* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Inborn Errors of Metabolic Etiology
Butyric acid wikipedia , lookup
Metabolomics wikipedia , lookup
Biosynthesis wikipedia , lookup
Citric acid cycle wikipedia , lookup
Metabolic network modelling wikipedia , lookup
Wilson's disease wikipedia , lookup
Basal metabolic rate wikipedia , lookup
Glyceroneogenesis wikipedia , lookup
Pharmacometabolomics wikipedia , lookup
Biochemistry wikipedia , lookup
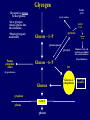
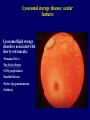
Inborn Errors of Metabolism Michael Marble, MD Professor of Clinical Pediatrics Division of Clinical Genetics Department of Pediatrics, LSUHSC and Children’s Hospital A 3 day old male is brought to the emergency room with a history of lethargy progressing to unresponsiveness. You take an initial history which reveals that the baby had been feeding normally for 24 hours but thereafter became irritable and lost interest in feeding. On exam, you notice that he is breathing fast and deep and is unresponsive. Along with other possible diagnoses, you suspect metabolic disease. (1) Which laboratory studies would you order to obtain quick evidence for or against metabolic disease? (2) You obtain a complete metabolic profile which shows a normal result. Urinalysis shows elevated specific gravity but is otherwise normal. Capillary blood gas shows respiratory alkalosis: 7.53/ pCO2 20/HCO3 nl, BE nl (3) Based on these results, what type of metabolic disease is most likely? Which test would you order next? Urea cycle disease; plasma ammonia (4) Plasma ammonia result is 1400 micromole/L (0-80). What is the most likely diagnosis? Which tests would you send to confirm a specific metabolic disorder? Ornithine transcarbamylase deficiency. Plasma amino acids, urine orotic acid (5) You confirm that the patient has ornithine transcarbamylase deficiency. What is the recurrence risk in the next pregnancy? Who else in the family should be tested? X-linked inheritance therefore 50% recurrence risk if mother is a carrier. (6) What is the treatment? Hemodialysis, low protein diet, arginine, phenylbutyrate Urea Cycle Disorders DIET Protein NH4+ + Hyperammonemia without metabolic acidosis (usually have respiratory alkalosis) Ornithine •Carbamoyl phosphate synthase deficiency (AR) •Citrullinemia (AR) OTC Citrulline UREA Urea cycle disorders: •Ornithine transcarbamylase deficiency (X-linked) Carbamoyl Phosphate HCO3 Arginine CYCLE urea(2N) Argininosuccinic Acid •Argininosuccinic acidemia (AR) •Argininemia (AR) OTC deficiency is the most common and is Xlinked Asp (N) Headaches, recurrent vomiting, avoids meat X-linked inheritance, partially affected female A 3 day old male is brought to the emergency room with a history of lethargy progressing to unresponsiveness. You take an initial history which reveals that the baby had been feeding normally for 24 hours but thereafter became irritable and progressively less interested in feeding. On exam, you notice immediately that he is breathing fast and deep and is unresponsive. Along with other possible diagnoses, you suspect metabolic disease. (1) Which laboratory studies would you order to obtain quick evidence for or against metabolic disease? (2) You obtain a blood gas, basic metabolic profile, urinalysis and plasma ammonia which show the following: 136 101 26 96 4.8 10 0.7 Capillary blood gas: UA 3+ ketones 7.11/CO2 19, HCO3 9, BE - 11 (3) Based on these results, what type of metabolic disease is most likely? Organic acidemias (this patient has propionic acidemia) (4) How would you confirm a specific metabolic disorder in this case? Urine organic acids, plasma acylcarnitine profile Ammonia 646 (0-36) Organic Acids Anabolic Catabolic ATP propionic acidemia Isoleucine Valine methylmalonic acidemia biotin Propionyl CoA Methionine Cholesterol B12 Methylmalonyl CoA Succinyl CoA Bicarb is used to buffer the propionic acid, leading to increased anion gap Odd chain fatty acids Krebs Cycle isovaleric acidemia leucine Isovaleryl CoA 