* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Ideal Ventricular Assist Device
Cardiovascular disease wikipedia , lookup
Remote ischemic conditioning wikipedia , lookup
Electrocardiography wikipedia , lookup
Rheumatic fever wikipedia , lookup
Cardiac contractility modulation wikipedia , lookup
Management of acute coronary syndrome wikipedia , lookup
Lutembacher's syndrome wikipedia , lookup
Heart failure wikipedia , lookup
Cardiac surgery wikipedia , lookup
Jatene procedure wikipedia , lookup
Coronary artery disease wikipedia , lookup
Quantium Medical Cardiac Output wikipedia , lookup
Dextro-Transposition of the great arteries wikipedia , lookup
Mitral insufficiency wikipedia , lookup
Ventricular fibrillation wikipedia , lookup
Hypertrophic cardiomyopathy wikipedia , lookup
Arrhythmogenic right ventricular dysplasia wikipedia , lookup
Cardiomyopathy, End-stage Heart
Disease & Transplantation
Seoul National University Hospital
Department of Thoracic & Cardiovascular Surgery
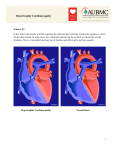
Hypertrophic Cardiomyopathy
Hypertrophic Cardiomyopathy
Definition
• A myocardial disease characterized by left and/or right
ventricular hypertrophy that is usually asymmetric and is
associated with microscopic evidence of myocardial fiber
disarray. Degree of hypertrophy at any given site can
vary substantially & influences clinical manifestations.
• Ventricular septal hypertrophy is the most common type of
asymmetric hypertrophy, with midventricular, apical, and other
types occurring much less frequently.
• Forms interfering with left ventricular emptying, termed
hypertrophic obstructive cardiomyopathy or idiopathic hypertrophic
subaortic stenosis, are surgical important and variable subaortic
obstruction and is associated with abnormal systolic anterior motion.
• The more commonly occurring nonobstructive forms are not
amenable to surgical treatment except for cardiac transplantation.
Hypertrophic Cardiomyopathy
Historical note
•
•
•
•
•
•
•
Hallopeau & Liouiville ; Pathologic finding compatible with HOCM in 19
century
Schmincke
; Pathologic finding in early 20 century
Davies
; Described family form in 1952
Brock
; Surgical report for diffuse muscular subaortic
stenosis
Braunwald & Goodwin ; Described respectively idiopathic hypertrophic
subaortic stenosis & hypertrophic obstructive cardiomyopathy
Bigellow
; Simple myotomy using an aortic approach in
1966
Morrow
; Excision of muscle in 1978
Coronary Flow Reserve
Determinants of microvascular dysfunction
• Narrowing of epicardial coronary arteries
• Structural changes (ie, vascular remodeling with reduced
lumen to wall ratio) or functional alterations involving
neurohumoral factors
• Small coronary arterioles may change their diameter as a
result of autonomic innervation
• Several extravascular mechanisms such as impaired
diastolic relaxation, compression of the coronary arteries
by high left ventricular filling pressures, and increased
force of contraction ("milking").
Hypertrophic Cardiomyopathy
Morphogenesis
• Hypertrophic cardiomyopathy is recognized a
heterogenous sarcomere diseases, and
mutations have been described in the betamyosin heavy chain gene (chromosome 14q11q12), in cardiac troponin-T( chromosome I ), in
alpha-tropomyosin ( chromosome 15q2), and in
two other chromosomes.
Hypertrophic Cardiomyopathy
Characteristics
• Ratio of thickness between septum and posterior wall is
1.3 or more in almost HCM.
• ASH tends to lessen or disappear with somatic growth
when present in early life in association with congenital
heart disease.
• Increased wall thickness is mainly caused by increased
fibrous tissue, particularly in the ventricular septum.
• Foci of disarrayed muscle cells are interspersed and also
abnormalities in orientation of myofibrils.
• The LV cavity is small and has a S or sigmoid shape in
systole.
• Rarely, LV cavity may become dilated in the late stages
of HOCM.
Hypertrophic Cardiomyopathy
Etiology
• HCM is a genetically determined disorder of cardiac
muscle transmitted as an autosomal dominant trait,
although nonfamilial cases probably occur as well.
• HCM can present at any age from early infancy to the
sixth or seventh decade.
• Echocardiographic studies of patients with HCM,
including those with isolated ASH suggest that
obstruction is present in only about 20%.
• It is uncertain whether isolated ASH, an asymptomatic
disease, develops into obstructive cardiomyopathy.
Hypertrophic Cardiomyopathy
Natural history
• The natural history of HCM is typically variable.
