susceptible host, cause a profound phenotypic change in various target cells of different lineages (immune cells, fibroblasts, and endothelial and vascular smooth-muscle cells). This phenotypic change could be caused by integration of genetic material (for example, of retroviral origin) within the genetic sequence of the target cells that through unknown mechanisms would induce the expression of specific regulatory genes, altering 6 January 2004 Annals of Internal Medicine Volume 140 • Number 1 47 Review Pathogenesis of Systemic Sclerosis Figure 5. Diagram of the pathogenesis of systemic sclerosis according to the hypothesis of phenotypic change in target cells. CTGF ⫽ connective tissue growth factor; HCMV ⫽ human cytomegalovirus; IFN ⫽ interferon-␥; IL ⫽ interleukin; NO ⫽ nitric oxide; PDGF ⫽ platelet-derived growth factor; TGF- ⫽ transforming growth factor-; TNF-␣ ⫽ tumor necrosis factor-␣. the function and behavior of the target cells. These alterations are manifested by increased collagen and extracellular matrix production in fibroblasts, generation of autoantibodies and cellular immune abnormalities in lymphocytes, and severe fibroproliferative and prothrombotic alterations in endothelial cells. The target cell effects of cytokines and growth factors, particularly TGF- and connective tissue growth factor, and of the various components of their regulatory cascades are downstream components common to all 3 pathways. All 3 hypotheses, and other plausible ones that can be construed on the basis of currently known molecular abnormalities in systemic sclerosis, may prove to be correct, since the disease exhibits a remarkably heterogeneous spec48 6 January 2004 Annals of Internal Medicine Volume 140 • Number 1 trum of clinical manifestations and its pathogenesis may be different in different individuals. Although the ultimate goal of research studies about the pathogenesis of a disease is to find a unifying mechanism, in the case of systemic sclerosis, a single common pathogenetic mechanism may not be the correct answer. In the past 30 years, the study of systemic sclerosis has evolved from the concept that the disease was purely due to collagen overproduction by affected fibroblasts to the current stage, in which it is possible to postulate various distinct and extremely complex processes during the initial phases. Regardless of whether the answer to the puzzle of systemic sclerosis pathogenesis is a single, unified mechanism or a constellation of different ones, the knowledge www.annals.org Pathogenesis of Systemic Sclerosis acquired in this search will undoubtedly uncover novel targets, including those involving regulatory transcription factors for collagen gene expression, such as Sp1; intracellular mediators, such as Smad proteins; and TGF-⫺signaling cascades. These targets may lead to the development of long-awaited and potentially effective therapeutic options for this incurable disease. From Thomas Jefferson University, Philadelphia, Pennsylvania. Acknowledgments: The authors thank Kate Salmon for expert assis- tance in the preparation of the manuscript and M. Sonsoles PieraVelazquez, PhD, for assistance in the preparation of some of the illustrations. Grant Support: By National Institutes of Health Grant AR19616. Dr. Derk was supported by National Institutes of Health Training Grant AR07583. Potential Financial Conflicts of Interest: None disclosed. Requests for Single Reprints: Sergio A. Jimenez, MD, Division of Rheumatology, Thomas Jefferson University, 233 South 10th Street, Room 509 BLSB, Philadelphia, PA 19107-5541. Current author addresses are available at www.annals.org. References 1. LeRoy EC, Black C, Fleischmajer R, Jablonska S, Krieg T, Medsger TA Jr, et al. Scleroderma (systemic sclerosis): classification, subsets and pathogenesis. J Rheumatol. 