* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download The Anatomical Substrates of Wolff-Parkinson-White
Survey
Document related concepts
Coronary artery disease wikipedia , lookup
Cardiac contractility modulation wikipedia , lookup
Hypertrophic cardiomyopathy wikipedia , lookup
Management of acute coronary syndrome wikipedia , lookup
Myocardial infarction wikipedia , lookup
Quantium Medical Cardiac Output wikipedia , lookup
Cardiac surgery wikipedia , lookup
Lutembacher's syndrome wikipedia , lookup
Atrial fibrillation wikipedia , lookup
Electrocardiography wikipedia , lookup
Heart arrhythmia wikipedia , lookup
Ventricular fibrillation wikipedia , lookup
Dextro-Transposition of the great arteries wikipedia , lookup
Arrhythmogenic right ventricular dysplasia wikipedia , lookup
Transcript
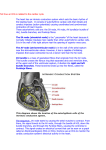
870 CIRCULATION Downloaded from http://circ.ahajournals.org/ by guest on June 16, 2017 16. Moore EN, Hoffman BF, Patterson DF, Stuckey JH: Electrocardiographic changes due to delayed activation of the wall of the right ventricle. Am Heart J 68: 347, 1964 17. Rosenbaum MB, Corrado G, Oliveri R, Castellanos A Jr, Elizari MV: Right bundle branch block with left anterior hemiblock surgically induced in tetralogy of Fallot. Am J Cardiol 26: 12, 1970 18. Bocala RR, Guller B, Danielson GK, Feldt RH: Left anterior hemiblock and complete repair of tetralogy of Fallot. (abstr) Pediatr Res 8: 347, 1974 19. Sonheimer HM, Izukawa T, Olley PM, Trusler GA, Mustard WT: Conduction disturbances after total correction of tetralogy of Fallot. Am Heart J 92: 278, 1976 20. Yabek SM, Jarmakani JM, Roberts N: Postoperative trifascicular block complicating tetralogy of Fallot repair. Pediatrics 58: 236, 1976 21. Boxer R, Krongrad E, Bowman FO Jr, Malm JR, Gersony WM: Conduction defects following ventricular septal defect closure with and without a right ventriculotomy. Pediatr Res 11: 386, 1977 22. Okoroma EO, Guller B, Maloney JD, Weidman WH: Etiology of right bundle branch block pattern after surgical closure of ventricular septal defects. Am Heart J 90: 14, 1975 23. Antar RA, Krongrad E, Bowman FO Jr, Malm JR: Etiology of right bundle branch block pattern following surgical repair of A-V cushion defects. Pediatr Res 10: 310, 1976 24. Kittle CF, Santos EM, Dimond EG: Persistent right bundle branch block due to pulmonic valvotomy and infundibulectomy. Am Surg 22: 80, 1956 25. Downing JW, Kaplan S, Bore KE: Post surgical left anterior hemiblock and right bundle branch block. Br Heart J 34: 263, 1972 26. Watt TB, Pruitt RD: Focal lesions in the canine bundle of His: Their effects on ventricular excitation. Circ Res 31: 351, 1972 VOL 57, No 5, MAY 1978 27. Krongrad E, Rosen MR, Merker C, Hoffman BF: Creation of counterclockwise superiorly oriented frontal plane loops in isolated blood perfused canine hearts. Pediatr Res 7: 300, 1973 28. Sugiura M, Okada R, Keisuke H, Shinichiro 0: Histological studies on the conduction system in 14 cases of right bundle branch block associated with left axis deviation. Jap Heart J 10: 121, 1969 29. Squarcia U, Merideth J, McGoon DC, Weidman WH: Prognosis of transient atrioventricular conduction disturbances complicating open heart surgery for congenital heart defects. Am J Cardiol 28: 648, 1971 30. Krongrad E, Rosen MR, Merker C, Fenoglio J, Hoffman BF: Creation of trifascicular block in isolated blood perfused canine hearts. (abstr) Circulation 48 (suppl IV): IV-21, 1973 31. El-Said G, Rosenberg HS, Mullins CE, Hallman GL, Cooley DA, McNamara DG: Dysrhythmias after Mustard's operation for transposition of the great arteries. Am J Cardiol 30: 526, 1972 32. Waldo AL, Krongrad E, Bowman FO Jr, Kaiser GA, Husson GS, Malm JR: Electrophysiological considerations during total repair of transposition of the great vessels. Circulation 45 & 46 (suppl II): 11-34, 1972 33. Clarkson PM, Barratt-Boyes BG, Neutze JM: Late dysrhythmias and disturbances of conduction following Mustard operation for complete transposition of the great arteries. Circulation 53: 519, 1976 34. Stanton RE, Lindesmith GG, Meyer BW: Arrhythmias following the Mustard operation for transposition of the great vessels. (abstr) Circulation 45 & 46 (suppl II): 11-225, 1972 35. James FW, Kaplan S, Chou T-C: Unexpected cardiac arrest in patients after surgical correction of tetralogy of Fallot. Circulation 52: 691, 1975 36. James FW, Kaplan S, Schwartz DC, Chou T-C, Sandker ET, Naylor V: Response to exercise in patients after total surgical correction of tetralogy of Fallot. Circulation 54: 671, 1976 The Anatomical Substrates of Wolff-Parkinson-White Syndrome A Clinicopathologic Correlation in Seven Patients ANTON E. BECKER, M.D., ROBERT H. ANDERSON, M.D., M.R.C. PATH., DIRK DURRER, M.D., AND HEIN J. J. WELLENS, M.D. SUMMARY Clinicopathological correlations were made on the hearts from seven patients known to have exhibited electrocardiographic evidence of the Wolff-Parkinson-White syndrome. In each case, clinical and pathological investigations were conducted independently, neither group of investigators having knowledge of the other's results. In all seven hearts, the entire atrioventricular junctions were serially sectioned. Accessory atrioventricular connections were predicted in all seven cases following electrocardiographic investigation. Connections were identified histopathologically in four hearts in the predicted site. In another case two connections were identified, one being considered responsible for the pre-excitation. In the sixth case a right lateral connection was anticipated, but only accessory nodo-ventricular fibers were identified following histopathologic studies. In the