• Progression of disease is more rapid in children and young
adults
• Symptomatic infants and young children represent the
more severe end of spectrum.
• Annual mortality of HCM has ranged 4-6% in children,
and 3-4% in adults.
• Sudden cardiac death is common and the risk factors are
young age, syncope, family history of malignancy,
myocardial ischemia, sustained VT, degree of outflow
obstruction.
Hypertrophic Cardiomyopathy
Morphology
Muscular hypertrophy present in HCM involves the
interventricular septum and left ventricle, and id variable
in its location and severity
•
•
•
•
•
•
•
•
•
•
Ventricular septum
Dynamic morphology of septum and mitral valve
Left ventricular free wall
Left ventricular cavity
Histopathology of left ventricle
Left atrium
Mitral valve
Right ventricle
Coronary arteries
Associated lesions
Hypertrophic Cardiomyopathy
Clinical features & diagnosis
• Symptoms ; angina, DOE, syncope, palpitation
• Signs ; late-onset ejection murmur, bifid arterial pulse,
palpable left atrial contraction
•
•
•
•
Ventricular function ; initial diastolic dysfunction
EKG ; LV strain, sometimes Q wave, LVH
Chest radiography ; variable cardiomegaly
Echocardiography & catheterization
Hypertrophic Cardiomyopathy
Mitral regurgitation
• SAM of the anterior leaflet is a constant features of
classic HOCM.
• It is likely that severity of mitral regurgitation,
magnitude of pressure gradient, and degree of
prolongation of LV ejection time are determined by
time of onset and duration of mitral leaflet-septal
contact.
• Mitral regurgitation occurs independent of SAM in
about 20% of patients with HOCM.
• It can result from mitral valve prolapse, chordal
rupture, anomalous attachment of a papillary muscle,
anterior leaflet fibrosis, congenital abnormalities,
rheumatic disease, or annular calcification
Systolic Anterior Motion
• The mechanism of SAM is probably multifactorial,
most likely, secondary to forward (anterior)
displacement of the elongated mitral valve relative to
the septum during systole.
• The Ventury effect of the high-velocity stream of blood
carries the protruding edge of anterior leaflet toward
the aortic annulus in early systole
• SAM is absent in the nonobstructive HCM
• SAN can occur in TGA with IVS
• SAM may also appear after inserting a rigid mitral
anuloplasty ring
Systolic Anterior Motion
Proposed Mechanism
A;
Coaptation point( arrow ) is in the body of anterior and posterior leaflets.
B & C ; Anterior and basal movement of the residual length of the anterior leaflet with
septal contact and failure of leaflet coaptation & subsequent mitral regurgitation
Technique of Operation
Myectomy by aortic approach
Adjunts to conventional myectomy
•
Extended myectomy & reconstruction of
subvalvular mitral apparatus
•
Plication of anterior leaflet with myectomy
•
Ventriculotomy with transaortic approach
Modified Konno operation
Mitral valve replacement
Hypertrophic Cardiomyopathy
Postoperative care
• LAP of 16-18 mmHg required early postoperatively for
adequate volume
• Digitalis, beta-receptor agonist should be avoided
• Hypovolemia and nitroglycerin, which can reduce LV
volume and exaggerate any residual gradient, should be
avoided.
• Atrial fibrillation may be poorly tolerated
• This can be best accomplished by use of beta-adrenergic
receptor blocking agents ( propranolol ), calcium
antagonists ( verapamil, diltiazem ), or amiodarone
Results of Operation
•
•
•
•
•
•
•
•
•
•
•
•
•
Early death
Time-related survival
Mode of death
Incremental risk factors
Myocardial metabolic changes
Conduction disturbance
Perioperative myocardial infarction
Iatrogenic defects
Postoperative pressure gradients
Mitral regurgitation
Left ventricular aneurysm
Symptomatic status
Left ventricular function
Postoperative Aortic Regurgitation
Causes after operation for HOCM
• Small aortic annulus probably by increased
operative difficulty & increased retraction &
possible injury of aortic valve cusp
• Loss of support of right coronary cusp as a result
of excising septal muscle beneath it may result in
aortic regurgitation as may altered velocity,
direction, and dynamics of the turbulent jet of
blood in the outflow tract
Incremental Risk Factors
•
•
•
•
•
•
•
Preoperative syncope
Increased NYHA functional class
Documented coronary artery disease
Concomitant procedures
Mitral valve replacement
Development of complete heart block
Outflow tract gradients greater than 15mmHg
were incremental risk factors for late death
Hypertrophic Cardiomyopathy
Indications for operation
• Symptomatic patients after appropriate medical
therapy, pacemaker therapy, or septal ablation and
who has LVOT gradient at rest more than 50mmHg
• Symptomatic patients with small gradient at rest but in
whom a gradient of 50mmHg or greater on provocation,
after ectopic beat, or after cessation of exercise
• Occurrence of atrial fibrillation is also an indication
• Less symptomatic patients with severe gradients, with
MR, history of syncope, asymptmatic young patients
with gradients more than 100mmHg
Hypertrophic Cardiomyopathy
Special situations & controversies
Alterative therapy
• Left ventricular-aortic conduit
• Dual-chamber pacing
• Percutaneous transluminal septal
myocardial ablation
• Cardioverter-defibrillator
• Cardiac transplantation
Heart Failure
Heart Failure
Definition
• A clinical syndrome that represents a complication or
common final pathway of many heart diseases in which
defective cardiac filling( diastolic heart failure ) or
impaired contraction( systolic heart failure ) or
emptying results in the heart’s ability to pump a
sufficient amount of blood to support tissue metabolism,
or to be able to do so only with elevated filling pressure.