1988;15:202-5. [PMID: 3361530] 2. Lally EV, Jimenez SA, Kaplan SR. Progressive systemic sclerosis: mode of presentation, rapidly progressive disease course, and mortality based on an analysis of 91 patients. Semin Arthritis Rheum. 1988;18:1-13. [PMID: 3187542] 3. Rodnan GP. Progressive systemic sclerosis: clinical features and pathogenesis of cutaneous involvement (scleroderma). Clin Rheum Dis. 1979;5:49-79. 4. Fritzler MJ, Kinsella TD. The CREST syndrome: a distinct serologic entity with anticentromere antibodies. Am J Med. 1980;69:520-6. [PMID: 6968511] 5. Block JA, Sequeira W. Raynaud’s phenomenon. Lancet. 2001;357:2042-8. [PMID: 11438158] 6. Pope JE. Musculoskeletal involvement in scleroderma. Rheum Dis Clin North Am. 2003;29:391-408. [PMID: 12841301] 7. Rose S, Young MA, Reynolds JC. Gastrointestinal manifestations of scleroderma. Gastroenterol Clin North Am. 1998;27:563-94. [PMID: 9891698] 8. Co HT, Block JA, Sequeira W. Scleroderma lung: pathogenesis, evaluation, and current therapy. Am J Ther. 2000;7:321-4. [PMID: 11317180] 9. Coghlan JG, Mukerjee D. The heart and pulmonary vasculature in scleroderma: clinical features and pathobiology. Curr Opin Rheumatol. 2001;13:495-9. [PMID: 11698727] 10. Deswal A, Follansbee WP. Cardiac involvement in scleroderma. Rheum Dis Clin North Am. 1996;22:841-60. [PMID: 8923599] 11. Steen VD. Scleroderma renal crisis. Rheum Dis Clin North Am. 2003;29: 315-33. [PMID: 12841297] 12. Gordon MB, Klein I, Dekker A, Rodnan GP, Medsger TA Jr. Thyroid disease in progressive systemic sclerosis: increased frequency of glandular fibrosis and hypothyroidism. Ann Intern Med. 1981;95:431-5. [PMID: 7283293] 13. Lally EV, Jimenez SA. Impotence in progressively systemic sclerosis. Ann Intern Med. 1981;95:150-3. [PMID: 6789736] 14. Cipoletti JF, Buckingham RB, Barnes EL, Peel RL, Mahmood K, Cignetti FE, et al. Sjogren’s syndrome in progressive systemic sclerosis. Ann Intern Med. 1977;87:535-41. [PMID: 921080] 15. Fleischmajer R, Perlish JS, Reeves JR. Cellular infiltrates in scleroderma skin. www.annals.org Review Arthritis Rheum. 1977;20:975-84. [PMID: 861067] 16. Jimenez SA. Cellular immune dysfunction and the pathogenesis of scleroderma. Semin Arthritis Rheum. 1983;13:104-13. [PMID: 6353581] 17. Feghali-Bostwick C, Medsger TA Jr, Wright TM. Analysis of systemic sclerosis in twins reveals low concordance for disease and high concordance for the presence of antinuclear antibodies. Arthritis Rheum. 2003;48:1956-63. [PMID: 12847690] 18. Jimenez SA, Diaz A, Khalili K. Retroviruses and the pathogenesis of systemic sclerosis. Int Rev Immunol. 1995;12:159-75. [PMID: 7650419] 19. Dang H, Dauphinee MJ, Talal N, Garry RF, Seibold JR, Medsger TA Jr, et al. Serum antibody to retroviral gag proteins in systemic sclerosis. Arthritis Rheum. 1991;34:1336-7. [PMID: 1930323] 20. Pandey JP, LeRoy EC. Human cytomegalovirus and the vasculopathies of autoimmune diseases (especially scleroderma), allograft rejection, and coronary restenosis. Arthritis Rheum. 