final case, a posterior septal connection was predicted but the entire septum had fibrosed following previous operation. These findings are discussed in the light of the investigative techniques used, the theories of pre-excitation and the embryogenetic mechanisms producing accessory atrioventricular connections. MANY CLINICAL AND ELECTROPHYSIOLOGICAL STUDIES of the Wolff-Parkinson-White (WPW) syndrome exist; in comparison, relatively little is known of the underlying pathology. Indeed at present there is still a dispute regarding the precise anatomy that underlies the abnormal ventricular activation pattern found in this syndrome. Paladino' and Kent2 had described myocardial fibers in normal hearts that connected atrial and ventricular myocardium which they considered to be responsible for normal atrioventricular conduction. However, these findings could not be considered to represent normality when Tawara3 demonstrated the existence of the atrioventricular node and its connection with the atrioventricular bundle,4 and then Hering' demonstrated experimentally that in the dog heart this bundle was the only conducting connection between atria and ventricles. In their classic study on the WPW syndrome Holzmann and Scherf 6 suggested two possible mechanisms, one of which assumed the existence of conducting muscular bridges between atrial and ventricular myocardia. Considerable time elapsed before such accessory pathways were actually demonstrated in a patient with WPW by Wood, Wolferth and Geckeler.7 Since then the number of cases in which these accessory connections have From the Departments of Pathology and Cardiology and Clinical Physiology, Wilhelmina Gasthuis, University of Amsterdam, Amsterdam, The Netherlands. Dr. Anderson is a British Heart Foundation Senior Research Fellow. His present address is Department of Pediatrics, Brompton Hospital, London, England. Address for reprints: Dr. Anton E. Becker, Department of Pathology, Wilhelmina Gasthuis, Eerste Helmersstraat 104, Amsterdam, The Netherlands. Received October 26, 1977; revision accepted November 28, 1977. ANATOMICAL SUBSTRATES OF WPW/Becker et al. 871 TABLE 1. Clinical and Electrophysiological Data of the Patients Studied Patient Age (yr) Sex 1 31 F 2 3 19 55 M M 4 45 M Mode of death Type of arrhythmia During surgery Atrial fibrillation, circus movement, tachycardia Syncopal attacks Atrial fibrillation, circus movement, tachyeardia Atrial fibrillation, circus movement, Downloaded from http://circ.ahajournals.org/ by guest on June 16, 2017 34 60 F M Atrial fibrillation Atrial fibrillation 7 61 F Atrial fibrillation, circus movement, tachyeardia been identified has considerably increased, so that at present approximately 25 well-documented cases have been reported.8 This still remains a small number when compared to the vast clinical experience of the syndrome. Precise knowledge concerning the anatomic substrates of the syndrome is desirable, because apart from the concept of an accessory pathway as an explanation of the abnormal ventricular activation pattern, many alternative mechanisms have been postulated, one of the most interesting of which is synchronized sino-ventricular conduction.9-" In view of this problem it seemed appropriate to report our study of a series of patients with WPW who died and in whom a morphologic study of the heart was undertaken. Suddenly 2 weeks post surgery Sudden Cardiogenic shock following anteroseptal MI Suddenly 3 yr after ligation of bundle of His I 1 2 3 4 5 6 7 I III TABLE 2. Predicted Localization of Accessory Pathway on ECG and Anatomic Localization ECG localization III Anatomic localization Anterior paraseptal 1. Left posterior (working myocardium) 2. Right anterior paraseptal (specialized) Left posterior Left posterior close to septum Left posterior Left posterior close to septum Right posterior septal Right septal ? Right lateral Left lateral Left lateral ? Left septal AVR lmv FIGURE 1. ECG from patient 1. On the basis of the criteria described by Frank"2 and Tonkin et al.13 the pre-excitation in this patient was predicted to be due to an accessory atrioventricular connection in right paraseptal position. The morphologicalfindings are illustrated in figures 7 and 8. Aortic incompetence, Ebstein's disease Hypertension (ECG) showing pre-excitation were available (figs. 1-5). Based upon the ECG the most likely site of the accessory pathway was predicted without knowledge of the anatomic findings, using the criteria of Frank12 and Tonkin et al.'3 (table 2). In a similar fashion, the histopathological studies in each case were conducted without prior knowledge of the results of clinical investigation. Materials and Methods Seven hearts from patients suffering from the WPW syndrome were studied. Clinical and electrocardiographic data are given in table 1. From all patients electrocardiograms Patient Ebstein's disease Sudden Sudden tachycardia 5 6 Additional abnormalities IAF 200msec VI VOL CIRCULATION 872 I -J.