3MCC HMG CoA Acetyl CoA ETS Even chain fatty acids Lysine Tryptophan glutaric acidemia Glutaryl CoA Crotonyl CoA Acetyl CoA Organic acids are the intermediates in the catabolism of amino acids, lipids and other compounds; specific enzyme deficiencies lead to characteristic urine organic acid profiles ATP Organic acids are metabolized in the mitochondria; blocks in their metabolism lead to elevation of specific acylcarnitines which are identified by plasma acylcarnitine profile Long chain fatty acid Fatty acid Detected by acylcarnitine profile Fatty acyl-CoA Free carnitine Fatty acyl-carnitine Propionyl CoA Fatty acyl-carnitine CoA propionylcarnitine Free carnitine acetyl CoA Fatty acyl-CoA Fatty acid oxidation Mitochondrion Plasma Cytoplasm ketones Krebs Selected Organic Acidemias Disease Cofactor Other features Wide anion gap ketoacidosis Propionic biotin Usually severe + Methylmalonic B12 Some respond to B12 + Isovaleric riboflavin Sweaty foot odor to urine + Glutaric riboflavin Macrocephaly, dystonia, Abnormal MRI + Maple syrup urine thiamine Maple syrup odor, elevated branched chain amino acids + Glutaric Acidemia Type 1 Severe movement disorder Glutaric acidemia type 1 (patient with viral illness) •Intercurrent illnesses (usually viral) greatly increase the risk of metabolic encephalopathy and long term disability; therefore preventive measures against catabolism are critical •The parents of organic acidemia patients should be given emergency protocols for management during intercurrent illnesses D10 + ¼ NS at 1.5 maintenance volume; IV carnitine Urea cycle disease versus organic acidemias UCD OA + + + ++ + +/- metabolic ketoacidosis - + primary respiratory alkalosis + - lethargy/coma vomiting hyperammonemia You are called to the newborn nursery regarding an 8 hour old female who is listless and not interested in feeding. The baby is severely hypotonic and lethargic but no other obvious abnormalities are noted. Accucheck shows normal glucose. Blood gas, complete metabolic profile, CBC, plasma ammonia, lactate and urinalysis all show normal results. Chest X-ray comes back normal. Along with other possibilities, you suspect a neuromuscular disorder and consult neurology. Maintenance IVFs are started. Pregnancy history is significant for decreased fetal movements. While awaiting neurology consult, the baby has apnea spells and develops myoclonic jerks. and is intubated. An EEG shows a “burst suppression” pattern. (1) What is the most likely diagnosis? Nonketotic hyperglycinemia (2) How would you confirm the diagnosis? CSF/plasma glycine ratio (3) What is the prognosis? Very poor, despite treatment Nonketotic hyperglycinemia *Defect in glycine catabolism •autosomal recessive •symptoms in first 24 hours •hypotonia/encephalopathy, seizures, burst suppression EEG •increased CSF/plasma glycine •Tx: benzoate, dextramethorphan •poor prognosis, diet ineffective *Diagnosis based on elevated CSF/Plasma glycine ratio Glycine NH3 + CO2 A 15 month old female, previously healthy, was brought to the emergency room after the mother had difficulty arousing her in the morning. Over the past 2 days, the child had had a low grade fever, cough, mild diarrhea and 3 episodes of vomiting. Due to poor appetite, the patient did not eat very much for dinner and missed her ususal bedtime snack the night before presentation. In the ER, she was noted to have a depressed mental status but was partially responsive. Exam was otherwise normal. Initial lab testing showed the following: CBC: WBC mildly elevated CMP shows sodium 139, Cl 104, CO2 13 BUN 28 Cre 0.6, glucose 37, mild elevation of ALT and AST Urinalysis negative for reducing substances and ketones, specific gravity is elevated The ER physician starts an IV and gives a bolus of glucose to correct hypoglycemia. The physician also gives normal saline boluses for rehydration. Then IVFs with D5 ¼ normal saline is started at 1.5 maintenance fluids. Followup labs show normal serum glucose but no change in acid-base status. The patient’s mental