* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Lecture Outlines
Psychoneuroimmunology wikipedia , lookup
DNA vaccination wikipedia , lookup
Complement system wikipedia , lookup
Immunocontraception wikipedia , lookup
Adaptive immune system wikipedia , lookup
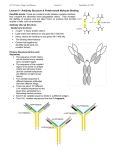
Innate immune system wikipedia , lookup
Adoptive cell transfer wikipedia , lookup
Immunosuppressive drug wikipedia , lookup
Molecular mimicry wikipedia , lookup
Cancer immunotherapy wikipedia , lookup
All contents copyright © 1998-1996 by Klaus D. Elgert and Wiley-Liss, Inc. All rights reserved. Lecture Transparencies for Antibody Structure and Function 1 Antibody Structure and Function n Definition: Antibodies (Abs) are recognition proteins produced by plasma cells (derived from activated B cells) found in serum and other body fluids of vertebrates that react specifically with the antigen (Ag) that induced their formation. n Antibodies belong to a class of proteins called Immunoglobulins (Igs) n Definition of Ig: A family of globular proteins that has in common the Ig fold as a structural domain. These proteins include Ab molecules and molecules having antigenic determinants in common with Abs. n What molecules other than Abs are Igs? Abs Ig Myeloma proteins 2 Antibody Structure and Function l Myeloma proteins are found in individuals who develop a type of cancer call multiple myeloma. l These proteins have the characteristic structure of Abs but arise from a disease state rather than an immune response. l A myeloma protein is an Ig whose Ag-binding specificity is unknown but all the protein molecules are IDENTICAL. n Importance ? Structure of Igs n Why is it important to understand the chemical structure of Igs? n Reason: Because the chemical structure of Igs determines three important functions. 3 Antibody Structure and Function l Versatility in Ag binding t t t l 105 to 109 Even to artificial Ags that do not exist in nature How can Ab molecules be so diverse to allow a response to this vast array of determinants? -And have a common structure. Specificity Why are Abs specific? That is, why don't Abs react with unrelated determinants? l Biological effector mechanisms How do Abs elicit these effector mechanisms? All these properties are explained by determining the CHEMICAL STRUCTURE of Igs 4 Antibody Structure and Function Electrophoresis studies by Tiselius & Kabat Abs = γ−globulins Studies showed that the MW of Abs = 150 to 1000 kD 5 Antibody Structure and Function Edelman & Porter's Work Fragmentation Studies Using Chemicals and Enzymes Chain Structure of Ig Rabbit IgG (150,000 mw)* (Edelman) reducing & IgG =========> subunits of IgG denaturing agents Heavy (H) + (50,000 mw) Light (L) chain (25,000 mw) *Found in equimolar amounts = 2 H + 2 L chains 6 Antibody Structure and Function PAPAIN (Porter): papain IgG =========> Fab + Fc (50 kD) (50 kD) n Fab = fragments Ag binding n Fc = fragment crystallizable n Molar ratio of Fab/Fc was 2:1, therefore there are two Ag-binding sites on each IgG molecule PEPSIN (Nisonoff): pepsin IgG =========> F(ab')2 (100,000 mw) n F(ab')2 fragments have both Ag-binding sites held together by disulfide bonds but missing Fc 7 Antibody Structure and Function n STRUCTURE: IgG composed of 2 L and 2 H chains, which are linked by interchain disulfide bonds and noncovalent interactions. Chemicals L Papain Fab H H L Fc Fab Pepsin n In any Ab molecule, the 2 L chains are IDENTICAL and the 2 H chains are IDENTICAL. ALL Igs HAVE THIS BASIC STRUCTURE! n Our purpose for looking at Ig structure: Three reasons (mentioned earlier) n To do this, we have to look more closely at Ig structure: Ig fine structure 8 Antibody Structure and Function Ig L chains Amino acid sequence comparisons of Bence-Jones κ L chains led to a startling finding NH2 1 110 214 COOH CL VL Ig H chains 1 113 446 VH VL CH CL CH VH VH VL CH CL n IMPLICATION: V regions of L and H chains explain versatility of Ab binding to 105 to 109 different antigenic determinants. Igs binding different determinants have different VL and VH regions. 