-- VIi V, ,-i I 57, No 5, MAY 1978 v2-w a A I III A AVR AVL AVF - 1 V2 V2 V3 V3 III V3 V54 AVR V4 V5' AVL v4JV-. AVR V5A AVL V6 AVF JL _>,V^ V2 4 V5 AVF I 1 fs Downloaded from http://circ.ahajournals.org/ by guest on June 16, 2017 200 msec Patient 2 Patient 3 Patient 4 FIGURE 2. ECGs from patients 2, 3 and 4. In patient 2, using the criteria of Frank12 and Tonkin et al.,'3 an accessory connection was predicted to exist in left posterior position. Morphologicfindings in this patient are illustrated in figures 7 and 9A. In patient 3, an accessory connection was again anticipated in left posterior position, and the bundle thus identified is illustrated in figure 10. A right septal accessory connection was predicted to exist in patient 4; the morphology of the connection discovered in this site is illustrated in figure 11. In all cases, both right and left atrioventricular (A-V) rings, together with the A-V septal area containing the A-V specialized conducting tissues were studied. The junctional regions were separated into multiple blocks, each of which was embedded for histologic serial sectioning. In each heart the number of blocks cut varied from 14 to 20 (fig. 6). The blocks were embedded in paraffin and serially sectioned at 7,u thickness in a plane at right angles to the atrioventricular junction. All the sections were retained in trays. As a routine, every fiftieth section was mounted; the remaining sections were studied, when deemed necessary, after examination of the initially mounted series. The sections were stained with hematoxylin and eosin, an elastic tissue stain counterstained with the van Gieson stain or with Masson's trichrome stain. Case Reports Patient 1. A 3 1-year-old woman was investigated because of atrial fibrillation and circus movement tachycardia. Study of the ECG suggested presence of a right, probably anterior septal accessory connection (fig. 1). At operation, the patient's penetrating atrioventricular (His) bundle was ligated, but she died during this procedure. The autopsy demonstrated the presence of Ebstein's malformation of the tricuspid valve. Histologic examination showed a marked deficiency of the atrioventricular fibrous anulus both in the septal area and posteriorly to the right. However, atrial and ventricular myocardium remained separated by adipose tissue throughout the whole area. The I II - t 1 V2 V3 III AVR V4 AVL V5_ AVF VeJ FIGURE 3. ECG from patient 5. A right lateral accessory connection was anticipated (fig. 7). No such connection was identified following morphological studies. V, 200 msec tv ANATOMICAL SUBSTRATES OF WPW/Becker et al. I 873 AVR AVR_v I AVL II 11 AVF III AVF III V3 Downloaded from http://circ.ahajournals.org/ by guest on June 16, 2017 V1 , 4 i' V4 V2 V5 V2 a V FIGURE 4. ECG from patient 6. A left lateral connection was predicted (fig. 7). The connection identified in this site is illustrated in figure 9B. FIGURE 5. ECG from patient 7. A septal connection on the left side was expected. Morphological studies revealed that the entire septal area had fibrosed following operation (fig. 7). mv v Epi FIGURE 6. Plane view of the atrioventricular junctional region viewed from above showing how the entire atrioventricular junction was divided into a series of blocks. The septal area, containing the specialized atrioventricularjunctional area (dotted tissue) was removed in three blocks. A typical section of this area is shown in the middle inset. The right and left parietal junctions were then divided into 5 10 blocks on each side depending upon the size of the heart. Sample sections of the parietal junctions are shown in the insets. AM = atrial myocardium; VM = ventricular myocardium; MV = mitral valve; TV = tricuspid valve; Epi = epicardium; PA = pulmonary artery. The anulusfibrosus is the cross-hatched tissue. - VOL 57, No 5, MAY 1978 CI RCULATION 874 7 Cases - Accessory Connections Casel ECG Right Septal Case 2 ECG Left Posterior Case 3 ECG Left Posterior Ant L Post Case4 ECG Right Septal Downloaded from http://circ.ahajournals.org/ by guest on June 16, 2017 Case 5 ECG Right Lateral Case 6 ECG Left Lateral Case 7 ECG Left Septal FIGURE 7. Diagram showing the correlation between electrocardiographic and morphologic findings in each of the seven patients studied. The meaning of the symbols is given in the key (A VA C = atrioventricular accessory connection). The ECG of patient I is shown in figure 1, the histological findings in figure 8. The ECGs of patients 2, 3 and 4 are illustrated in figure 2, and those of patients 5, 6 and 7 in figures 3, 4 and 5, respectively. The histological findings from patient 2 are illustrated in figure 9A, patient 3 in figure 10; patient 4 in figure 11 and patient 6 in figure 9B. A-V node and penetrating bundle were extensively traumatized, their normal architecture being completely obliterated. Examination of the parietal junctional tissues revealed presence of two accessory atrioventricular connections (figs. 7 and 8). One of these was composed of working myocardium and was located in a left posterior site close to the crux of the heart. This bundle ran through the epicardial fat, close to a well formed fibrous anulus (fig. 8A). The bundle had an overall length of 10 mm with a maximal diameter of I mm. At its ventricular side it divided into smaller W-1 .- -I-, -6. 1, FIGIURE 8. Morphological findings in patient /. A shows the left sided accessory connection (A C), which had not been anticipatedfrom the ECG. Note that the connection is on the epicardial aspect of a wellformed anulusfibrosus (A F), and joins atrial (AM) and ventricular (VM) myocardium by traversing the epicardial fat rather than by passing through a defect in the anulus. Note also the hemorrhage (H) following surgical intervention in the case. Figures 8B-D illustrate the second accessory connection, which was composed ofspecialized tissue. Figure 8B illu-strates the anterior node-like structure (in box) situated at the insertion of the atrial myocardium into the tricuspid valve (TV). A section further anterior (fig. 8C) at higher magnification shows that the anterior node (A N) is composed of a basket-like collection ofspecialized cells, and that the accessory connection (A C) originates from the node, the connection itself also being composed of specialized tissue. Figure 8D shows the course of the bundle in