• It is commonly characterized by secondary organ
abnormalities in the skeletal muscles( fatigue ),
lungs( dyspnea ), and kidneys( salt & fluid retention )
Heart Failure
Pathophysiology
•
•
•
•
Cardiorenal mechanism
Hemodynamic mechanisms
Neurohumoral mechanisms
Myocardial hypertrophy & ventricular
remodeling mechanisms
• Other factors
Mechanisms of Remodeling
• Transition from compensatory hypertrophy to heart
failure is related to alterations in cell organization and
changes in coronary blood flow to the increased cell mass
of the hypertrophied ventricle.
• Alterations in myocyte biology include excitationcontraction coupling, myosin heavy chain or fetal gene
expression, beta-adrenergic desensitization.
• Alteration in the extracellular matrix of the myocardium
include replacement fibrosis.
• Changes in configuration of the ventricular chamber
include dilation, change in shape( increased sphericity),
thinning of wall and regurgitation.
Heart Failure
Clinical features & diagnostic criteria
• Stage A ; patient at high risk for developing heart
failure but has no structural disorder of the heart
• Stage B ; patient with structural disorder of the heart
but has never developed symptoms of heart failure
• Stage C ; patient with past or current symptoms of
heart failure associated with underlying structural
heart disease
• Stage D ; patient with end-stage disease
Heart Failure
Natural history
• About 3% of the adult population is treated for heart
failure and occurrence of heart failure increases with
age so that 6% to 10% of people older than 65 years
have heart failure
• Heart failure accounts about 5-10% of all hospital
admissions.
• Heart failure results in nearly 300,000 deaths per year
in the United States, 60% sudden.
• Sudden death may be completely unexpected(1/3), a
consequence of worsening heart failure(1/3), or a result
of progression of heart failure alone(1/3)
Cardiomyopathy
Definition
• A cardiac muscle disease process that leads to
clinical myocardial dysfunction
• The disease process results in morphologic
changes in the heart that are typically classified
as (1) dilated cardiomyopathy, (2) hypertrophic
cardiomyopathy, (3) restrictive cardiomyopathy,
and (4) arrhythmogenic right ventricular
dysplasia
Dilated Cardiomyopathy
Definition
• A cardiac muscle disease characterized by dilatation of
one or both ventricles and impairment of at least systolic
function. Dilated cardiomyopathy may be considered the
final outcome of pathways produced by a variety of
agents of myocardial insult
• These include selenium deficiency , alcohol, smoking,
and a variety of viral agents and in some patients, the
causative factor may be immune, genetic, or familial.
• In many patients, none of these can be identified, & the
condition is termed idiopathic dilated cardiomyopathy
Dilated Cardiomyopathy
Morphology
• Enlargement (increased volume) of ventricles
and, to a lesser extent, the atria
• Variable degree of hypertrophy is often present
• Extensive interstitial & perivascular fibrosis,
occasionally calcification, in the ventricular
myocardium in microscopic examination
• Myocardial cell degeneration is usually seen
• The specific diagnosis of DCM usually cannot be
made by endocardial biopsy
Dilated Cardiomyopathy
Clinical features & diagnosis
• DCM frequently is of unknown etiology
• Speculation as to possible progression of infective,
particularly viral, myocarditis to full-blown dilated
cardiomyopathy, particularly frequent in children
• Alcoholism, pregnancy, and systemic hypertension may
provide a background for its development
• About 25% of patients have familiar disease (X-linked)
• Characterized by impaired systolic function, but in late,
decreased left ventricular compliance may develop
• Al forms of cardiomyopathy may have a nonspecific
prodromal phase, lasting weeks or months
Dilated Cardiomyopathy
Natural history
• DCM is a serious disease, and about 80% of patients
are dead within 10 years of its evident onset
• The course is variable, with some patients dying within 1
to 2 years and a few having more fulminating course
• Cardiac antibodies play a functional role and their
removal may induce hemodynamic improvement
• A few patients with dilated cardiomyopathy recover
spontaneously.