9 Antibody Structure and Function n Analogy: l l l P-S-Y-C-H O-L-O-G-Y P-H-Y-S-I O-L-O-G-Y I-M-M-U-N O-L-O-G-Y Closer Look at Ab Fine Structure n Further amino acid sequencing of Ig chains showed areas or “hotspots” of increased variability in the V region. n These hotspots are called hypervariable regions or complementarity-determining regions (CDRs) n Less variable amino acid residues/regions between CDRs are called framework residues 10 Antibody Structure and Function Wu and Kabat Plot FR = Framework residues CDRs = Complementarity-determining regions; also called hypervariable regions 11 Antibody Structure and Function Domains n Igs are not linear chains of proteins -- they are globular proteins. n Each Ig chain has homology units called DOMAINS. l Domains are regions of compact globular structure; for example, IgG domains = light chain: V & C and heavy chain: V, Cγ1, Cγ2, Cγ3, & hinge region (IgM and IgE have an additional heavy chain C domain but no hinge region domain) l Two types of Ig-like domains: V and C t l These domains are the prototype of the Ig Superfamily domain structure, also called the Ig fold. What is this Ig fold? All Ig domains contain two layers of β-pleated sheet with 3 or 4 strands of antiparallel polypeptide chain (a "sandwich" structure). This characteristic structure is the Ig fold. t Significance: holds loops out from sandwich 12 Antibody Structure and Function The Ig Superfamily n The Ig superfamily is characterized by their 3D structure because all the members contain at least one Ig fold. n The Ig fold, so-called because it is a single-stranded β-sheeted barrel composed of a sandwich of two extended protein sheets roughly 110 amino acid residues long. n The two antiparallel sheets (purple) are connected with one disulfide bridge (yellow). Side-view of Ig fold 13 Human Ab Light Chain Folding Into Ig Domains CL domain VL domain b strands COOH Disulfide bridge CDRs NH2 Location of CDRs in VL domain CDR1 CDR2 CDR3 Light chain 28-34 89-97 50-56 CL domain 14 3D Structure of Complete IgG VH domain CL domain CH1 VH domain Antigen-binding site Antigen-binding site VL domain Carbohydrate chain VL domain CH2 Heavy chains CH3 (a) VH Antigenbinding site VL CH1 CL S VL S CH2 CH2 Carbohydrate CH3 (b) VH Antigenbinding site 15 Antibody Structure and Function Summary of Ig Fine Structure n Roughly, the first 110 amino acid residues at the NH2 termini of both L and H chains comprise the V region (the Ag-binding site). n Within the V region, some areas are more variable than others. l Areas of greatest variability are called Hypervariable Regions or Complementarity-Determining Regions (CDRs) Responsible for the shape of the Ag-binding site of Igs l Amino acid residues between CDRs are called Framework Residues n L and H chains are divided into homology units, domains, which have in common the Ig fold. n The COOH termini contain the C regions. 16 Ig Domains Antigen-binding clefts Light chains NH2 Fab S S S Fab S VL S S S S CL VH S S S S S S Variable region S S S S S S S S CH1 S S Hinge region Heavy chain S S CH3 S S S S CH2 Carbohydrate S S COOH Fc Biological activity 17 Antibody Structure and Function All Known Ab-Mediated EFFECTOR FUNCTIONS Are Ascribed to the Fc Region n They can be divided into three generic categories: l l l Activation of the classical complement pathway Interaction with effector cells t For example: Mφs, NK cells, or mast cells Compartmentalization of Abs (where Abs localize) n There are three main ways Abs contribute to immunity: l l l Neutralization Opsonization Complement activation n Abs protect the host by: l l l l l binding to bacterial toxins and viruses, leading to their neutralization enhancing phagocytosis of microorganisms activation of complement, leading to lysis of cells localizing at mucous membrane surfaces, they bind to organisms and prevent colonization binding to Fc receptors on cells to mediate ADCC 18 Antibody Structure and Function Human Ig Classes and Subclasses n There are five major groups of human Igs, called Immunoglobulin Classes, they include: IgG, IgA, IgM, IgD, and IgE ? How do you know what class an Ig belongs to? n Igs are divided into classes on the basis of the antigenic determinants they possess on their heavy chains. n Concept: Abs can be IMMUNOGENIC 19 Antibody Structure and Function Heavy (H) Chains of Igs n Abs specific for H chain antigenic determinants define the Ig class an Ab belongs to Human Ig = Ab against human Ig n There are 5 antigenic types of H chains in humans. The antigenic determinant that defines the H chain is: H chain type Ig class 1. γ IgG 2. α IgA 3. µ IgM 4. δ IgD 5. ε IgE 20 Antibody Structure and Function n Ab specific to a particular H chain will react only with that chain and not other H chains. H chains always γ n IgG L chains can be κ or λ n Similar for other Igs n Ig subclasses l l Even within a class of Ig (e.g. IgG) the H chains have minor antigenic variations. Examples: IgG H chain has 4 variations, therefore IgG is divided into: H chain γ1 γ2 γ3 γ4 IgG IgG1 IgG2 IgG3 IgG4 21 Antibody Structure and Function n IgA H chain has 2 variations, therefore IgA is divided into: H chain IgA α1 IgA1 α2 IgA2 LIGHT (L) CHAINS OF Igs kappa (κ) n Light chains lambda (λ) n An Ab molecule will have either κ or λ L chains but not both together. n κ and λ chains are found on all classes of Igs. 22 Antibody Structure and Function n Three categories of antigenic determinants found on Igs: l l l Isotypic Allotypic Idiotypic n These determinants define isotypes, allotypes, and idiotypes of Igs. n Isotypic antigenic determinants: determinants present on some Igs of ALL members of a species. The antigenic differences that characterize the class and subclass of H chains and the type and subtype of L chains are called isotypes. Thus, isotype refers to an Ab class. l Each normal individual expresses all the isotypes, characteristic of the species. 23 Antibody Structure and Function n Allotypic antigenic determinants: determinants present on the Igs of only SOME members of a species, because these epitopes are encoded by alleles (or alternative genes) at a single locus. The Igs are called allotypes. They are due to polymorphisms within species in the amino acid sequence of Ab isotypes. l Example: The IgG1 of one person is not necessarily absolutely indentical to the IgG1 of another person. n Idiotypic antigenic determinants: determinants that are common to Igs having specificity for the same epitope on Ag molecules. (idiotypic determinants reflect the Ag-bining site structure) l Example: two Igs both IgG1 with κ L chains. t Both have the same allotypic markers; one Ab is specific for Ag epitope A, the other is specific for Ag epitope B. The difference in chemical structure of Abs needed to bind these epitopes, define their idiotypes. 24 Antibody Structure and Function Structure of Human Ab Classes When in Their Soluble Form (As part of the B-cell antigen receptor complex: their carboxyl termini have hydrophophic sequences, they are monomeric, and they contain two identical Ig-α/Ig−β heterodimers) 25 Antibody Structure and Function Human Ab Classes n Immunoglobulin G (IgG): the predominant class of Abs found in humans. l IgG is the only Ab that can cross the placenta from mother to fetus, and is responsible for immunologic protection of an infant during its first few months after birth. l IgG is: t the primary Ab induced by protein Ags, the main Ab in the secondary Ab response (has higher affinity for antigen) and t the main Ab involved in neutralization (any isotype can), opsonization, ADCC, complement activation, and precipitation reactions. n Immunoglobulin A (IgA): is the predominant Ab found in human bodily secretions such as saliva, tears, and nasal mucous. l It is a monomeric molecule in the blood. l In bodily secretions, it is found as a dimer composed of two Ab molecules, a secretory component, and a joining (J) chain; therefore, this form is called secretory IgA. l It can bind to 4 epitopes. l It can activate the alternative complement pathway. 