1998;41:10-5. [PMID: 9433864] 21. Neidhart M, Kuchen S, Distler O, Bruhlmann P, Michel BA, Gay RE, et al. Increased serum levels of antibodies against human cytomegalovirus and prevalence of autoantibodies in systemic sclerosis. Arthritis Rheum. 1999;42:389-92. [PMID: 10025936] 22. Mayes MD. Epidemiologic studies of environmental agents and systemic autoimmune diseases. Environ Health Perspect. 1999;107 Suppl 5:743-8. [PMID: 10502540] 23. Diot E, Lesire V, Guilmot JL, Metzger MD, Pilore R, Rogier S, et al. Systemic sclerosis and occupational risk factors: a case-control study. Occup Environ Med. 2002;59:545-9. [PMID: 12151611] 24. Garabrant DH, Lacey JV Jr, Laing TJ, Gillespie BW, Mayes MD, Cooper BC, et al. Scleroderma and solvent exposure among women. Am J Epidemiol. 2003;157:493-500. [PMID: 12631538] 25. Johnson RW, Tew MB, Arnett FC. The genetics of systemic sclerosis. Curr Rheumatol Rep. 2002;4:99-107. [PMID: 11890874] 26. Arnett FC, Howard RF, Tan F, Moulds JM, Bias WB, Durban E, et al. Increased prevalence of systemic sclerosis in a Native American tribe in Oklahoma. Association with an Amerindian HLA haplotype. Arthritis Rheum. 1996; 39:1362-70. [PMID: 8702445] 27. Reveille JD. Ethnicity and race and systemic sclerosis: how it affects susceptibility, severity, antibody genetics, and clinical manifestations. Curr Rheumatol Rep. 2003;5:160-7. [PMID: 12628048] 28. Hata R, Akai J, Kimura A, Ishikawa O, Kuwana M, Shinkai H. Association of functional microsatellites in the human type I collagen alpha2 chain (COL1A2) gene with systemic sclerosis. Biochem Biophys Res Commun. 2000; 272:36-40. [PMID: 10872800] 29. Crilly A, Hamilton J, Clark CJ, Jardine A, Madhok R. Analysis of transforming growth factor beta1 gene polymorphisms in patients with systemic sclerosis. Ann Rheum Dis. 2002;61:678-81. [PMID: 12117671] 30. Awad MR, El-Gamel A, Hasleton P, Turner DM, Sinnott PJ, Hutchinson IV. Genotypic variation in the transforming growth factor-beta1 gene: association with transforming growth factor-beta1 production, fibrotic lung disease, and graft fibrosis after lung transplantation. Transplantation. 1998;66:1014-20. [PMID: 9808485] 31. Tan FK, Wang N, Kuwana M, Chakraborty R, Bona CA, Milewicz DM, et al. Association of fibrillin 1 single-nucleotide polymorphism haplotypes with systemic sclerosis in Choctaw and Japanese populations. Arthritis Rheum. 2001; 44:893-901. [PMID: 11315929] 32. Artlett CM, Smith JB, Jimenez SA. Identification of fetal DNA and cells in skin lesions from women with systemic sclerosis. N Engl J Med. 1998;338:118691. [PMID: 9554859] 33. Artlett CM. Microchimerism and scleroderma: an update. Curr Rheumatol Rep. 2003;5:154-9. [PMID: 12628047] 34. Nelson JL. Microchimerism and HLA relationships of pregnancy: implications for autoimmune diseases. Curr Rheumatol Rep. 2001;3:222-9. [PMID: 11352791] 35. Maloney S, Smith A, Furst DE, Myerson D, Rupert K, Evans PC, et al. Microchimerism of maternal origin persists into adult life. J Clin Invest. 1999; 104:41-7. [PMID: 10393697] 36. Ohtsuka T, Miyamoto Y, Yamakage A, Yamazaki S. Quantitative analysis of microchimerism in systemic sclerosis skin tissue. Arch Dermatol Res. 2001;293: 387-91. [PMID: 11686513] 6 January 2004 Annals of Internal Medicine Volume 140 • Number 1 49 Review Pathogenesis of Systemic Sclerosis 37. Burastero SE, Galbiati S, Vassallo A, Sabbadini MG, Bellone M, Marchionni L, et al. Cellular microchimerism as a lifelong physiologic status in parous women: an immunologic basis for its amplification in patients with systemic sclerosis. Arthritis Rheum. 