• Mode of death is usually chronic cardiac failure, or
occasionally intractable arrhythmias, and sometimes
sudden
Dilated Cardiomyopathy
Risk factors for death
• Marked cardiomegaly
• Cardiac rhythm other than sinus,
especially ventricular arrhythmia
• Pulmonary hypertension
• Elevated right atrial pressure
• Thromboembolism in great LV with
atrial fibrillation
Restrictive Cardiomyopathy
Definition
• A cardiac muscle disease that results in impaired
diastolic function with loss of compliance
Morphology
• Characterized by diffuse ventricular hypertrophy. The
ventricular walls are excessively rigid, resulting in
restrictive filling and reduced volume of ventricle with
normal or near normal systolic function.
Microscopically, fibrosis and hypertrophy of myocytes
are usually apparent.
Restrictive Cardiomyopathy
Clinical features & diagnosis
•
Restrictive cardiomyopathy may be secondary to
amyloid infiltration and other process, with or
without eosinophilia. In number of cases the
etiology is unknown
• This condition simulates chronic constrictive
pericarditis with severe impairment of compliance
• Generally, symptoms are of long duration, & death
is delayed for 5 to 20 years after abnormalities of
cardiac function and not well defined
Endomyocardial Fibroelastosis
Definition
• A form of restrictive cardiomyopathy with unknown
etiology in which the pathologic process is restricted to
the endocardium
Morphology
• Fibrous endocardial lesions involving primarily the
inflow portions of right and left ventricles
• The outflow of the ventricle is usually spared.
• Both ventricles are commonly involved, but 40% purely
in LV and 10% in RV involvement
• A thick layer of hyalinized fibrous tissue, calcification
in endocardium and sparsity of elastic fiber
• Possible role of diet in banana, malnutrition, and various
infections as well as an immunologic response
Endomyocardial Fibroelastosis
Clinical features & diagnosis
• As progressively increasing endomyocardial fibrosis
develops, with consequent restriction of ventricular
filling, ventricular end-diastolic pressure elevate as do
pulmonary or systemic venous pressure, depending on
which ventricle is involved
• Involvement of AV valves then adds valvar regurgitation
to the already impaired hemodynamic state
• Endomyocardial fibrosis(or obliterative cardiomyopath )
tends to affect children and young adults with. It occurs
primarily in Uganda, Nigeria, and India.
• This type of cardiomyopathy is generally unfavorable,
slowly deteriorating course and death within 5 to 10
years, often within 1 to 2 years.
Secondary Cardiomyopathies
Associated with cardiac or systemic disorders
Ischemic
Valvar
Hypertensive
Inflammatory - Myocarditis, Chanas disease, HIV
Metabolic – Thyrotoxicosis, Hypothyroidism, Storage diseases
Systemic diseases - Systemic lupus erthematosus, Sarcoidosis
Muscular dystrophies - Duchenne’s, Becker-type
Neuromuscular disorders - Friedreich’s ataxia
Sensitivity and toxic reactions – Alcohol, Radiation,
Anthracyclines
Peripartum (pregnancy)
Treatment of Heart Failure
Therapy for Heart Failure
•
•
•
•
Non-drug therapy
Dietary sodium restriction
Exercise training
Treatments of no benefit or harm
Calcium-channel blockers
Positive inotropic therapy
Heart Failure
Drug therapy
• Angiotensin-converting enzyme inhibitors, ACE1
Enalpril 10mg bid
• Angiotensin-receptor blocker, RBs
Losartan 50mg, captopril
• Beta-blocker ; Carvedilol, metoprolol, bisoprolol
• Aldosterone receptor-blocker
Spironolactone 25~50mg/day
• Vasodilator ; Hydralazine & isosorbide dinitrate
• Digoxin
• Diuretics
• Antiplatelet therapy & anticoagulation
End-Stage Heart Disease
Surgical Options
1. Ventricular assist device
2. Dynamic cardiomyoplasty
3. Ventricular volume reduction
4. Heart transplantation
Treatment of Dilated Ventricle
Options to Reserve Compensatory
Mechanism
1. Increase the LV mass (cardiomyoplasty)
2. Decrease the wall tension (vasodilator)
3. Reduce the LV radius (cardioreduction)
Myocardial Infarction
Sequence
• Acute & chronic inflammatory reaction after
infarction and myocardial fibrosis
• Ventricular pressure stretches & thins the
healing area including ventricular dilation.
