* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download interaction of tcr with mhc and antigen
Immune system wikipedia , lookup
DNA vaccination wikipedia , lookup
Human leukocyte antigen wikipedia , lookup
Innate immune system wikipedia , lookup
Duffy antigen system wikipedia , lookup
Cancer immunotherapy wikipedia , lookup
Monoclonal antibody wikipedia , lookup
Adaptive immune system wikipedia , lookup
Immunosuppressive drug wikipedia , lookup
Adoptive cell transfer wikipedia , lookup
Molecular mimicry wikipedia , lookup
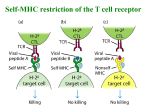
NATIONAL CHENG KUNG UNIVERSITY MEDICAL COLLEGE DEPARTMENT OF MEDICAL TECHNOLOGY Friday, September 21, 2007, 8:10-10:00 a.m. ANTIBODIES T CELL RECEPTORS AND MHC MOLECULES Yee-Shin Lin, Ph.D. A. BACKGROUND Two distinct types of molecule are involved in the recognition of foreign antigen which leads to the initiation of adaptive immune response -- the immunoglobulins (Ig) and the T-cell antigen receptors (TCR). Characteristic features of Ig and TCR are: (1) diversity and heterogeneity, (2) gene rearrangements. The Ig, or antibodies, are produced by B lymphocytes and are found associated with the globulin fraction of serum. Some are carried on cell surfaces where they act as receptors. They are glycoproteins. Each Ig molecule is bifunctional: one region (Fab, variable region) binds to antigen while a different region (Fc region) mediates binding of Ig to host tissues including various cells of the immune system, some phagocytic cells, and C1q. Serum Ig levels are an important indication to the physician in assessing the state of health of the patient. Antibodies can be administered to a patient to help combat disease. TCR are expressed on the membrane surface of T lymphocytes, there is no production of soluble molecules, analogous to the circulating Ab. This lecture will also discuss the means that B cells and T cells are able to generate a great diversity of antigen specific receptors (i.e., surface Ig and TCR) within each individual. The immune system has a specific cell for each the millions of possible epitopes. In fact, B and T cells bear specific cell surface receptors prior to antigen exposure. All antigen specificities are determined during embryonic and neonatal life before the immune system is exposed to Ag. Antibody and TCR: Features in common: 1. Both have variable (V) and constant (C) domains. 2. Both have the processes of gene recombination. Differences: 1. Antibody recognizes antigens in solution or on cell surfaces in their native conformation, while TCR sees antigen in association with MHC molecules on cell surfaces. 2. Antibody may be produced in two forms, either as the B cell antigen receptor or as secreted antibody, whereas the TCR is an integral membrane protein. Reading: Male, Brostoff, Roth & Roitt [chapters 3 and 5] B. LECTURE SUMMARY ANTIBODIES Immunoglobulins (Ig) are a family of serum proteins with antibody activity. Although displaying heterogeneous specificity, they have a number of common physical properties: 1. Globular proteins classified according to electrophoretic mobility in gel. 2. Basic monomeric molecular structure is the same for all Ig. Each Y-shaped Ig monomer consists of 2 identical heavy and 2 identical light chains linked by disulfide bonds. [Fig. 3.1, 3.4] 3. There are only 2 light (L) chain classes in man called kappa or lambda. Both types associate with all heavy chain classes. However, a single B cell expresses only one type of light chain, i.e. a B cell expresses either 2 kappa or 2 lambda chains, but never both. molecular weight: approximately 25,000 daltons molecule contains two domains, each containing an intrachain disulfide bond. Constant domain at the COOH terminus, amino acid sequence is the same for all light chains of a given type (i.e. either kappa or lambda). Variable domain at the NH2 terminus, amino acid sequence differs from one B cell to the next. 4. There are 5 major heavy chain classes in man, these, in turn, determine the isotype or class of Ig molecule: IgG, IgM, IgA, IgD, IgE. Each heavy chain contains one variable and 3 or 4 constant region domains. Similar to light chains, each domain contains a single intrachain disulfide bond. Ig subclasses are determined by slight constant region amino acid sequence differences. Subclass differences within an Ig class are less than those between the different classes. [Fig. 3.8] 5. Variable regions. Ab class or isotype is determined by CH1-4 and light chain type is determined by CL. The variable regions of both heavy and light chains (VH and VL) determine the Ag-binding specificity. 