Catabolic pathways
... broken down into their component building blocks. For example, proteins are degraded to amino acids, polysaccharides to monosaccharides, and fats (triacyl glycerols) to free fatty acids and glycerol. ...
... broken down into their component building blocks. For example, proteins are degraded to amino acids, polysaccharides to monosaccharides, and fats (triacyl glycerols) to free fatty acids and glycerol. ...
Carbon Based Compounds
... Organic macromolecules: An organic compound is any member of a large class of gaseous, liquid, or solid chemical compounds whose molecules contain carbon. ...
... Organic macromolecules: An organic compound is any member of a large class of gaseous, liquid, or solid chemical compounds whose molecules contain carbon. ...
The Necessities of Life
... description of the expected workload by explaining whether the building block will have to work constantly or sporadically. Indicate whether the ...
... description of the expected workload by explaining whether the building block will have to work constantly or sporadically. Indicate whether the ...
Biosynthesis of Macromolecules
... • Build small molecules into larger molecules or cell structures (ex. Glucose, amino acids, fatty acids=>lipid, PG) • Diversity of metabolic systems (chemoheterotrophs, etc.) ...
... • Build small molecules into larger molecules or cell structures (ex. Glucose, amino acids, fatty acids=>lipid, PG) • Diversity of metabolic systems (chemoheterotrophs, etc.) ...
Metabolism08
... breaks triglycerides into glycerol and fatty acids Most of the energy is stored in the fatty acids (glycerol can be converted to glucose or pyruvate) Fatty acids usually produce substantially more ATP than glucose (16 carbon fatty acid = 129 ATP) ...
... breaks triglycerides into glycerol and fatty acids Most of the energy is stored in the fatty acids (glycerol can be converted to glucose or pyruvate) Fatty acids usually produce substantially more ATP than glucose (16 carbon fatty acid = 129 ATP) ...
Foundations in Microbiology
... resembles normal substrate competes with substrate for active site 2. Feedback inhibition – concentration of product at the end of a pathway blocks the action of a key enzyme 3. Feedback repression – inhibits at the genetic level by controlling synthesis of key enzymes 4. Enzyme induction – enzymes ...
... resembles normal substrate competes with substrate for active site 2. Feedback inhibition – concentration of product at the end of a pathway blocks the action of a key enzyme 3. Feedback repression – inhibits at the genetic level by controlling synthesis of key enzymes 4. Enzyme induction – enzymes ...
Ch. 2 Fill-In Study Guide
... Thee examples would be (fruit, potato, pasta, honey, juice, fiber) 5. Simple carbohydrates manufactured by plants are (sugars, starches) which give quick energy. Thee examples would be (fruit, potato, pasta, honey, juice, fiber) 6. Cells use which 5 choices for energy and energy storage: (fats, suga ...
... Thee examples would be (fruit, potato, pasta, honey, juice, fiber) 5. Simple carbohydrates manufactured by plants are (sugars, starches) which give quick energy. Thee examples would be (fruit, potato, pasta, honey, juice, fiber) 6. Cells use which 5 choices for energy and energy storage: (fats, suga ...
Metabolism - California Science Teacher
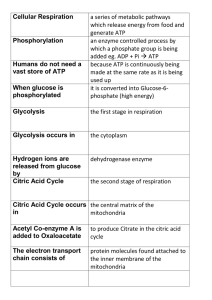
... During respiration most energy flows in a sequence: glucose, NADH, electron transport chain, proton, motive force, ATP. ...
... During respiration most energy flows in a sequence: glucose, NADH, electron transport chain, proton, motive force, ATP. ...
Introduction to metabolism. Specific and general pathways of
... are oxidized to common metabolite (acetyl CoA) Stage III. Acetyl CoA is oxidized in citric acid cycle to CO2 and water. As result reduced cofactor, NADH2 and FADH2, are formed which give up their electrons. Electrons are transported via the tissue respiration chain and released energy is coupled dir ...
... are oxidized to common metabolite (acetyl CoA) Stage III. Acetyl CoA is oxidized in citric acid cycle to CO2 and water. As result reduced cofactor, NADH2 and FADH2, are formed which give up their electrons. Electrons are transported via the tissue respiration chain and released energy is coupled dir ...
File
... because ATP is continuously being made at the same rate as it is being used up it is converted into Glucose-6phosphate (high energy) ...
... because ATP is continuously being made at the same rate as it is being used up it is converted into Glucose-6phosphate (high energy) ...
Control of intra-cellular (enzyme regulator)
... In eukaryotes, anabolic and catabolic pathways that interconvert common products may take place in specific sub cellular compartments. ...
... In eukaryotes, anabolic and catabolic pathways that interconvert common products may take place in specific sub cellular compartments. ...