• The dilated heart may result in congestive
heart failure.
• Ventricular aneurysm may form, further
compromising heart function.
Ischemic Cardiomyopathy
Ventricular Reconstruction
• Recommended in patients with coronary
disease as a treatment for heart failure, angina,
and thromboembolic complications or to
control ventricular arrhythmias
Technical modifications
• Purse-string technique
• Endoaneurysmorrhaphy technique
• Endoventricular circuloplasty
Volume Reduction Surgery
1. Selection
1) Dilated cardiomyopathy (LVEDD>70mm)
2) Contraindication to transplantation
3) Hemodynamic deterioration waiting
transplantation
2. Exclusion
1) Ischemic cardiomyopathy
2) Cardiac fibrosis
3) Active myocarditis
Heart Reduction Surgery
Cardiac Function after Reduction
1. Increases in end-systolic elastance & preload
recruitable stroke work, and ejection fraction
due to decrease in LVEDV with no little
change in stroke volume.
2. Decreases in LVEDV and increases in diastolic
chamber stiffness.
3. At any ventricular pressure, mass reduction
results in a decrease in ventricular wall stress.
(reduction of myocardial afterload, and
subendocardial ischemia)
Dor Procedure
Pathophysiology
• Relieve ischemia by revascularization
• Diminish ventricular volume
• Restore the ventricle to more normal
geometry
• Further diminishes volume overload
by mitral valve repair when appropriate
Overlapping Ventriculoplasty
• Schema of integrated overlapping ventriculoplasty with PMP
Septal Aneurysm Patch Exclusion
A, Apical aneurysm with significant thinning and aneurysmal involvement of
distal septum. B, Pericardial patch sewn to the preserved normal portion of
the septum on three sides. C, The patch effectively excludes the aneurysmal
portion of the septum
Dor Procedure
Indications
• Anteroseptal infarction and dilated left ventricle
(enddiastolic volume index >100mL/m2)
• Depressed ejection fraction(even below 20%)
• Left ventricular regional asynergy greater than
35%
• Symptoms of angina, heart failure, arrhythmias
• Inducible ischemia in asymptomatic patients
Dor Procedure
Contraindications
• Systolic PA pressure more than 60mmHg
( when not associated with severe MR)
• Severe RV dysfunction as assessed by
tricuspid annulus plane systolic excursion
• Regional asynergy without dilation of the
ventricle ( risk of too small a residual
ventricle)
Mechanisms of Cardiomyoplasty
• Girdling effect on the left ventricle
reduce chronic dilation
decrease diastolic strain
• Enhance systolic performance
decrease myocardial workload
decrease cardiac oxygen consumption
improve myocardial efficiency
Cardiac Transplantation
History
• 1960 ; R. Lower & N. Sumway
Successful canine orthotopic cardiac transplantation
• 1967 ; C. Barnard
Successful human orthotopic cardiac transplantation
• 1972 ; P. Caves
Percutaneous transvenous RV endomyocardial biopsy
• 1974 ; M. Billingham
Standard grading system for cardiac biopsy
• 1979 ; Cyclosporin A in clinical immunosuppresion
• 1985 ; L. Bailey
Successful infant orthotopic transplantation
Cellular Transplantation
•
•
•
•
•
Cardiomyocyte
Bone marrow cell
Human mesenchymal stem cells(hMSCs)
Mouse embryonic stem cell(ESCs)
Intracoronary or transendocardial
transplantation of autologous mononuclear
bone marrow cells
• Cellular transplantation by autologous,
allogeneic, or xenogeneic cells for cardiac
repair
Cellular Xenotransplantation
Cells for cardiac repair
• Somatic cells
• Adult stem cells
Bone marrow stem cells
Satellite cells
• Embryonic stem cells
Heart Transplantation
Indications
Infants
•
•
•
•
•
Hypoplastic left heart syndrome
Dilated cardiomyopathy
Aortic stenosis with endocardial fibroelastosis
Unstable ventricular tachycardia
Others
Children
•
•
•
•
•
Cardiomyopathy, dilated and restrictive
Single ventricle s/p Fontan procedure
Complex cardiac anomalies s/p palliative surgery
Other complex cardiac anomalies s/p corrective procedure
Retransplantation, TCAD, Rejection, Early graft failure
Heart Transplantation
Contraindication in pediatric age
•
•
•
•
•
•
•
Fixed PVRI more than 6 unit/BSA
Fixed TPG more than 15mmHg