status worsens and she becomes comatose. She is transferred to the PICU. Plasma ammonia level is found to be mildly elevated at 101 micromoles/L . Patient who presented with hypoglycemia and altered mental status Based on the above presentation and lab results, the patient most likely has a disorder within which category of inborn error of metabolism? Fatty acid oxidation defects (specifically MCAD in this patient) How would you confirm a specific diagnosis? Plasma acylcarnitine profile Diagnosis of fatty acid oxidation disorders by acylcarnitine analysis Long chain fatty acid Fatty acid Detected by acylcarnitine analysis MCAD deficiency Fatty acyl-CoA Fatty acyl-carnitine Fatty acyl-carnitine Free carnitine + (C6-C12) fatty acyl CoAs (C6-C12) Fatty acyl-carnitine acetyl CoA Fatty acyl-CoA SCAD 18 16 14 12 8 6 4 MCAD Mitochondrion Plasma Cytoplasm ketones Fatty acid oxidation Brain CPT1/CPT2 Fatty acids VLCAD LCHAD MCAD fasting *key pathway for adaptation to fasting •Distinguishing feature of FAOD is hypoketotic hypoglycemia •Medium chain acyl CoA dehydrogenase deficiency(MCAD) is most common and has a 25% risk of death with first episode •LCHAD, VLCAD and carnitine uptake disorder are variably associated with, hepatomegaly, liver disease, hypertrophic cardiomyopathy and potential arrythmias •All are autosomal recessive SCAD ketones + acetyl CoA Krebs cycle LCHAD deficiency Hypoketotic hyoglycemia, hypotonia, failure to thrive At diagnosis On dietary treatment Variable Clinical presentations of fatty acid oxidation •Hyoketotic hypoglycemia in neonatal period •Later onset hypoketotic hypoglycemia •Sudden infant death syndrome •Hypertrophic cardiomyopathy, arrythmias •Liver disease •Adolescent or adult onset myopathy •Acute rhabdomyolysis •Asymptomatic Fatty acid oxidation disorders Disease Typical presentation SCAD Probably benign MCAD Hypoketotic hypoglycemia Most common FAOD, may be associated with “SIDS” VLCAD Variable: hypoketotic hypoglycemia, hypertrophic cardiomyopathy, myopathy, liver dz Extemely variable ranging from neonatal to adult onset LCHAD Variable: hypoketotic hypoglycemia, hypertrophic cardiomyopathy, myopathy, liver dz Extremely variable, need low fat diet Comments N/A Diagnosis is based on the specific pattern of acylcarnitine elevations Disorders of carnitine metabolism (1) Carnitine transports long chain fatty acids into the mitochondria (2) Carnitine deficiency can be primary or secondary (3) Primary carnitine deficiency is caused by abnormal transport of carnitine itself into the cells (carnitine uptake disorder, AKA “systemic carnitine deficiency”) (4) Secondary carnitine deficiency is caused by other metabolic disorders through the formation of carnitine esters (acylcarnitines) by abnormal organic/fatty acids Primary (CUD) Plasma: Decreased total carnitine Decreased free carnitine Normal acyl/free ratio Urine: Normal total carnitine MCAD, organic acidemias etc Plasma: Decreased/normal total carnitine Decreased free carnitine Increased acyl/free ratio Urine: Decreased/normal total carnitine Normal or increased free carnitine Decreased free carnitine Normal acyl/free ratio Increased acyl/free ratio A 6 day old female who is breast fed is brought to the emergency room due to poor feeding, vomiting and jaundice? Initial laboratory studies show the following: Total Bilirubin 19 136 115 26 4.8 10 0.7 73 Direct bilirubin 5.2 AST 987 ALT 767 Which metabolic disorder do you suspect? galactosemia Which other routine tests should you order? PT, PTT, urine reducing substances How would you confirm the diagnosis? Enzyme assay, DNA How would you treat this patient? Galactose free diet What are the acute and long term complications of this disorder? Liver disease, E coli sepsis, cataracts, MR, speech delay, ovarian failure Galactose Metabolism glucose Breast milk, cow’s milk Lactose (galactose-glucose) Galactose (cataracts) galactokinase epimerase (benign) Treatment: galactose free diet, ophthalmology and developmental followup Gal-1-P UDP glucose galactose-1-P