SCH3U Course Review
... increase with increasing atomic radii decrease with increasing nuclear charge decrease across a period from left to right increase across a period from left to right increase as you go down a family ...
... increase with increasing atomic radii decrease with increasing nuclear charge decrease across a period from left to right increase across a period from left to right increase as you go down a family ...
chemical*equations
... “Success'is'not',inal,'failure' is'not'fatal:'it'is'the'courage' to'continue'that'counts.” ''7Winston'Churchill ...
... “Success'is'not',inal,'failure' is'not'fatal:'it'is'the'courage' to'continue'that'counts.” ''7Winston'Churchill ...
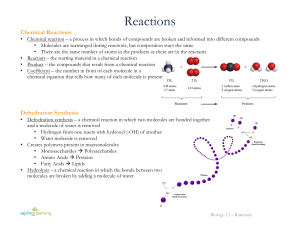
Reactions
... • Dehydration synthesis – a chemical reaction in which two molecules are bonded together and a molecule of water is removed • Hydrogen from one reacts with hydroxyl (-OH) of another • Water molecule is removed • Creates polymers present in macromolecules • Monosaccharides à Polysaccharides • Amino ...
... • Dehydration synthesis – a chemical reaction in which two molecules are bonded together and a molecule of water is removed • Hydrogen from one reacts with hydroxyl (-OH) of another • Water molecule is removed • Creates polymers present in macromolecules • Monosaccharides à Polysaccharides • Amino ...
Nothing Lost, Nothing Gained
... the ashes from the burned paper. Let's add up the thing we wrote above . . . H and H are H2. Now just add an O and we will get H2O or . . . water! See? That was not too hard. ...
... the ashes from the burned paper. Let's add up the thing we wrote above . . . H and H are H2. Now just add an O and we will get H2O or . . . water! See? That was not too hard. ...
Begin Chemical Equations Practice
... How many? • How many of each atom are shown by 4Al2(SO4)3 • How many of each atom are shown by 3(NH4)3PO4 ...
... How many? • How many of each atom are shown by 4Al2(SO4)3 • How many of each atom are shown by 3(NH4)3PO4 ...
Classifying Chemical Reactions 9-3
... Can’t change the formula, because it describes what it is (carbon monoxide in this example) ...
... Can’t change the formula, because it describes what it is (carbon monoxide in this example) ...
CHEMICAL EQUATIONS NAME PERIOD_______ DATE________
... A ______________ is a well-defined example of a chemical change. A chemical ___________ can be used to show the changes that occur in a chemical reaction. In a chemical equation, the substances on the left side of the arrow are the starting substances. These substances are called ______________. The ...
... A ______________ is a well-defined example of a chemical change. A chemical ___________ can be used to show the changes that occur in a chemical reaction. In a chemical equation, the substances on the left side of the arrow are the starting substances. These substances are called ______________. The ...
Reading-Chem v Phys
... signify a physical change. Table salt (sodium chloride) is a white solid at room temperature. It can also exist in each of the three physical states. However, the temperatures at which the changes of state occur in sodium chloride are much higher than for the corresponding changes in water. Sodium c ...
... signify a physical change. Table salt (sodium chloride) is a white solid at room temperature. It can also exist in each of the three physical states. However, the temperatures at which the changes of state occur in sodium chloride are much higher than for the corresponding changes in water. Sodium c ...
Notes for Types of Reactions:
... NaHCO3(s) + CH3COOH(l) CO2(g) + NaC2H3O2(aq) + H2O(l) 3. Formation of a _________. Many reactions take place between substances that are _________ in liquids. If a ______ appears after two _________ are mixed, a reaction has likely occurred. precipitate = a _____ that is produced as a result of ...
... NaHCO3(s) + CH3COOH(l) CO2(g) + NaC2H3O2(aq) + H2O(l) 3. Formation of a _________. Many reactions take place between substances that are _________ in liquids. If a ______ appears after two _________ are mixed, a reaction has likely occurred. precipitate = a _____ that is produced as a result of ...