Active infection
Severe metabolic disease
Multiple severe congenital anomalies
Advanced multiple organ failure
Active malignancy
Pretransplant Evaluation
Critical determinants
• Pulmonary vascular resistance index
PVRI (units/square M) = PAP-PAWP(mmHg)
*mmHg= mean pressure. CI(L/min/square M)
• Transpulmonary artery gradients
TPG (mmHg) = PAP-PAWP(mmHg)
• Fixed PVRI of 6 units or greater and /or a
TPG>15mmHg that do not respond to vasodilator
therapy (nitroglycerin, milinone, dobutamine, oxygen,
nitric oxide) are contraindications
Heart Transplantation
Donor Criteria
•
•
•
•
•
•
•
Meets requirements for brain death
Consent from next of kin
ABO compatible
Weight compatible (1 to 3 times recipient )
Normal echcardiogram
Age under 40 years
Normal heart morphology at harvest
Neonatal Transplantation
Fetal Listing
• A heart defect currently not considered
correctable
• Normal pulmonary artery anatomy
• Estimated fetal weight more than 2 Kg
• Greater than 35 weeks gestation
• Normal chromosome
• No significant extracardiac defects
Heart Transplantation
Indications
Risk Factors for Transplantation
Right Atrial Heart Transplantation
• Devised by Lower and Shumway
Bicaval Heart Transplantation
• Devised by Dreyfus & simplified by Sievers et al
Postoperative Management
• Immediate treatment
Isolation room with anteroom
Lines & tubes are removed by progression
Usually all catecholamines are used
• Immunosuppression
• Rejection surveillance
• Graft coronary artery diseases
• Childhood diseases
• Growth and development
• Posttransplant lymphoproliferative disease
Sequence of Immunosuppression
• The transplant recipient recognizes proteins encoded
by major histocompatibility complex of donor gene,
referred to human leukocyte antigen
• The macrophages react to these foreign antigen, which
are then recognized by T-cells, promoting the release of
interleukin-1 from the macrophages
• T-cells are stimulated, and cellular proliferation and
differentation occur with production of lymphokines
( interleukin 2) & other mediators of rejection response
• The production of cytotoxic T lymphocytes,
macrophages, and lymphokines attempt to bring about
destruction of donor graft
Allo-immune Reaction
• T-cell activation through three signals
Immunosuppressive Action Site
Anti-CD 154 antibody has been withdrawn from clinical trial but remains
of interest. FTY720 engagement of sphingosine-1-phosphate(S-1-P)
receptors triggers and internalizes and alters lymphocyte recirculation,
causing lymphopenia. Antagonists of chemokine receptors are also being
developed in preclinical models. MPA denotes mycophenolic acid.
Rejection Surveillance
Clinical assessment
• Heart rate and activity change
• Atrial & ventricular ectopy & resting tachycardia
Echocardiography
• LVED dimension increase of 20%
• LV posterior wall thickness increase of 20%
• LV shortening fraction decrease of 20%
Endomyocardial biopsy
•
•
•
•
Analysis of humoral and vascular rejection
Initially every 2 weeks during 1-3 months
Monthly during 3-6 months
Repeated every 3-6 months until 2 years
Graft Coronary Artery Disease
Potential etiology
•
•
•
•
Chronic cellular rejection episodes
Hyperlipidemia
Cytomegalovirus infection
Vascular rejection
Characteristics
• Concentric intimal proliferation with intact internal
elastic laminae and is different from naturally
occuring atherosclerosis
• The media is normal and thickened intima consists
of smooth muscle cells with macrophages
Heart Transplantation
Posttransplant tricuspid regurgitation
• Complication of the endomyocardial biopsy procedure
• The presence of pulmonary hypertension may cause
right ventricular and annular dilatation, causing TR
• Superimposition of pulmonary injury or right
ventricular dysfunction may contribute to a process
leading to development of TR.
• Right ventricular dysfunction can be caused in the
intraoperative period by preservation or reperfusion
injury, air embolus, donor risk factors, or accelerated
rejection.
• Distortion of the right atrial–right ventricular
relationship caused by the implantation technique.