the tricuspid valve, lying alongside and ramifying into the ventricular myocardium in anterior paraseptal position. Note the lack of a discrete anulus fibrosus in this heart. ANATOMICAL SUBSTRATES OF WPW/Becker et al. IA I II aI Downloaded from http://circ.ahajournals.org/ by guest on June 16, 2017 FIGURE 9. Histological findings in patients 2 (fig. 9A) and 6 (fig. 9B). In patient 2, the accessory connection (arrows) was in the left posterior position; in patient 6, the connection (arrows) was in left lateral position. Note that in both cases the connections skirt a well formed an ulus fibros us (AIF) and course through the epicardial fat (EF) to join atrial (AM) and ventricular (VM) myocardia. Note also the bridging of the coronary artery (CA) in patient 6. MV mitral valve. bundles which became continuous with ventricular myocardium. The second connection was located in a right anterolateral position (fig. 7). A node-like structure was found at the insertion of atrial myocardium into the anulus (fig. 8B), being composed of cells typical of those found in the specialized atrioventricular junctional area (fig. 8C). They formed an interweaving, basket-like structure reminiscent of the normal compact atrioventricular node (fig. 8C). From this node-like structure a bundle originated composed of specialized cells, which coursed caudally toward the anterior tricuspid valve leaflet (fig. 81D). The latter exhibited dysplastic features, as commonly encountered in Ebstein's malformation, having myocardium present on its ventricular aspect. The ventricular extension of the specialized 875 accessory connection became continuous with the myocardium in the valve leaflet (fig. 8D). The overall length of the connection was 10 mm, with a maximal width of 1 mm. Patient 2. A 19-year-old man was first seen because of syncopal attacks. The site of the anticipated accessory atrioventricular connection was predicted to be left sided and posterior, close to the crux of the heart (fig. 2). The patient died suddenly at home. There were no abnormal findings during gross examination of the heart. Histopathological studies revealed an accessory atrioventricular connection located at the anticipated site (fig. 7). The bundle was composed of working myocardium. At its atrial end the bundle was well delineated with a width of slightly less than 1 mm. The bundle ran through epicardial adipose tissue, skirting a well formed fibrous anulus at its epicardial aspect (fig. 9A). Close to the ventricular myocardium, on its epicardial aspect, the bundle ramified into smaller bundles. The smaller bundles connected directly to the ventricular myocardium like the roots of a tree. The overall length of the connection was estimated at approximately 6 mm. There was deficiency of the fibrous anulus posteriorly in the septum, but no atrioventricular connection could be established at that site. The specialized atrioventricular junctional area was considered normal. Patient 3. A 55-year-old man first presented with signs and symptoms of a circus movement tachyeardia and atrial fibrillation. Following examination of the ECG, it was considered that an accessory atrioventricular connection would be present in a left posterior position, close to the crux of the heart (fig. 2). The patient died suddenly. Gross examination of the heart revealed normal anatomy. Histological studies demonstrated the presence of a connection at the anticipated site (fig. 7). The bundle was composed of working myocardium. At its atrial side the bundle was approximately 1 mm in width. It ran through epicardial adipose tissue, skirting the fibrous anulus (fig. 10). The latter was well formed. During its course the bundle was accompanied by a small artery and vein and a small nerve trunk (fig. 1OC). On reaching the ventricular myocardium the bundle ramified into K jk. . I. 1; j,. A q'i . I$ XfI' FIGURE 1 0. Series ofphotographs illustrating the left posterior connection identified in patient 3. Figure I OA illustrates the origin of the accessory connection (arrow) from the atrial myocardium. Note that it originates very close to a well formed anulusfibrosus and "points'" into the epicardial fat (A F). Figure lOB shows a section across the bundle (arrow) as it courses through the epicardialfat, skirting the anulus. The area within the box is shown in higher magnification infigure JOC. The accessory connection is a small bundle (B) of working myocardial cells, and is accompanied by a nerve (N), artery (A ) and vein ( V). Figure IOD shows the insertion of one "root" of the connection (arrow) into the ventricular myocardium (VM) some appreciable distance outside the well-formed anulus fibrosus. 876 CIRCULATION VOL 57, No 5, MAY 1978 Downloaded from http://circ.ahajournals.org/ by guest on June 16, 2017 FIGURE 1 1. Serial photographs of the septal accessory connection identified in patient 4. A is a posterior section showing the origin of the accessory connection (solid arrow) from the distal point of the right side of the interatrial septal myocardium (IAS). Note that the interatrial and interventricular septal (IVS) myocardia are separated only by epicardial fat (open arrows), the anulusfibrosus being deficient in this area. B, taken more anteriorly to A, illustrates the connection as afree running strand in the atrioventricular sulcus (solid arrow). C, again more anterior, shows the insertion of the connection into the ventricular mvocardium (solid arrow). several smaller bundles each of which anchored itself into ventricular myocardium (fig. lOD). The area of insertion had an approximate diameter of 1 mm. The overall length of the bundle was 10 mm. No other accessory