6. Disulfide bonds. The heavy and light chains are held together by disulfide bonds. Disulfide bonds join the heavy chains at or near a flexible, proline-rich portion of the heavy chain known as the hinge region. The hinge region confers flexibility on the Ig molecule. 7. Particular forms of certain classes of Ig have polymeric structures that are multiples of the basic 2H2L structure. In addition, an accessory polypeptide J is associated with IgM and IgA. IgA also contains a secretory component (SC). Concept: During its lifespan, a B cell (and its progeny) synthesizes a single variable region that binds only a particular epitope. However, the B cell can switch the isotype expressed. The B cell has the capacity to switch heavy chain constant region genes, but not variable region genes. 8. Antigen binding site resides in the Fab portion of the Ig molecule. As you must have guessed, the variable portion of the both H and L chains confers antigen-binding specificity to the Ig molecule. In fact when a number of variable region amino acid sequences were compared, three segments of exceptional variability were discovered and are termed hypervariable regions. The interaction of variable regions of both the H and L variable regions determine the antigen-binding site. These polypeptide chains are folded into a pocket-like binding site. Note that each Ig molecule has 2 identical Ag-binding sites and an intact Ig molecule is divalent. 9. Ig Fc portion is determined by the CH regions of heavy chain. This portion of the molecule is important in many effector mechanisms. You will recall that both macrophages and B cells bear FcR, and that these are thought to be important in concentrating and removal of Ag-Ab complexes from the circulation. Ag binding human IgM, IgG1 and IgG3 activates the complement system (classical pathway) and plays an important role in inflammatory reactions [see chapter 4]. In man, only IgG (all subclasses) readily crosses the placenta. Thus, the mother is able to confer passive immunity to her fetus. 5 different Ig classes (or isotypes) in humans and mice: IgG -- Subclasses: IgG1, IgG2, IgG3, IgG4 (human) IgG1, IgG2a, IgG2b, IgG3 (mouse) They are monomeric (2H, 2L) molecules. IgM -- the heaviest isotype with an m.wt. approaching 1,000,000 daltons. Found as either a monomeric surface-bound (2H, 2L) or as a secreted pentamer of five monomers (each containing 2H, 2L) for a total of 10H and 10L, linked by disulfide bonds. IgA -- usually associated with mucous membranes and their fluids. Found in monomeric (2H, 2L) and dimeric (joined monomers) forms. Monomeric form is found in serum. Dimeric form, together with secretory component (synthesized by epithelial cells not by plasma cells) is found in fluids of mucous tissues. The secretory component is thought to facilitate sIgA into mucous secretions as well as to protect sIgA from proteolytic attack [Fig. 3.11]. IgD -- very low serum concentration, monomeric structure (2H, 2L) found almost exclusively on B cell surface, function unknown although it may play a role in antigen-triggered lymphocyte differentiation. IgE -- relatively low serum concentration. Atopic individuals (people susceptible to allergic reactions) often show greater concentrations of IgE. Crosslinking of IgE on mast cell surface by Ag results in degranulation of cells with histamine release. The structure, serum concentration, and function of each Ig class are summarized in next page. [see also Fig. 3.3] INTERACTION OF ANTIBODIES WITH ANTIGENS Interaction of Ab with Ag involves the formation of multiple non-covalent biochemical interactions [p. 68]. This can be represented by the simple formula: Ab + Ag -------------> AbAg <----------- [Fig. 3.13] Ag-Ab interactions are reversible. The strength of interaction (i.e. how far to the right it is driven) is termed affinity. And an affinity constant is derived: [AbAg] K = --------------[Ab] [Ag] [Fig. 3.13] Ig show a wide range of affinity (10-4 to 10-14) with most antibodies falling in the 10-5 to 10-7 range. Affinity: monovalent binding between Ab and Ag Avidity: multivalent binding between Ab and Ag [Fig. 3.14] Antibody specificity: specific reaction, cross-reaction, no reaction [Fig. 3.15] configurational specificity [Fig. 3.17] BIOLOGICAL PROPERTIES OF IMMUNOGLOBULINS [Fig. 3.2, 3.18, 3.19] ANTIBODY RECEPTORS 1. Receptors for IgG: FcRI (CD64), FcRII (CD32), FcRIII (CD16) [Fig. 3.20] 2. Receptors for IgE: FcRI, FcRII (CD23) [Fig. 3.22] IMMUNOGLOBULIN VARIABILITY: [Fig. 3.24] 1. Isotypic variation: refers to the different heavy and light chain classes and subclasses. Variants are present in all members of a species (in constant region). 