Heterotopic Heart Transplantation
Heterotopic transplant in Children
1 Advantages
1)
2)
3)
4)
Better use of donor organ
Suitability of procedure in high PVR
Survival not entirely dependent on donor organ
Possible recovery of the recipient heart
2 Disadvantages
1) Lack of wider experience and data
2) Technical problems related to the size
3) Doubt about the reversibility of the PVR
Heterotopic Heart Transplantation
Indications
1. Presence of fixed high PVR
2. Availability of undersized donor
3. Expectation of a certain degree of
recipient heart recovery
Transplantation Immunology
•
Primary immune response
1. Recognition of substance as nonself
2. Proliferation of immunocompetent cells
3. Effector phase
Antigen (HLA)
Most important antigen in human coded for the genes
of major histocompatibility complex and these genes
are present on the 6th chromosome
•
Antibody(immunoglogulin)
IgG, M, A, D, E
•
Complement system
Composed by protein and activated by classic pathway,
and alternative pathway
Immunologic Concepts
•
•
•
•
•
•
Immunological tolerance
Negative selection
Autograft
Isograft, syngeneic homograft
Allograft, homograft
Xenograft, heterograft
Special Immune Cells
•
T Cell
1. Cytotoxic T cell ; class I proteins of MHC (HLA-A,B,C)
2. Helper T cell ; class II protein of MHC (HLA-Dr)
3. T cell producing delayed hypersensitivity (T-DTH)
; class III protein of MHC (HLA-Dr)
4. Suppresser cell (Ts) ; release suppress factor
TH – OKT4, TS – OKT5
•
Killer or K-lymphocyte
Non B, non T cell, subpopulation of nonphagocytic monocyte.
K-cells have a receptor for the Fc portion of immunoglobulin and
are the effector cells of Ab-dependant cellular cytotoxicity.
Acute Cellular Rejection
ISHLT categories(Grade)
• G 0 ; No evidence of cellular rejection
• G 1A ; Focal perivascular or interstitial infiltrate
without myocyte injury
• G 1B ; Multifocal or diffuse sparse infiltrate without
myocyte injury
• G 2 ; Single focus of dense infiltrate with myocyte injury
• G 3A ; Multifocal dense infiltrates with myocyte injury
• G 3B ; Diffuse, dense infiltrates with myocyte injury
• G 4 ; Diffuse and extensive polymorphous infiltrate with
myocyte injury; may have hemorrhage, edema,
microvasvular injury
Accelerated Graft Rejection
Mechanism with elevated Troponin-T donor
1. Increased vascular permeability allowing
early post-transplant infiltration
2. Activation of nonspecific inflammatory
mediator
1) Lead to increased expression of donor
histocompatibility antigen
2) Increase in passenger leucocyte interact
aggressively with recipient lymphocyte
Class of Immunosuppressives
Use of Sirolimus
Methods of cyclosporin change
Common Immunosuppressives
1. OKT3 (murine monoclonal antibody)
Pan T-cell agent against the CD3 antigen on all T-cell
2. Antilymphocyte serum
3. Antithymocyte globulin
4. RATG (pan-anti T-cell globulin)
5. Monoclonal murine anti-T cell antibody
Immunosuppressive Agents
Class and Agent
Antiinflammatory Steroid
Adrenocorticosteroids
Action
Multiple mechanisms of action. Decrease production
of γ-interferon and interleukins, impair macrophage
function, and decease circulating lymphocytes.
Inhibitors of Interleukin-2
Cyclosporin A
Blocks production and release of interleukin-2, which
is essential for proliferation of cytotoxic and helper T
cells. Reduces interleukin-1 release from macrophages.
Tacrolimus
Blinds to T-cell binding protein to prevent synthesis of
interleukin-2 and other lymphokins.
Rapamycin
Investigational agent with action similar to cyclosporine
and tacrolimus.
Interleukin-2 Receptor Blockers
Daclizumab
Humanized interleukin-2 receptor blocker prevents
interleukin-2 to cytotoxic and helper T cells necessary
for cell proliferation.
Basiliximab(simulect)
Chimeric monoclonal antibody, single IV, 12mg/BSA,
on day 0, 4th for prophylaxis , not treatment
Immunosuppressive Agents
Class and Agent
Action
Inhibitors of Purine Biosynthesis
Azathioprine
Purine antimetabolite metabolized to 6-mercaptopurine,
which inhibits DNA and RNA synthesis
Methotrexate
Folic acid analog inhibits dihydrofolate reductase, thus
inhibiting purine synthesis.
Cyclophosphamide
A type of nitrogen mustard is activated by cytochrome
P450 in liver to form an alkylating species
cross-linking DNA
Mycorphenolate mofetil
Inhibits guanosine monophosphate synthesis primarily
in T and B lymphocytes, thereby inhibiting purine
synthesis DNA and RNA synthesis.
Immunosuppressant Gamma Globulin
OKT3 (anti-CD3 antibody) Murine monoclonal lgG antibody raised against the CD3
receptor-complex, which is present on 95% of all T
cells. Antibody binding causes both removal of receptors
from cells and removal of cells from the circulation.