connections were found; the A-V node and penetrating bundle were considered normal. Patient 4. A 45-year-old man with aortic insufficiency was investigated because of tachycardia. He was shown to have a circus movement tachycardia and atrial fibrillation. The findings were considered to indicate the presence of a right septal accessory atrioventricular connection (fig. 2). The patient's aortic valve was replaced by a ball valve prosthesis and at the same procedure the penetrating atrioventricular (His) bundle was ligated. The patient died suddenly two weeks after operation, following a previously uneventful postoperative period. At gross examination of the heart, the tricuspid valve was found to exhibit Ebstein's malformation. The anulus fibrosus in the septal area was highly deficient. Histopathological studies revealed the presence of an accessory atrioventricular connection, which was right sided and posterior to the orifice of the coronary sinus (fig. 7). The bundle was composed of working myocardium. At its atrial side it had a width of approximately 3 mm (fig. 1 IA). It coursed anteriorly from atrium to ventricle tapering down to a diameter of approximately 1.5 mm (fig. 1 B). Close to its ventricular insertion the bundle broke up into a number of small branches. The total length of the bundle was estimated at 5 mm. Throughout its course the bundle ran in subendocardial position, and passed through an area in which the fibrous anulus was completely deficient. Atrial and ventricular myocardium in this position were separated by only adipose tissue (fig. 11). Examination of the remaining junctional tissues failed to demonstrate any further connections. The area of the A-V node and penetrating bundle had been extensively traumatized by sutures. Patient 5. A 34-year-old woman had been known to have hypertension for many years. When first seen her electrocar- diogram revealed atrial fibrillation and a pattern typical for the WPW syndrome (fig. 3). The ECG was interpreted as being compatible with presence of an accessory atrioventricular connection in a right lateral position. The patient died suddenly at home before any further investigations could be undertaken. At autopsy, there were no abnormalities detected during gross examination of the heart. Histopathological studies revealed that the connections between atrial transitional cells and the compact node were normal. The penetrating bundle was normally formed, but there were extensive accessory nodo-ventricular connections.14 These were the only accessory connections found. The right and left parietal atrioventricular junctions exhibited the anticipated architecture with the right fibrous ring being deficient at many sites. Continuity between atrial and ventricular myocardium was never established. Because of the electrocardiographic interpretations the right lateral region was further studied by mounting all sections taken from that area. It still proved impossible to demonstrate any accessory connections. Patient 6. A 60-year-old man had been known to have atrial fibrillation for a prolonged time. The ECG was considered suggestive of a left lateral accessory atrioventricular connection (fig. 4). He also suffered from angina pectoris. As a consequence of his atherosclerotic disease acute anteroseptal myocardial infarction occurred and he died suddenly one week following onset of infarction. Gross examination of the heart confirmed the diagnosis of anteroseptal transmural myocardial infarction. Two vessel coronary artery disease was identified with multiple stenoses of more than 75% in the dominant right coronary artery. The left anterior descending coronary artery showed total occlusion in its proximal segment. Serial sectioning of the right and left atrioventricular rings revealed an accessory atrioventricular connection at the predicted left lateral position (fig. 7). The left circumflex artery, positioned in the ANATOMICAL SUBSTRATES OF WPW/Becker et al. Downloaded from http://circ.ahajournals.org/ by guest on June 16, 2017 atrioventricular sulcus, was extensively bridged at that site by atrial myocardium. From this bridging myocardium an accessory bundle of 1 mm diameter took origin (fig. 9B). It skirted the anulus fibrosus on its epicardial aspect and, just prior to its insertion into the ventricular myocardium, divided into two smaller bundles. The overall length of the connection was 3 mm. No other connections were found and the A-V node and penetrating bundle were considered normal. Patient 7. A 61-year old woman was known to have coronary artery disease. She also suffered from a circus movement tachycardia. Following study of the ECG, an accessory atrioventricular connection was predicted to be present posteriorly in the left side of the septum (fig. 5). At operation, her penetrating atrioventricular (His) bundle was ligated. Her postoperative electrocardiogram showed conduction over the anomalous pathway. She recovered uneventfully but died suddenly three years after the operation. Gross examination revealed considerable fibrosis of the septal junctional area. Histopathology revealed that the fibrosis had obliterated both the specialized junctional area and the posterior part of the septum. No accessory connections were identified (fig. 8). Discussion It is important to stress that the histopathological findings presently reported in each patient were derived without prior knowledge of the clinical and electrocardiographic data from that patient. Similarly, the interpretation of the electrocardiogram, using the criteria of Frank12 and Tonkin et al.,13 was made without any knowledge of the histopathological results. Bearing