Fermentation and Biosynthetic Pathways File
... intermediates produced during glycolysis, Krebs cycle and from lipids or amino acids. Bacteria may assemble it into the more complex polysaccharides. Biosynthesis of Lipids Lipids vary in chemical composition, cells synthesize fats by joining glycerol and fatty acids. The glycerol portion of the fat ...
... intermediates produced during glycolysis, Krebs cycle and from lipids or amino acids. Bacteria may assemble it into the more complex polysaccharides. Biosynthesis of Lipids Lipids vary in chemical composition, cells synthesize fats by joining glycerol and fatty acids. The glycerol portion of the fat ...
Cell Metabolism Review
... During catabolism (breakdown rxns of metabolism), energy is released in the form of ATP. Often energy is also released in the form of electrons (i.e. the organic molecule being broken down is oxidized). These electrons are carried by electron carriers such as NADH and FADH2 (Note: when not carrying ...
... During catabolism (breakdown rxns of metabolism), energy is released in the form of ATP. Often energy is also released in the form of electrons (i.e. the organic molecule being broken down is oxidized). These electrons are carried by electron carriers such as NADH and FADH2 (Note: when not carrying ...
Organic Compounds - West Branch Schools
... Nucleotides made of 3 parts: 1. Nitrogenous bases (A, G, C, T) ...
... Nucleotides made of 3 parts: 1. Nitrogenous bases (A, G, C, T) ...
Metabolism Metabolism refers to all the chemical reactions within an
... The citric acid cycle (Krebs cycle, tricarboxylic acid cycle) The citric acid cycle is a series of reactions in mitochondria that oxidize acetyl residues (as acetyl-CoA) and reduce coenzymes that upon reoxidation are linked to the formation of ATP. The citric acid cycle is the final common pathway f ...
... The citric acid cycle (Krebs cycle, tricarboxylic acid cycle) The citric acid cycle is a series of reactions in mitochondria that oxidize acetyl residues (as acetyl-CoA) and reduce coenzymes that upon reoxidation are linked to the formation of ATP. The citric acid cycle is the final common pathway f ...
BIO 6.3 Carbon - Steinbach Science
... (e.g., starch, glycogen, cellulose) Lipids are organic compounds that have a large portion (much greater than 2 to 1) or C—H bonds and less oxygen than carbohydrates (e.g., beef fat has the formula ...
... (e.g., starch, glycogen, cellulose) Lipids are organic compounds that have a large portion (much greater than 2 to 1) or C—H bonds and less oxygen than carbohydrates (e.g., beef fat has the formula ...
Recitation 3 - Department of Chemistry ::: CALTECH
... are bounced off and things that are bounced off are these high energy carriers. High energy carriers bounce down a staircase A series of oxidation reactions then ensue with the result that two carbons are expelled as CO2, (G/A)TP is produced and the electron carriers (NAD+ and FAD+) are reduced. ...
... are bounced off and things that are bounced off are these high energy carriers. High energy carriers bounce down a staircase A series of oxidation reactions then ensue with the result that two carbons are expelled as CO2, (G/A)TP is produced and the electron carriers (NAD+ and FAD+) are reduced. ...
Metabolism

Metabolism (from Greek: μεταβολή metabolē, ""change"") is the set of life-sustaining chemical transformations within the cells of living organisms. These enzyme-catalyzed reactions allow organisms to grow and reproduce, maintain their structures, and respond to their environments. The word metabolism can also refer to all chemical reactions that occur in living organisms, including digestion and the transport of substances into and between different cells, in which case the set of reactions within the cells is called intermediary metabolism or intermediate metabolism.Metabolism is usually divided into two categories: catabolism, the breaking down of organic matter by way of cellular respiration, and anabolism, the building up of components of cells such as proteins and nucleic acids. Usually, breaking down releases energy and building up consumes energy.The chemical reactions of metabolism are organized into metabolic pathways, in which one chemical is transformed through a series of steps into another chemical, by a sequence of enzymes. Enzymes are crucial to metabolism because they allow organisms to drive desirable reactions that require energy that will not occur by themselves, by coupling them to spontaneous reactions that release energy. Enzymes act as catalysts that allow the reactions to proceed more rapidly. Enzymes also allow the regulation of metabolic pathways in response to changes in the cell's environment or to signals from other cells.The metabolic system of a particular organism determines which substances it will find nutritious and which poisonous. For example, some prokaryotes use hydrogen sulfide as a nutrient, yet this gas is poisonous to animals. The speed of metabolism, the metabolic rate, influences how much food an organism will require, and also affects how it is able to obtain that food.A striking feature of metabolism is the similarity of the basic metabolic pathways and components between even vastly different species. For example, the set of carboxylic acids that are best known as the intermediates in the citric acid cycle are present in all known organisms, being found in species as diverse as the unicellular bacterium Escherichia coli and huge multicellular organisms like elephants. These striking similarities in metabolic pathways are likely due to their early appearance in evolutionary history, and their retention because of their efficacy.