uridyltransferase UDP galactose (classical) Glucose-1-P Glucose-6-P glycolysis pyruvate A 9 year old male is brought to the emergency room due to acute vomiting and lethargy shortly after a birthday party. Past medical history is significant for failure to thrive in late infancy which resolved without determination of a diagnosis. He had had several bouts of vomiting in the past, usually after consuming candy or soft drinks at parties. He has had no dental cavities. Laboratory results in the ER are as follows: Total Bilirubin 6.4 136 115 26 4.8 10 0.7 73 Direct bilirubin 5.2 What is the most likely metabolic diagnosis? Hereditary fructose intolerance AST 767 ALT 987 A 3 month old female is found to have hepatomegaly on routine exam. She is asymptomatic. Lab testing shows hypoglycemia, lactic acidemia, hyperuricemia, hyperlipidemia and elevated AST and ALT. What is the most likely diagnosis? Glycogen storage disease How would you confirm the diagnosis? DNA, liver biopsy What is the treatment? dietary Glycogen Storage Disease 1a “Von Gierke disease” Glycogen Storage Disease 1b facial features weakness hepatomegaly Hypoglycemia, lactic acidosis, hyperuricemia, hyperlipidemia, neutropenia Sibling with same disorder Autosomal recessive Glycogen Krebs cycle Glycogen is a storage form of glucose: Lactic acidosis •Liver glycogen releases glucose into the circulation •Muscle glycogen is used locally Acetyl CoA Glucose – 1- P pyruvate Malonyl CoA gluconeogenesis glycolysis Pentose phosphate shunt Stimulates fatty acid synthesis and inhibits fatty acid breakdown (Hyperlipidemia) Glucose – 6- P ER (hyperuricemia) Glucose cytoplasm Glut 2 plasma glucose Glucose-6phosphatase GSD types 1a and 1b Selected glycogen storage diseases Disease Typical presentation Von Gierke (GSDIa) Hepatomegaly, lactic acidosis, hyperuricemia, hyperlipidemia GSDIb Hepatomegaly, lactic acidosis, hyperuricemia, hyperlipidemia Pompei (GSD II) Weakness, hypotonia, cardiomyopathy Other features Puffy cheeks Puffy cheeks, neutropenia Treatment Nocturnal NG feedings, avoid fasting Nocturnal NG feedings, avoid fasting, neutropenia precautions EKG: short PR intervals, wide QRS Enzyme replacement Debrancher deficiency (GSD III) Similar to Von Gierke but milder, normal lactate Muscle, including cardiac may be involved Similar to GSD1a Brancher deficiency (GSD IV) Fatal liver disease (amylopectinosis) Other organ involvement ? transplant McCardle disease (GSD VI) Only muscle involvement Risk of rhabdomyolysis Avoid excess excercise Patient with developmental regression Apparently normal development for the first 6 months but begins to slow down. She was able to sit unassisted by 1 year. She was very socially interactive and could grasp objects. Gradually lost her ability to sit and grasp objects. Became less and less interactive, and lost interest in eating and became emaciated. She had splenomegaly. Ophthalmology exam revealed a cherry red spot macula: •What type of disorder do you suspect? Lysosomal storage disease •How would you confirm a diagnosis? Enzyme assay •What is the differential diagnosis of cherry red macula? Lysosomal storage disease: ocular features Lysosomal lipid storage disorders associated with cherry red macula: •Niemann-Pick A •Tay-Sachs disease •GM1 gangliosidosis •Sandhoff disease •Farber lipogranulomatosis •Sialidosis Cell membranes, organelles Bone, connective tissue, skin, cornea,joints etc Mucoploysaccharides (glycosaminoglycans) Sphingolipids, glycolipids etc Glycoproteins Glycogen Food particles Acid hydrolases Lysosome Abnormal lysosomal storage leads to developmental regression “The cells wrecking crew” Bacteria, viruses Metachromatic Leukodystrophy •Rapid developmental regression starting in late infancy •Lysosomal accumulation of sulfatides GM1 Gangliosidosis Neonatal presentation: hypotonia, ascites A 14 month old female presented with developmental delay to your clinic. She was reportedly normal at birth but at 8 months was noted to have mild kyphosis when sitting. She had