in mind this approach it is therefore of considerable significance that accessory atrioventricular connections were identified in five of seven hearts (fig. 7). A connection was considered to be present when atrial and ventricular myocardia became continuous with each other due to the interposition of a strand of muscle cells. In patients 2, 3, 4 and 6 a positive correlation was obtained with the ECG findings. In patient 1 the accessory connection was predicted to be in the anterior paraseptal region. The anatomical study revealed the presence of two connections, neither being in the precise situation. However, the specialized bundle originating from the anterior node-like structure in this patient coursed toward the anterior septum. This case is of considerable significance and will be discussed separately below. In the two remaining patients, our studies failed to demonstrate the presence of an accessory connection, although such connections had been anticipated following study of the ECG. In patient 5 the accessory connections had been predicted to be in a right lateral position. Even following retrospective study, we failed to demonstrate any connection at that site. Our retrospective study included the mounting of all sections from the different blocks covering the area. It must be realized, however, that this failure could easily be a consequence of the necessity to cut the specimen prior to histological sectioning. This immediately introduces the danger of cutting through a connection or destroying it by the proximity of the incision. This possibility was increased in our cases since the pathologist cutting the specimen was unaware of the predicted site of the connec- 877 tion. This could be regretted in retrospect but we remain convinced that our elaborate methodology has led to an unbiased interpretation of the histology. The heart of this patient did show extensive accessory nodo-ventricular (Mahaim) connections but the ECG was atypical for preexcitation of this type. It should therefore be questioned whether these connections in this particular case had any functional significance. In our experience such connections are frequently encountered in hearts of patients without signs of pre-excitation,15 although it must be emphasized that they have been directly implicated in another case following histopathological correlation.'6 In this latter instance, however, the ECG was not as expected for classical WPW pre-excitation. The final patient in whom we failed to demonstrate a connection had undergone surgery, ligation of the penetrating bundle having been performed (patient 7). In this case the predicted site of the connection had been the posterior left septal area. The operative procedure had led to fibrosis of the specialized junctional area, which extended posteriorly into the posterior part of the septum. It is surprising, therefore, that the electrocardiogram had shown conduction over the anomalous pathway, although we may speculate about progressive fibrosis eventually affecting the pathway itself and hampering the microscopic investigations. In summary, we strongly feel that failure to demonstrate an abnormal atrioventricular connection at the anticipated sites in these two hearts relates to the method of cutting blocks "in the blind" rather than to the existence of other mechanisms eliciting pre-excitation. It is important in this respect that no anatomical substrate could be detected to accept a phenomenon of synchronized sino-ventricular con- duction.9-"1 As indicated above, our case with dual accessory connections (patient 1) is of particular significance. In this heart, one of the connections was situated in the left posterior wall, being composed of ordinary working myocardium. The second connection was located in the anterolateral margin of the tricuspid orifice, and was composed of specialized cells. This connection took origin from an anteriorly located A-V node-like structure, which we believe represents a vestige of the anterior expansion of the specialized ring tissue present in the embryonic heart.17 Indeed, this node is remarkably similar to that described in a human heart as contacting the ventricular myocardium by Kent in 1914,1' although Kent did not describe a bundle originating from the node as in the present case. Furthermore, the node in this case is similar to those recently described in congenitally malformed hearts, notably corrected transposition19 and univentricular hearts.2'-22 The diameters of the cells composing the bundle which took origin from the ant r node were smaller than those of working myocardium and strikingly similar to those cells which make up a sizable portion of the A-V junctional specialized tissues." It must remain speculative as to whether this node and bundle could have displayed functional acteristics of specialized tissues. To the best of our knowledge only two previous connections have been reported to resemble specialized tissue. One of the connections reported by Verduyn Lunel was stated to be composed of Purkinje cells.22asecond case was reported by James et al. and a left lateral connection was said to be composed of p cells.24 It is significant, however, that the occurrence of a 878 CIRCULATION Downloaded from http://circ.ahajournals.org/ by guest on June 16, 2017 specialized connection such as that presently described had been predicted on the basis of previous morphological studies in embryos and adult human hearts.'7 The present case is of further interest, since in addition to the presence of dual accessory connections it also exhibited a deficient anulus fibrosus as a consequence of Ebstein's anomaly. Multiple