*6th Grade Science-Chapter 5 Study Guide Lesson 5.1: Observing
... *Chemical reactions involve changes in properties and changes in energy that you can often observe. Physical change-any change that alters the form or appearance of a substance but does not change it into another substance. Ex: bending, cutting, freezing, and boiling are examples of physical changes ...
... *Chemical reactions involve changes in properties and changes in energy that you can often observe. Physical change-any change that alters the form or appearance of a substance but does not change it into another substance. Ex: bending, cutting, freezing, and boiling are examples of physical changes ...
Chemical Building Blocks Chapter One
... Characteristic Property: a quality of a substance that never changes and can be used to identify the substances (pg. 15) Boiling Point: the temperature at which a substance changes from a liquid to a gas (pg. 16) Physical Change: a change in a substance that does not change its identity; for example ...
... Characteristic Property: a quality of a substance that never changes and can be used to identify the substances (pg. 15) Boiling Point: the temperature at which a substance changes from a liquid to a gas (pg. 16) Physical Change: a change in a substance that does not change its identity; for example ...
Chemical Reactions - Mr. Brown`s Science Town
... An Intro. To Chemical Reactions What is a chemical reaction? How do we represent chemical reactions? ...
... An Intro. To Chemical Reactions What is a chemical reaction? How do we represent chemical reactions? ...
Molecule: two or more atoms held together by
... Lipid: nonpolar molecule composed of carbon, hydrogen, and oxygen; includes fats and oils. Fatty acid: hydrocarbon chain often bonded to glycerol in a lipid Protein: polymer composed of amino acids linked by peptide bonds; folds into a particular structure depending on bonds between amino acids. Am ...
... Lipid: nonpolar molecule composed of carbon, hydrogen, and oxygen; includes fats and oils. Fatty acid: hydrocarbon chain often bonded to glycerol in a lipid Protein: polymer composed of amino acids linked by peptide bonds; folds into a particular structure depending on bonds between amino acids. Am ...
File - Flipped Out Science with Mrs. Thomas!
... Chemical formula - tells us the number of atoms of each element in a compound Chemical property – the ability of a substance to combine with or change into one or more other substances. Chemical symbol – a shorthand method of representing an element. Instead of writing out the name of an element, we ...
... Chemical formula - tells us the number of atoms of each element in a compound Chemical property – the ability of a substance to combine with or change into one or more other substances. Chemical symbol – a shorthand method of representing an element. Instead of writing out the name of an element, we ...
File - Flipped Out Science with Mrs. Thomas!
... Chemical formula - tells us the number of atoms of each element in a compound Chemical property – the ability of a substance to combine with or change into one or more other substances. Chemical symbol – a shorthand method of representing an element. Instead of writing out the name of an element, we ...
... Chemical formula - tells us the number of atoms of each element in a compound Chemical property – the ability of a substance to combine with or change into one or more other substances. Chemical symbol – a shorthand method of representing an element. Instead of writing out the name of an element, we ...
Topic2890 Thermodynamics and Kinetics A given system at
... water (l) , the solvent, together with n 0X and n 0Y moles of chemical substances X and Y respectively at time ‘t = 0’. The molalities of these solutes are m 0X (= n 0X / n 1 ⋅ M 1 = n 0X / w 1 ) and m 0Y (= n 0Y / n 1 ⋅ M 1 = n 0Y / w 1 ) respectively at time ‘t = 0’; the concentrations are c 0X (= ...
... water (l) , the solvent, together with n 0X and n 0Y moles of chemical substances X and Y respectively at time ‘t = 0’. The molalities of these solutes are m 0X (= n 0X / n 1 ⋅ M 1 = n 0X / w 1 ) and m 0Y (= n 0Y / n 1 ⋅ M 1 = n 0Y / w 1 ) respectively at time ‘t = 0’; the concentrations are c 0X (= ...
Atoms, Elements, Compounds File
... SOL 6.4 Atoms, Elements, compounds The student will investigate and understand that all matter is made up of atoms. Key concepts include ...
... SOL 6.4 Atoms, Elements, compounds The student will investigate and understand that all matter is made up of atoms. Key concepts include ...