2. Allotypic variation: refers to the different alleles at a given locus. Variants are present within a species due to genetic differences (mostly in constant region). Idiotypic variation: usually specific for the individual antibody clone (in variable region). In particular relates to the hypervariable regions (complementarity-determining regions, CDRs) [Fig. 3.6]. Isotype Structure mw IgG monomer 146,000 to 170,000 IgM monomer IgA % of total Ig Serum halflife Serum conc. mg/ml 70-75 21d 13-14 most abundant; major Ab of 2nd immune response; 1 & 3 > 2 fix C (4 does not); cross placenta; bind to FcR on phagocytes (opsonization) — — surface form Biological properties pentamer (+J) 970,000 10 10 d 1.5 major Ab in primary immune response; fix C; bind to FcR on phagocytes (opsonization) monomer 160,000 15-20 6d 3.5 serum form dimer (+J+SC) 385,000 — 0.05 major Ig in external secretions to protect mucous surface against infectious agents at their “ports of entry”; aid in protection of infants against infection IgD monomer 184,000 <1 3d 0.03 most prevalent on B cell surface as an Ag-specific triggering receptor IgE monomer 188,000 trace 2d 0.00005 bind to FcR on mast cells and trigger their degranulation resulting in an immediate hypersensitivity response IMMUNOGLOBULIN GENE RECOMBINATION There are 3 unlinked families of genes for Ig on 3 different chromosomes: those for lambda L chains, kappa L chains, and the H chain family (includes all heavy chains). In each family, variable and constant regions for the particular chains are linked on the same chromosome. 1. Heavy chain gene recombination (VDJ) [Fig. 3.25] 2. Light chain gene recombination (VJ) [Fig. 3.26, 3.27] At the DNA level, variable segment is encoded as a series of genes divided into three groups: variable or VH genes, diversity or D genes, and joining or JH genes. There are a number of genes in each group. During the embryonic development of a particular B cell lineage (clonal), but prior to contact with antigen, this segment of chromosome undergoes a rearrangement whereby one VH gene, one D gene and one JH gene are united by clipping out and discarding the DNA between them. This VH-D-JH unit now encodes the variable portion of the Ig heavy chain. Because the DNA encoding the other VH, D, and JH genes was clipped out and discarded, this cell and its descendants are limited to producing only a single type of variable domain for its heavy chains. 3. Mechanisms for the generation of Ig diversity: [Fig. 3.28-3.33] a. multiple germ line V genes: there are multiple V genes for each of the 3 families -- lambda chain, kappa chain, and heavy chain. b. VJ and VDJ recombinations [Fig. 3.28, 3.29] Lambda and kappa light chains: each of the multiple V genes can combine with one of several J genes, and additional diversity is generated. Heavy chain: operates similarly as light chains, except that there is another set of genes (D genes) which together with V and J genes recombine to encode the V region of the heavy chains. The potential diversity thus is multiplied by yet another factor. c. N-nucleotide addition d. gene conversion e. recombinational inaccuracies (variable recombination) f. somatic point mutation g. assorted heavy and light chains Heavy chain switching: involves sequential deletion of particular CH genes allowing same VH-D-JH genes to be expressed with different CH genes. [see chapter 8] T CELL RECEPTORS AND MHC MOLECULES T CELL RECEPTORS Two main groups of T cell receptors (TCR) are expressed on separate lineages of cells and show quite distinct anatomical locations. The TCR is present on about 95% of peripheral T cells and on the majority of TCR-expressing thymocytes. The T cells are abundant in various epithelia. The TCR is a heterodimer with a molecular weight of 90,000. Each chain has a distinct constant (C) region and a distinct variable (V) region. Each of these