accessory connections have previously been reported, both anatomically25 and clinically,26 and it has been indicated how this feature may hamper the electrocardiographic interpretation.28 In this case, we would suggest that the specialized connection was responsible for the preexcitation, since the distribution of the descending bundle would have involved the anterior segment of the right ventricular myocardium. It has previously been postulated that the accessory atrioventricular connections present in the WPW syndrome are the result of an embryologic fault in the formation of fibrous tissue separating both structures.23 This supposition has not been borne out by our data. Of the five hearts with identified connections there were four with a left-sided bundle. Each of these four left-sided connections occurred in the presence of a well formed fibrous anulus. Indeed the fibrous ring was in no way different from that encountered in the normal heart. The bundle in each instance skirted the anulus on its epicardial aspect, albeit that the bundle spatially was more closely related to the inner surface of the heart rather than the epicardial surface (fig. 12). It is significant that careful review of the cases reported in the literature similarly demonstrates that the majority of described left sided connections skirt a well formed fibrous anulus. These findings indicate that the presence of left-sided bundles cannot simply be explained on the basis of faulty development of the fibrous ring. An alternative explanation for their existence is thus far lacking. In contrast to left-sided bridges, the single heart in our series with a right-sided septal connection exhibited this connection in subendocardial position. The heart also displayed an Ebstein's malformation and therefore does not necessarily reflect the usual configuration in the case of a right-sided connection. Careful review of the cases reporting this morphology of WPW cases with right-sided connections demonstrates that the majority of these connections had a subendocardial course. This should not be surprising, since the normal anatomy of the right atrioventricular fibrous anulus is quite different from the left. The left sided anulus is usually well formed, and nearly always complete. The rightsided anulus, on the other hand, is poorly formed and deficient at many sites (Becker AE, Anderson RH, unpublished observations). From this anatomy one may anticipate the existence of right-sided accessory connections which indeed pass through an area of incomplete development. In conclusion, our study demonstrates that the electrocardiogram can be a good indicator of the site of the accessory connection when employing the criteria of Frank22 and the Duke University investigators,13 and when the accessory connection contributes significantly to ventricular activation." However, we would hesitate to use the predicted sites as a basis for surgery without having the facility for further electrophysiological studies before and during operation. It is noteworthy that the incision suggested by Sealy et al.28 to sever left-sided accessory connections probably VOL 57, No 5, MAY 1978 ANULUS 'CONNECTION FIGURE 12. Diagrammatic representation of left-sided accessory atrioventricular connections, based on our four identified connections (patients 1, 2, 3 and 6) and others reported in literature. It is important to note that the connections skirt through the epicardial fat, being outside a well formed anulus fibrosus. However, being close to the anulus they are still closer to the endocardial rather than the epicardial surfaces of the heart. It is also important to appreciate that, owing to the proximity of connection to anulus, the surgical incision advocated by Sealy et al.28 probably divides only the atrial input to the connection. As recently suggested by Sealy et al.29 in order to divide the connection itself, it will be necessary to scrape away the fat from the epicardial aspect of the anulus, removing the connection at the same time. divides the atrial input to the connection rather than the connection itself (fig. 12). This fact and the possibility of multiple pathways has led Sealy et al.28 to advocate an extensive incision. Moreover, in order to divide the ventricular connection itself, Sealy and co-workers29 recently proposed dissecting all fat tissue and vessels away from the ventricular surface. Our observations, which revealed atrioventricular connections skirting the anulus fibrosus, endorse the need for such a meticulous method (fig. 12). Finally, it should be emphasized that in two of our cases, Ebstein's malformation was discovered only at autopsy, and the failure of its earlier demonstration may well have contributed to the dismal outcome in these patients. We would therefore advocate rightsided angiography as a preoperative study in all patients scheduled for the WPW syndrome. Acknowledgment We are indebted to Miss H.J. Dijk and Messrs E.M.E. Heeren and M.J. Klaver for their technical assistance; to Mr. R.H. Verhoeven for photographic assistance and to Miss M.I. Schenker for secretarial assistance. References 1. Paladino G: Contribuzione all'anatomia, istologia e fisiologia del cuore. Mov Med-Chir (Napoli) 8: 428, 1876 2. Kent AFS: Researches on the structure and function of the mammalian heart. J Physiol 14: 233, 1893 3. Tawara S: Das Reizleitungssystem des Sauigetierherzens. Eine anatomischhistologische Studie fiber das Atrioventrikularbiundel und die Purkinjeschen Faden. Gustav Fisher, Jena, 1906 4. His W Jr: Die Thatigkeit des embryonalen Herzens und deren Bedeutung ANATOMICAL SUBSTRATES OF WPW/Becker et al. 5. 6. 7. 8. 