Chemistry Review: Unit2 - Menno Simons Christian School
... difficult to reverse. 7) In the table below list whether there is a chemical or physical change and how you know. Observation Sugar dissolves in water. ...
... difficult to reverse. 7) In the table below list whether there is a chemical or physical change and how you know. Observation Sugar dissolves in water. ...
Introductory Chemistry Test Review
... and oxygen gas according to the following equation. Calculate how much oxygen in grams is produced when 35.0 grams of potassium chlorate decomposes. 2 KClO3(s) ...
... and oxygen gas according to the following equation. Calculate how much oxygen in grams is produced when 35.0 grams of potassium chlorate decomposes. 2 KClO3(s) ...
Yearly Plan for MYP 1 Science
... without them - recognizing common chemical reactions in our world - understanding what happens in a chemical change - noticing and identifying common chemicals we use in our everyday lives ...
... without them - recognizing common chemical reactions in our world - understanding what happens in a chemical change - noticing and identifying common chemicals we use in our everyday lives ...
Biochemistry Objectives Concepts of Matter and Energy (pp. 59
... Distinguish between organic and inorganic compounds. Differentiate clearly between a salt, an acid, and a base. List several salts (or their ions) vitally important to body functioning. Explain the importance of water to body homeostasis and provide several examples of the roles of water. Explain th ...
... Distinguish between organic and inorganic compounds. Differentiate clearly between a salt, an acid, and a base. List several salts (or their ions) vitally important to body functioning. Explain the importance of water to body homeostasis and provide several examples of the roles of water. Explain th ...
673 lab three
... A) DISCUSS CHEMICAL REACTIONS: start with a definition and apply the law of conservation of mass in a chemical reaction to the reaction in this lab. Discuss balanced reactions and give three example reactions and SHOW that they are balanced. Clearly indicate the role of COEFFICIENTS., B) DISCUSS CHE ...
... A) DISCUSS CHEMICAL REACTIONS: start with a definition and apply the law of conservation of mass in a chemical reaction to the reaction in this lab. Discuss balanced reactions and give three example reactions and SHOW that they are balanced. Clearly indicate the role of COEFFICIENTS., B) DISCUSS CHE ...
Chemical weapon

A chemical weapon (CW) is a munition that uses chemicals formulated to inflict death or harm on human beings. The Organisation for the Prohibition of Chemical Weapons (OPCW) states: The term chemical weapon may also be applied to any toxic chemical or its precursor that can cause death, injury, temporary incapacitation or sensory irritation through its chemical action. Munitions or other delivery devices designed to deliver chemical weapons, whether filled or unfilled, are also considered weapons themselves.They are classified as weapons of mass destruction (WMDs), though they are distinct from nuclear weapons, biological weapons (diseases), and radiological weapons (which use radioactive decay of elements). All may be used in warfare known by the military acronym NBC, for nuclear, biological, and chemical warfare. Weapons of mass destruction are distinct from conventional weapons, which are primarily effective due to their explosive, kinetic, or incendiary potential. Chemical weapons can be widely dispersed in gas, liquid and solid forms, and may easily afflict others than the intended targets. Nerve gas, tear gas and pepper spray are three modern examples.Lethal, unitary, chemical agents and munitions are extremely volatile and they constitute a class of hazardous chemical weapons that are now being stockpiled by many nations. (Unitary agents are effective on their own and require no mixing with other agents.) The most dangerous of these are nerve agents GA, GB, GD, and VX, and vesicant (blister) agents which are formulations of sulfur mustard such as H, HT, and HD. All are liquids at normal room temperature, but become gaseous when released. Widely used during the First World War, the effects of so-called mustard gas, phosgene gas and others caused lung searing, blindness, death and maiming.Pepper spray is of common use today. It is potentially lethal. There are no recent records of pepper spray being used in war, despite the fact that it inflicts fewer injuries and side-effects compared with impact and explosive weapons.Under the Chemical Weapons Convention (1993), there is a legally binding, world-wide ban on the production, stockpiling, and use of chemical weapons and their precursors. Notwithstanding, large stockpiles thereof continue to exist, usually justified as only a precaution against putative use by an aggressor.