chronic rhinorrhea. Late in infancy, the parents noticed gradual changes in craniofacial features including thickening of the eyebrows, large tongue, prominence of forehead. The patient hand been pulling to stand but lost this ability and seemed to be regressing in overall development. On exam, you notice a scaphocephalic head shape, frontal bossing, relatively thick eyebrows, cloudy cornea and stiff elbows. The patient most likely has a disorder within which category of inborn error of metabolism? Lysosomal storage disease (mucopolysaccharidosis) How would you confirm a specific diagnosis? Enzyme assay, urine mucopolysaccharies (glycosaminoglycans), skeletal survey Mucopolysaccharidosis • Hurler Syndrome: comparison with sibs Hurler syndrome Mucopolysaccharidosis • Hurler syndrome – alpha L-iduronidase def. organomegaly Sanfilipo Syndrome (MPS 3) • facial features •Sanfilipo (MPS III) •Less severe somatic features •Developmental delay •Behavioral problems •Neurological regression Maroteaux-Lamy (MPS VI) MaroteauxLamy syndrome (MPS6) Morquio (MPS IV) Lysosomal storage disease: laboratory diagnosis •Urine mucopolysaccharides •Urine oligosaccharide •Enzyme assay •DNA (for genetic counseling and to rule out pseudoalleles) Disease Typical presentation Hurler (MPS1) Developmental regression, dysosotosis multiplex, cloudy cornea, organomegaly, cardiac valve disease Hunter (MPS2) Similar to Hurler but no cloudy cornea San Filippo (MPS3) Later onset, mild somatic features Inheritance Treatment Autosomal recessive BMT/ERT X-linked BMT/ERT Autosomal recessive Morquio (MPS4) Mainly skeletal involvement Autosomal recessive Maroteaux-Lamy (MPS6) Similar to Hurler but “CNS sparing” Autosomal recessive ?ERT BMT/ERT One year old female with failure to thrive, developmental delay and hypotonia, MRI showed basal ganglia abnormalities. Labs show mild elevation of lactate. Mitochondrial genome sequencing: mutation m.8993T>G in a subunit of ATP synthase •Maternal inheritance •Heteroplasmy •Replicative segregation Mitochondrial genome disorders Mitochondrial genome disorders •Myoclonic epilepsy, lactic acidosis, stroke-like episodes (MELAS) •Myoclonic epilepsy ragged red fibers (MERRF) •Neuropathy, ataxia, retinintis pigmentosa (NARP) •Nonsyndromic deafness/diabetes •Kearn Sayres: sporadic giant deletions •Pearson syndrome: sporadic giant deletions •Leigh syndrome •other PKU Adult with Mental Retardation: born before newborn screening era PAH Dietary protein Phe Tyr Neurotransmitters, melanin etc •Phenylalanine hydroxylase defect •Autosomal recessive •Normal infant at birth Severe mental retardation, microcephaly, behavioural problems PKU: Clinical Problems if Untreated • mental retardation • seizures • hypopigmentation • rash Tx: low phenylalanine diet *Due to newborn screening, the above problems rarely occur. “Guthrie cards” Heel stick: •Obtain at about 48 hours •If obtained too early, false negative Filter paper with blood spots and demographic information Phenylketonuria Patients with PKU: low Phe diet, frequent monitoring of Phe, dietary counseling •Studies have shown that NBS has virtually eliminated mental retardation due to PKU Normal growth and development Selected Presentations/Diagnostic Considerations Lysosomal storage (glycolpids)) DEVELOPMENTAL REGRESSION ORGANOMEGALY CHERRY RED MACULA RESPIRATORY ALKALOSIS HYPERAMMONEMIA Lysosomal storage (MPS) DEVELOPMENTAL REGRESSION SKELETAL DYSPLASIA ORGANOMEGALY VARIABLE CLOUDY CORNEA ORGANIC ACIDEMIA HYPOGLYCEMIA HEPATOMEGALY INFANT/CHILD WITH SUSPECTED METABOLIC DISEASE GLYCOGEN STORAGE DISEASE (LIVER) HYPERCHLOREMIC METABOLIC ACIDOSIS LIVER DISEASE CATARACTS HYPERBILIRUBINEMIA REDUCING SUBSTANCES GALACTOSEMIA UREA CYCLE DISEASE WIDE ANION GAP METABOLIC ACIDOSIS, KETONURIA, HYPERAMMONEMIA WEAKNESS RHABDOMYOLYSIS GLYCOGEN STORAGE DISEASE (MUSCLE) Or FAOD KETONES NEGATIVE ENCEPHALOPATY < 24 HRS OLD, BURST SUPPRESSION EEG NON KETOTIC HYPERGLYCINEMIA METABOLIC ACIDOSIS HYPOGLYCEMIA INAPPROPRIATELY LOW KETONES FATTY ACID OXIDATION DEFECT