9. 10. 11. 12. 13. Downloaded from http://circ.ahajournals.org/ by guest on June 16, 2017 14. 15. 16. fur die Lehre von Herzbewegung beim Erwachsenen. Arbeit aus der medicin Klinik zu Leipzig: 14, 1893 Hering HE: Nachweis dass die Verzogerung der Erregungsulberleitung zwischen Vorhof und kammer des Sauigetierherzens im Tawara, schen Knoten erfolgt. Pfluger Arch Ges Physiol 131: 572, 1910 Holzmann M, Scherf D: Ueber Elektrocardiogramme mit verkurzter Vorhof-Kammer-Distanz und positiven P-Zacken. Z Klin Med 121: 404, 1932 Wood FC, Wolferth CC, Geckeler GD: Histologic demonstration of accessory muscular connections between auricle and ventricle in a case of short P-R interval and prolonged QRS complex. Am Heart J 25: 454, 1943 Frank R, Brechenmacher C, Fontaine G: Apport de l'histologie dans l'etude des syndromes de pre-excitation ventriculaire. Coeur Med Int 15: 337, 1976 James TN: The Wolff-Parkinson-White syndrome: Evolving concepts of its pathogenesis. Prog Cardiovasc Dis 13: 159, 1970 Sherf L, James TN: A new electrocardiographic concept: Synchronized sinoventricular conduction. Dis Chest 55: 127, 1969 Sherf L: The atrial conduction system: Clinical implications. Am J Cardiol 37: 814, 1976 Frank R: Apport des investigations endocaitaires et des cartographies epicardiques dans l'etude des syndrome de pre-excitation ventriculaire. Thesis, Paris, 1974 Tonkin AM, Wagner GS, Gallagher JJ, Cope GD, Kasell J, Wallace AG: Initial forces of ventricular depolarization in the Wolff-Parkinson-White syndrome. Circulation 52: 1030, 1975 Anderson RH, Becker AE, Brechenmacher C, Davies MJ, Rossi L: Ventricular pre-excitation. A proposed nomenclature for its substrates. Eur J Cardiol 3: 27, 1975 Becker AE, Anderson RH: Morphology of the human atrioventricular junctional area. In The Conduction System of the Heart, edited by Wellens HJJ, Lie KI, Janse MJ. Leiden, HE Stenfert Kroese BV, 1976, chapter 15 Lev M, Fox SM, Bharati S, Greenfield JC, Rosen KM, Pick A: Mahaim and James fibers as a basis for a unique variety of ventricular preexcitation. Am J Cardiol 36: 880, 1975 879 17. Anderson RH, Davies MJ, Becker AE: Atrioventricular ring specialized tissue in the normal heart. Eur J Cardiol 2: 219, 1974 18. Kent AFS: The right lateral auriculo-ventricular junction of the heart. J Physiol 48: 22, 1914 19. Anderson RH, Becker AE, Arnold R, Wilkinson JL: The conducting tissues in congenitally corrected transposition. Circulation 50: 911, 1974 20. Anderson RH, Arnold R, Thapar MK, Jones R, Hamilton DI: Cardiac specialized tissue in hearts with an apparently single ventricular chamber (double inlet left ventricle). Am J Cardiol 33: 95, 1974 21. Bharati S, Lev M: The course of the conduction system in single ventricle with inverted (L-) loop and inverted (L-) transposition. Circulation 51: 723, 1975 22. Wilkinson JL, Anderson RH, Arnold R, Hamilton DI, Smith A: The conducting tissues in primitive ventricular hearts without an outlet chamber. Circulation 53: 930, 1976 23. Verduyn Lunel AA: Significance of annulus fibrosus of heart in relation to AV conduction and ventricular activation in cases of Wolff-ParkinsonWhite syndrome. Br Heart J 34: 1263, 1972 24. James TN, Puech P: De subitaneis mortibus IX. Type A WolffParkinson-White syndrome. Circulation 50: 1264, 1974 25. Deerhake HG, Kimball JL, Burch GE, Henthorne JC: Wolff-ParkinsonWhite syndrome: Histologic study of the cardiac septum and atrioventricular groove in one case. Quoted by Kimball JL, Burch GE: Ann Intern Med 27: 239, 1947 26. Denes P, Amat-y-Leon F, Wyndham C, Wu D, Levitsky S, Rosen K: Electrophysiological demonstration of bilateral anomalous pathways in a patient with Wolff-Parkinson-White syndrome (Type B pre-excitation). Am J Cardiol 37: 93, 1976 27. Wellens HJJ: Contribution of cardiac pacing to our understanding of the Wolff-Parkinson-White syndrome. Br Heart J 37: 231, 1975 28. Sealy WC, Wallace AG, Ramming KP, Gallagher JJ, Svenson RH: An improved operation for the definitive treatment of the Wolff-ParkinsonWhite syndrome. Ann Thorac Surg 17: 107, 1974 29. Sealy WC, Gallagher JJ, Wallace AG: The surgical treatment of WolffParkinson-White syndrome: Evolution of improved methods for identification and interruption of the Kent bundle. Ann Thorac Surg 22: 443, 1976 The anatomical substrates of wolff-parkinson-white syndrome. A clinicopathologic correlation in seven patients. A E Becker, R H Anderson, D Durrer and H J Wellens Downloaded from http://circ.ahajournals.org/ by guest on June 16, 2017 Circulation. 1978;57:870-879 doi: 10.1161/01.CIR.57.5.870 Circulation is published by the American Heart Association, 7272 Greenville Avenue, Dallas, TX 75231 Copyright © 1978 American Heart Association, Inc. All rights reserved. Print ISSN: 0009-7322. Online ISSN: 1524-4539 The online version of this article, along with updated information and services, is located on the World Wide Web at: http://circ.ahajournals.org/content/57/5/870 Permissions: Requests for permissions to reproduce figures, tables, or portions of articles originally published in Circulation can be obtained via RightsLink, a service of the Copyright Clearance Center, not the Editorial Office. Once the online version of the published article for which permission is being requested is located, click Request Permissions in the middle column of the Web page under Services. Further information about this process is available in the Permissions and Rights Question and Answer document. Reprints: Information about reprints can be found online at: http://www.lww.com/reprints Subscriptions: Information about subscribing to Circulation is online at: http://circ.ahajournals.org//subscriptions/