2003;48:1109-16. [PMID: 12687555] 38. Ho KT, Reveille JD. The clinical relevance of autoantibodies in scleroderma. Arthritis Res Ther. 2003;5:80-93. [PMID: 12718748] 39. Roumm AD, Whiteside TL, Medsger TA Jr, Rodnan GP. Lymphocytes in the skin of patients with progressive systemic sclerosis. Quantification, subtyping, and clinical correlations. Arthritis Rheum. 1984;27:645-53. [PMID: 6375682] 40. Kraling BM, Maul GG, Jimenez SA. Mononuclear cellular infiltrates in clinically involved skin from patients with systemic sclerosis of recent onset predominantly consist of monocytes/macrophages. Pathobiology. 1995;63:48-56. [PMID: 7546275] 41. Chizzolini C. T lymphocyte and fibroblast interactions: the case of skin involvement in systemic sclerosis and other examples. Springer Semin Immunopathol. 1999;21:431-50. [PMID: 10945035] 42. Sakkas LI, Xu B, Artlett CM, Lu S, Jimenez SA, Platsoucas CD. Oligoclonal T cell expansion in the skin of patients with systemic sclerosis. J Immunol. 2002;168:3649-59. [PMID: 11907131] 43. Ferrarini M, Steen V, Medsger TA Jr, Whiteside TL. Functional and phenotypic analysis of T lymphocytes cloned from the skin of patients with systemic sclerosis. Clin Exp Immunol. 1990;79:346-52. [PMID: 1969335] 44. Shimizu Y, Van Seventer GA, Horgan KJ, Shaw S. Regulated expression and binding of three VLA (beta 1) integrin receptors on T cells. Nature. 1990; 345:250-3. [PMID: 2139716] 45. White B. Immunopathogenesis of systemic sclerosis. Rheum Dis Clin North Am. 1996;22:695-708. [PMID: 8923591] 46. Postlethwaite AE. Role of T cells and cytokines in effecting fibrosis. Int Rev Immunol. 1995;12:247-58. [PMID: 7650423] 47. Roberts AB. Molecular and cell biology of TGF-beta. Miner Electrolyte Metab. 1998;24:111-9. [PMID: 9525693] 48. Varga J, Rosenbloom J, Jimenez SA. Transforming growth factor beta (TGF beta) causes a persistent increase in steady-state amounts of type I and type III collagen and fibronectin mRNAs in normal human dermal fibroblasts. Biochem J. 1987;247:597-604. [PMID: 3501287] 49. Ihn H, Yamane K, Kubo M, Tamaki K. Blockade of endogenous transforming growth factor beta signaling prevents up-regulated collagen synthesis in scleroderma fibroblasts: association with increased expression of transforming growth factor beta receptors. Arthritis Rheum. 2001;44:474-80. [PMID: 11229480] 50. Massague J. How cells read TGF-beta signals. Nat Rev Mol Cell Biol. 2000; 1:169-78. [PMID: 11252892] 51. Attisano L, Wrana JL. Signal transduction by the TGF-beta superfamily. Science. 2002;296:1646-7. [PMID: 12040180] 52. Varga J. Scleroderma and Smads: dysfunctional Smad family dynamics culminating in fibrosis. Arthritis Rheum. 2002;46:1703-13. [PMID: 12124852] 53. Dong C, Zhu S, Wang T, Yoon W, Li Z, Alvarez RJ, et al. Deficient Smad7 expression: a putative molecular defect in scleroderma. Proc Natl Acad Sci U S A. 2002;99:3908-13. [PMID: 11904440] 54. Leask A, Holmes A, Abraham DJ. Connective tissue growth factor: a new 50 6 January 2004 Annals of Internal Medicine Volume 140 • Number 1 and important player in the pathogenesis of fibrosis. Curr Rheumatol Rep. 2002; 4:136-42. [PMID: 11890879] 55. Jimenez SA, Saitta B. Alterations in the regulation of expression of the alpha 1(I) collagen gene (COL1A1) in systemic sclerosis (scleroderma). Springer Semin Immunopathol. 