Antithymocyte globulin
Polyclonal T-cell antibodies derived from injecting rabbits,
goats, or horses with human lymphocytes or
lymphoblasts. Antibodies formed are against a variety of
human lymphocytes.
Pediatric Immnusuppression
Pediatric Heart Transplant Immunosuppression-ISHLT,2001
Percentage of Children on Various
Immunosuppression Drugs
Posttransplant
CSA
Tacrolimus
Azathioprine
MMF
Prednisone
Discharge
80
15
75
15
75
Year 1
75
25
65
20
70
Year 3
75
25
55
25
50
Risk Factors for Rejection
Incremental Risk Factors
for a Rejection Episode
(Younger)
(Female)
(Female)
(Higher)
(Shorter)
(Increased)
Patient age at transplant
Gender
Patient-donor interaction
Gender of donor
Number of HLA mismatches
Procedural
Globat myocardial isochemic time
Immunosuppression
Trip drug + induction therapy
Posttransplantation
Interval since transplantation
No. of previous rejection episodes
Hazard Phase P value
Early
Constant
-
.001
.002
-
.002
.008
-
.01
.006
<.0001
<.0001
Pediatric Heart Transplantation
Results
Pediatric heart transplantation actuarial survival (1982-1999)
Pediatric Heart Transplantation
Results
Pediatric heart transplantation actuarial survival by era
Lung Transplantation
End-stage respiratory failure as a result of
•
•
•
•
Cystic fibrosis
Primary pulmonary hypertension
Interstitial lung disease
Congenital heart disease with Eisenmenger
syndrome
• Others such as obliterative bronchiolitis,
bronchopulmonary dysplasia, bronchiectasis
Donor Selection Criteria
Criteria for living donor lobar transplantation
Age < 55 years
No significant past medical history
No recent viral infections
Normal echocardiogram
Normal electrocardiogram
Normal chest radiograph
Oxygen tension > 80mm Hg on room air
FEV₁and FVC > 85% predicted
No significant pulmonary pathology on CT
No previous thoracic operation on donor side
Preservation of Heart & Lung
Single Left Lung Harvest
Single Left Lung Harvest
• The lung is triple bagged in a sterile plastic
container with iced saline slush for transport
Single Left Lung Transplantation
Anastomosis of donor pulmonary vein to recipient
left atrium, pulmonary artery and bronchus
Bronchiolitis Obliterans Syndrome
Scoring system
0. No significant abnormality: FEV₁>80% of baseline value
a. Without pathologic evidence of obliterative bronchiolitis
b. With pathologic evidence of obliterative bronchiolitis
1. Mild BOS: FEV₁66%-80% of baseline value
a. Without pathologic evidence of obliterative bronchiolitis
b. With pathologic evidence of obliterative bronchiolitis
2. Moderate BOS: FEV₁51%-65% of baseline value
a. Without pathologic evidence of obliterative bronchiolitis
b. With pathologic evidence of obliterative bronchiolitis
3. Severe BOS: FEV₁50% or less of baseline value
a. Without pathologic evidence of obliterative bronchiolitis
b. With pathologic evidence of obliterative bronchiolitis
Pulmonary Rejection
Classification & grading
A. Acute rejection-solitary or multiple
B*. Airway inflammation-lymphocytic
perivascular and interstitial
bronchitis/bronchiolitis
mononudear cell infitrates present
Grade B0: none
with/without B*
Grade B1: minimal
Grade 0:none
Grade B2: moderate
Grade 1: minimal (scattered infiltrates )
Grade B3: severe
Grade 2: mild (frequent infiltrates)
Grade BX: ungradeable
Grade 3: moderate (dense infiltrates)
Grade 4: severe (diffuse infiltrates)
C. Chronic airway rejection-bronchiolitis obliterans
Active (fibrosis with infiltrates)
Inactive (fibrous scarring without infiltrates)
D. Chronic vascular rejection-accelerated graft vascular sclerosis
Lung Transplantation
Immunosuppressive protocol
INTRAOPERATIVE
Methylprednisolone 15 mg/kg Ⅳ
Azathioprine 2.5mg/kg Ⅳ
POSTOPERATIVE
Atgam: day 1 = 15 mg/kg Ⅳ
day 2 = 10 mg/kg Ⅵ
day 3 = 7.5 mg/kg Ⅳ
Methylprednisolone 15 mg/kg/day Ⅳ in 3 divided doses;
when taking PO, then prednisone 0.5 mg/kg/day
Azathioprine 2 mg/kg Ⅳ and then switch to PO
Cyclosporine (Neoral) 10-20 mg/kg/day PO divided in two doses
if patient older than 6 years of age, three doses if younger than
6 years of age