TERM 2 Unit 3 YR 9 SCI It is elementary
... understandings of atomic structure. Students model an atom according to currently accepted understandings. They will identify patterns in atomic structure that allow prediction of the products of chemical reactions and are reflected by the periodic table. They recognise that new substances are forme ...
... understandings of atomic structure. Students model an atom according to currently accepted understandings. They will identify patterns in atomic structure that allow prediction of the products of chemical reactions and are reflected by the periodic table. They recognise that new substances are forme ...
Answer Key - La Quinta High School
... dilute, the temperature may change only by a fraction of a degree and may not be noticed. 2. A chemical equation indicates the substances necessary for a chemical reaction to take place, as well as what is produced by that chemical reaction. In addition, if a chemical equation has been balanced, the ...
... dilute, the temperature may change only by a fraction of a degree and may not be noticed. 2. A chemical equation indicates the substances necessary for a chemical reaction to take place, as well as what is produced by that chemical reaction. In addition, if a chemical equation has been balanced, the ...
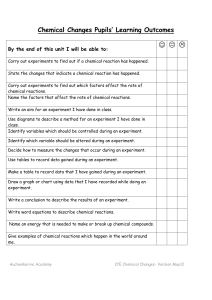
Chemical Reactions Unit Pupils` Learning Outcomes
... Chemical Changes Pupils’ Learning Outcomes ...
... Chemical Changes Pupils’ Learning Outcomes ...
Unit 2: Chemical Reactions
... • A chemical formula is an abbreviation for a chemical compound using chemical symbols and numbers. • The subscript number tells how many atoms of the element are present in the compound • Example: CO2 = Carbon Dioxide – Di = 2 – 1 Carbon atom and 2 oxygen atoms ...
... • A chemical formula is an abbreviation for a chemical compound using chemical symbols and numbers. • The subscript number tells how many atoms of the element are present in the compound • Example: CO2 = Carbon Dioxide – Di = 2 – 1 Carbon atom and 2 oxygen atoms ...
Chemical Reaction Basics
... A ____________ ____________ is a process by which one or more substances are changed into one or more ____________ substances. Chemical reactions are represented by some type of ____________. The general form is as follows: ...
... A ____________ ____________ is a process by which one or more substances are changed into one or more ____________ substances. Chemical reactions are represented by some type of ____________. The general form is as follows: ...
Physical and Chemical Prop/changes
... Every substance has characteristic properties that are used to distinguish it from others. ...
... Every substance has characteristic properties that are used to distinguish it from others. ...
Unit 5 Chemical Properties and Changes Video Notes A ______ is a
... ________________________ A change that alters the identity of a substance resulting in a new substance or substances with different properties ________________________ Those characteristics that can be observed when a chemical reaction changes the identity of the substance, such as potential to rus ...
... ________________________ A change that alters the identity of a substance resulting in a new substance or substances with different properties ________________________ Those characteristics that can be observed when a chemical reaction changes the identity of the substance, such as potential to rus ...
Deconstructed HS-PS1-2
... trends in the periodic table, and knowledge of the patterns of chemical properties.[Clarification Statement: Examples of chemical reactions could include the reaction of sodium and chlorine, of carbon and oxygen, or of carbon and hydrogen.] [Assessment Boundary: Assessment is limited to chemical rea ...
... trends in the periodic table, and knowledge of the patterns of chemical properties.[Clarification Statement: Examples of chemical reactions could include the reaction of sodium and chlorine, of carbon and oxygen, or of carbon and hydrogen.] [Assessment Boundary: Assessment is limited to chemical rea ...
CHM 2045C - State College of Florida
... CHM 1025C with a grade of “C” or better or one year of high school college preparatory honors or AP chemistry within last three years. This course meets Area V for the A.A./A.S. general education requirements. A rigorous study of chemistry principles for students who have already studied basic conce ...
... CHM 1025C with a grade of “C” or better or one year of high school college preparatory honors or AP chemistry within last three years. This course meets Area V for the A.A./A.S. general education requirements. A rigorous study of chemistry principles for students who have already studied basic conce ...
Chapter 2 SWBATS Content Standards Cell Biology 1. The
... What are the two main types of chemical bonds? Give an example of each. Why are water molecules polar? What are acidic solutions? What are basic solutions? Give an example of each. What are the functions of each group of organic compounds? What happens to chemical bonds during chemical reactions? Ho ...
... What are the two main types of chemical bonds? Give an example of each. Why are water molecules polar? What are acidic solutions? What are basic solutions? Give an example of each. What are the functions of each group of organic compounds? What happens to chemical bonds during chemical reactions? Ho ...
CHEMICAL INVENTORY - Web based software (chemoventory v3.0)
... SOFTWARE – Chemoventory 3.0 Chemoventory is a web ...
... SOFTWARE – Chemoventory 3.0 Chemoventory is a web ...
Chemical weapon

A chemical weapon (CW) is a munition that uses chemicals formulated to inflict death or harm on human beings. The Organisation for the Prohibition of Chemical Weapons (OPCW) states: The term chemical weapon may also be applied to any toxic chemical or its precursor that can cause death, injury, temporary incapacitation or sensory irritation through its chemical action. Munitions or other delivery devices designed to deliver chemical weapons, whether filled or unfilled, are also considered weapons themselves.They are classified as weapons of mass destruction (WMDs), though they are distinct from nuclear weapons, biological weapons (diseases), and radiological weapons (which use radioactive decay of elements). All may be used in warfare known by the military acronym NBC, for nuclear, biological, and chemical warfare. Weapons of mass destruction are distinct from conventional weapons, which are primarily effective due to their explosive, kinetic, or incendiary potential. Chemical weapons can be widely dispersed in gas, liquid and solid forms, and may easily afflict others than the intended targets. Nerve gas, tear gas and pepper spray are three modern examples.Lethal, unitary, chemical agents and munitions are extremely volatile and they constitute a class of hazardous chemical weapons that are now being stockpiled by many nations. (Unitary agents are effective on their own and require no mixing with other agents.) The most dangerous of these are nerve agents GA, GB, GD, and VX, and vesicant (blister) agents which are formulations of sulfur mustard such as H, HT, and HD. All are liquids at normal room temperature, but become gaseous when released. Widely used during the First World War, the effects of so-called mustard gas, phosgene gas and others caused lung searing, blindness, death and maiming.Pepper spray is of common use today. It is potentially lethal. There are no recent records of pepper spray being used in war, despite the fact that it inflicts fewer injuries and side-effects compared with impact and explosive weapons.Under the Chemical Weapons Convention (1993), there is a legally binding, world-wide ban on the production, stockpiling, and use of chemical weapons and their precursors. Notwithstanding, large stockpiles thereof continue to exist, usually justified as only a precaution against putative use by an aggressor.