regions is characterized by an intrachain disulfide bridge and some sequence homology with the Ig domains. These two chains are linked by a single interchain disulfide bridge. The CD3 complex is closely associated with the TCR and mediates signal transduction when T cells are activated by antigen binding to the TCR [Fig. 5.1, 5.3]. T-CELL RECEPTOR GENES There are four sets of genes (, , ) which generate the antigen/MHC binding portion of the T cell receptor (TCR). The general arrangement of TCR genes is remarkably similar to that of Ig heavy chains. Diversification of the TCR genes occurs by recombination of VJ or VDJ joining. [Fig. 5.4] MAJOR HISTOCOMPATIBILITY COMPLEX (MHC) MOLECULES Concept: Gene products of the MHC play a fundamental role in immune response. Genetic map of MHC: [Fig. 5.5] Mouse H-2 complex Human HLA complex Structure and biochemistry of class I and class II molecules: [Fig. 5.9] Glycoproteins on cell surface Two polypeptides (Class I: 45 kDa heavy chain and 12 kDa 2-microglobulin; Class II: 30-34 kDa chain and 26-29 kDa chain) Class I: H-2K, H-2D, H-2L (mouse), HLA-A, HLA-B, HLA-C (human) Class II: H-2I (A, E) (mouse), HLA-D (DP, DQ, DR) (human) Cellular distribution of MHC antigens: Class I antigens are expressed on all nucleated cells. Class II antigens are characteristic of the cells in the immune system. MHC polymorphism: A hallmark of the MHC is the extreme degree of polymorphism encoded within it. Polymorphism refers to the large number of variants seen in different individuals at the same gene locus and hence in the proteins which that locus encodes. Each common variant of a polymorphic gene present in the population is called an allele. For polymorphic genes, an individual can have the same allele at a genetic locus on both chromosomes of the pair or two different alleles, one on each chromosome. [p. 120] MHC molecules are co-dominantly expressed. This means that in one individual, all of the principle gene loci are expressed from both the maternal and paternal chromosomes. [p.121] INTERACTIONS OF MHC MOLECULES WITH ANTIGENIC PEPTIDES The peptide association with MHC class I and class II molecules are shown in Fig. 5.12, 5.13. The MHC class I and class II molecules present peptides derived from endogenous and exogenous antigens, respectively. [see chapter 7] INTERACTION OF TCR WITH MHC AND ANTIGEN T cells do not see free antigen. They recognize it on the surface of other cells (antigen-presenting cells, APCs). Antigens are internalized by APCs and are degraded by proteolytic enzymes in the phagolysosomes (antigen processing). The partially degraded material is re-expressed at the cell surface in association with MHC molecules (antigen presentation) [see chapter 7]. T cell antigen recognition involves a 3-way interaction involving the TCR, MHC molecule and processed antigen [Fig. 5.15]. In general, CD4+ cells recognize antigen plus class II MHC molecules while CD8+ cells recognize antigen plus class I MHC molecules. [Fig. 5.20] ANTIGEN PRESENTATION BY CD1 CD1 is MHC class I-like molecule which presents lipid antigens. The CD1 genes are located outside the MHC and are not polymorphic. CD1 was originally identified on cortical thymocytes as a T-cell differentiation marker, and was subsequently found on B cells and dendritic cells for antigen presentation. C. ImmunObjectives -- a self assessment 1. Define: H and L chains kappa and lambda chains hinge region V and C region Fab and Fc fragments Ig and Ig, Ig Isotypic/allotypic/idiotypic variations affinity/avidity cross-reactivity TCR and CD3 H-2/HLA genes and proteins CD1 MHC polymorphism MHC co-dominant 2. Name the 5 classes of antibody -- approx. molecular weight of each, serum concentration, major physiological roles. 3. Diagram the structure of typical Ig molecules of each class. Label heavy and light chains, Fc, Fab, J chains, antigen binding sites, disulfide bonds, and secretory component. 4. How is it possible to generate so many variable domains of the chains which make up Ig? How the different genes involved are brought together? 5. Describe the common and distinct characteristics of Ig and TCR. 6. How is it possible to generate so many variable domains of the chains which make up TCR? How the different genes involved are brought together? 7. Describe interactions of TCR with MHC and antigen. What molecules do T cells see on antigen-presenting cells?