1999;21:397-414. [PMID: 10945033] 56. Trojanowska M. Molecular aspects of scleroderma. Front Biosci. 2002;7: d608-18. [PMID: 11861221] 57. Ghosh AK. Factors involved in the regulation of type I collagen gene expression: implication in fibrosis. Exp Biol Med (Maywood). 2002;227:301-14. [PMID: 11976400] 58. Hitraya EG, Varga J, Artlett CM, Jimenez SA. Identification of elements in the promoter region of the alpha1(I) procollagen gene involved in its up-regulated expression in systemic sclerosis. Arthritis Rheum. 1998;41:2048-58. [PMID: 9811061] 59. Ihn H, Tamaki K. Increased phosphorylation of transcription factor Sp1 in scleroderma fibroblasts: association with increased expression of the type I collagen gene. Arthritis Rheum. 2000;43:2240-7. [PMID: 11037883] 60. Rippe RA, Almounajed G, Brenner DA. Sp1 binding activity increases in activated Ito cells. Hepatology. 1995;22:241-51. [PMID: 7601417] 61. Nehls MC, Brenner DA, Gruss HJ, Dierbach H, Mertelsmann R, Herrmann F. Mithramycin selectively inhibits collagen-alpha 1(I) gene expression in human fibroblast. J Clin Invest. 1993;92:2916-21. [PMID: 7504695] 62. Gaidarova S, Jimenez SA. Inhibition of basal and transforming growth factor-beta-induced stimulation of COL1A1 transcription by the DNA intercalators, mitoxantrone and WP631, in cultured human dermal fibroblasts. J Biol Chem. 2002;277:38737-45. [PMID: 12138160] 63. Saitta B, Gaidarova S, Cicchillitti L, Jimenez SA. CCAAT binding transcription factor binds and regulates human COL1A1 promoter activity in human dermal fibroblasts: demonstration of increased binding in systemic sclerosis fibroblasts. Arthritis Rheum. 2000;43:2219-29. [PMID: 11037881] 64. Widom RL, Culic I, Lee JY, Korn JH. Cloning and characterization of hcKrox, a transcriptional regulator of extracellular matrix gene expression. Gene. 1997;198:407-20. [PMID: 9370309] 65. Campbell PM, LeRoy EC. Pathogenesis of systemic sclerosis: a vascular hypothesis. Semin Arthritis Rheum. 1975;4:351-68. [PMID: 1135634] 66. Drenk F, Deicher HR. Pathophysiological effects of endothelial cytotoxic activity derived from sera of patients with progressive systemic sclerosis. J Rheumatol. 1988;15:468-74. [PMID: 3379623] 67. Kahaleh MB, LeRoy EC. Endothelial injury in scleroderma. A protease mechanism. J Lab Clin Med. 1983;101:553-60. [PMID: 6339651] 68. Renaudineau Y, Revelen R, Levy Y, Salojin K, Gilburg B, Shoenfeld Y, et al. Anti-endothelial cell antibodies in systemic sclerosis. Clin Diagn Lab Immunol. 1999;6:156-60. [PMID: 10066646] 69. Herrick AL. Vascular function in systemic sclerosis. Curr Opin Rheumatol. 2000;12:527-33. [PMID: 11092203] 70. Xu S, Denton CP, Holmes A, Dashwood MR, Abraham DJ, Black CM. Endothelins: effect on matrix biosynthesis and proliferation in normal and scleroderma fibroblasts. J Cardiovasc Pharmacol. 1998;31 Suppl 1:S360-3. [PMID: 9595482] www.annals.org Current Author Addresses: Dr. Jimenez: Division of Rheumatology, Thomas Jefferson University, 233 South 10th Street, Room 509 BLSB, Philadelphia, PA 19107-5541. Dr. Derk: Division of Rheumatology, Thomas Jefferson University, 1025 Walnut Street, Room 613 Curtis, Philadelphia, PA 19107-5541. © American College of Physicians E-15