* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Living things are energy rich complex chemical structures
Isotopic labeling wikipedia , lookup
Photosynthetic reaction centre wikipedia , lookup
Nucleic acid analogue wikipedia , lookup
Carbon sink wikipedia , lookup
Basal metabolic rate wikipedia , lookup
Evolution of metal ions in biological systems wikipedia , lookup
Metalloprotein wikipedia , lookup
Photosynthesis wikipedia , lookup
Biosequestration wikipedia , lookup
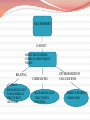
Living things are energy rich complex chemical structures BIOCHEMISTRY IS ABOUT ENERGY RICH COMPLEX CHEMICAL STRUCTURE OF THINGS RELATING 4 TYPES OF MACROMOLECULES TO BIOCHEMICAL STRUCTURE OF ORGANISMS CORRELATING MACROMOLECULAR STRUCTURE TO FUNCTION DETERMINEDED BY CALCULATIONS ENERGY IS STORED IN COMPOUNDS TERMS TO KNOW- BIOCHEMISTRY ORGANIC : relating to living things ORGANIC MOLECULES: composed of long chains or rings of carbon atoms attached to other elements like nitrogen, hydrogen, oxygen. MACROMOLECULE: A LARGE COMPLEX MOLECULE : EX. CARBOHYDRATES, LIPIDS, NUCLEIC ACIDS, PROTEINS ( these are organic molecules) Classes of Macromolecules Class sub units & composition Carbohydrates Sugars lipids Carbon Hydrogen water Fatty acids proteins Carbon Hydrogen And sometimes oxygen Amino acids Carbon Hydrogen Oxygen nitrogen functions/roles examples energy storage Structural components Starch, glycogen Cellulose, chitin Store energy Form membranes Steroid hormones Body fat Cell membrane testosterone Serve as enzymes Structural components Peptide hormones Lactase Hair, cartilage insulin Nucleic acids Nucleotide Carbon Hydrogen oxygen Nitrogen phosphorus -Store genetic DNA information RNA -Direct production of’ proteins THE PARTY ANIMAL OF THE PERIODIC TABLE- CARBON CARBON IS AN ATOM THAT CAN BOND IN A VARIETY OF WAYS WITH OTHER ATOMS. CARBON IS THE BUILDING BLOCK OF ALL LIVING THINGS (ORGANIC MOLECULES) THE HUMAN BODY IS ABOUT 18% CARBON BY WEIGHT CARBON IS THE 4TH MOST ABUNDANT ELEMENT IN THE UNIVERSE. CARBON BECAUSE IT HAS 4 VALENCE ELECTRONS IT CAN BOND WITH LOTS OF DIFFERENT ATOMS. BECAUSE OF ITS BONDING CAPABILITIES IT CAN FORM LONG CHAINS OR RINGs THE STRUCTURE OF AN ORGANIC MOLECULE DETERMINES IT’S FUNCTION ( WHAT IT DOES) Diamonds are made up of an allotrope of carbon What is it? Characteristics of living things 1. Made of complex chemical substances - nucleic acids (DNA, RNA) - pass on genetic information 2. Made of cells ( basic unit of living things) - single celled - ex. bacteria - multi cellular - ex. Humans 3. Require water & a source of energy usually in the form of ATP - for metabolism 4. Reproduce and grow - asexual - sexual - get larger , more complex More characteristics of living things 5. Have limited life spans - dead organisms become food for other organisms 6. Respond to their surroundings - homeostasis – maintaining a balanced internal state. ie. Body temperature ( humans) 7. Move - to find food - escape from danger 8. Able to repair injuries to themselves - if not too severe - ex. Salamander ( grow new arms or legs) 9. Can adapt to changes in their environments Characteristics of non-living things Not made of cells Do not obtain & use energy Do not grow & develop Do not reproduce Do not respond to their environment Do not adapt to their environment What do living & nonliving things have in common with each other? THEY ARE BOTH MADE UP OF- COMPOUNDS WHICH ARE THEMSELVES MADE UP OF ELEMENTS JOINED BY ENERGY RICH BONDS. LIKE THOSE IN ATP BONDS???????? OKAY WHAT DOES ENERGY HAVE TO DO WITH IT? endergonic reactions- bonds are formed and energy absorbed. exergonic reactions – bonds are broken and energy is released.