26 Antibody Structure and Function n Immunoglobulin M (IgM): is present, as a monomer, along with IgD on the surface of mature B cells. l In blood, IgM normally exists in clusters of five (pentamer), connected at the bottoms of the Y. l The monomers are held together by disulfide bridges to a polypeptide called the J chain. l IgM can bind to 10 epitopes. l It is prominent in the primary Ab response to most Ags. l IgM is the main Ab: t induced by polysaccharide Ags and t involved in agglutination and complementmediated cytolytic reactions. n Immunoglobulin E (IgE): is involved in allergic reactions, because it naturally binds to certain cells causing release of compounds that mediate these reactions. n Immunoglobulin D (IgD): is the predominant Ab class found on Ag-naive B cells. l It may be involved in B-cell growth and maturation. l It is found in trace amounts in human blood. 27 Antibody Structure and Function Monoclonal Abs n Heterogeneous response vs. need for homogeneous Ab n Homogeneous Ab: l l l Specific for a single determinant of the Ag Same class and subclass All the Ab would bind the determinant with the same affinity Why search for homogeneous Ab? 28 Antibody Structure and Function n Studies of the interaction between epitopes and the Abs’ Ag-combining sites l Use myeloma proteins t l l Problem: Is the Ag-binding site of the myeloma protein really an accurate representation of Ab produced during an immune response? Limit cross-reactivity of Ab Standardization of diagnostic reagents 29 Antibody Structure and Function n In 1975, Kohler and Milstein developed homogeneous Ab against any antigenic determinant in almost unlimited quantity (1984 Nobel Prize) n Two problems to overcome: l l Ab-forming cell = production of specific Ab but it is a terminal cell Myeloma cell = Ab of unknown specificity but it is an immortal cell n Fused with a myeloma cell that had two unusual properties: l l It did NOT secrete Ig Metabolically deficient t t Lacked the enzyme Hypoxanthine-Guanine Phosphoribsyl Transferase (HGPRT−) It cannot use the exogenous hypoxanthine to synthesize purines. 30 Antibody Structure and Function Hybrid-myeloma HYBRIDOMA Resulting fusions Plasma cell FUSION = Myeloma cell How to select for proper hybrid? n Culture cells in a selective medium (Hypoxanthine-Aminopterin-Thymidine; HAT) for a few weeks n Grow-up selected clone of desired Ab specificity = Monoclonal Ab 31 Conventional Antisera vs. Monoclonal Antibody Production CONVENTIONAL ANTIBODY PRODUCTION MONOCLONAL ANTIBODY PRODUCTION Antigenic determinants Repeated antigen inoculation Antigen Remove spleen The mouse produces B cells specific for each antigenic determinant. FUSION Spleen cells Myeloma cells Hybrid-myeloma cells Antiserum (contains a mixture of antibodies specific for each antigenic determinant) Clone 1 Grow up individual cells into clones Clone 2 Clone 3 Individual populations of antibodies 32 Development of Monoclonal Antibodies Repeated antigen inoculation Fusion Remove spleen Nonsecretor myeloma cells (HAT sensitive) Culture may contain: 1 2 Spleen cells grown in HAT medium (HAT resistant) 3 Culture in HAT medium 5 1 2 3 Test for positive cultures (contain antibody against antigen) 4 5 Clone antibody producers Spleen cells (end cells, thus will die with time) Myeloma cells (killed by HAT medium) Fused spleen cells (same as no. 1) Fused myeloma cells (same as no. 2) Fused spleen cells—myeloma cells (hybrid cell = hybridoma; lives forever [myeloma cells] and produces antibodies [spleen cells or plasma cells]) CELLULAR DNA SYNTHESIS 1 2 De novo pathway (endogenous sources) Salvage pathway (exogenous sources) H HGPRT Ribonucleotide De novo DNA Ribonucleotide TK T 4 33 Transfectomas: the production of hybrid antibody molecules IN VITRO MANIPULATIONS Human H Mouse H chain gene chain gene Leader Leader VDJ VDJ 1. CH1 2. INTRODUCTION INTO E. coli plasmid CH1 Hinge Hinge CH2 CH2 CH3 CH3 + L chain gene Proliferating E. coli containing the plasmid Mouse–human recombinant heavy chain gene 3. TRANSFECTION + E. coli spheroplast 4. Myeloma cell TRANSFECTANT Selection EXPANSION Tissue culture HUMAN–MOUSE RECOMBINANT ANTIBODY Ascites