Protein folding
... aggregation, and early degradation and thus to a loss of functional PAH proteins • BH4 – molecular chaperone – the restoration of enzyme function which might be transmitted by correction of protein misfolding. • About 75% of PAH mutations, characterized by high residual activity, have been found to ...
... aggregation, and early degradation and thus to a loss of functional PAH proteins • BH4 – molecular chaperone – the restoration of enzyme function which might be transmitted by correction of protein misfolding. • About 75% of PAH mutations, characterized by high residual activity, have been found to ...
Protein structure
... viewed via the Research Collaboratory for Structural Bioinformatics (www.rcsb.org) database. ...
... viewed via the Research Collaboratory for Structural Bioinformatics (www.rcsb.org) database. ...
Design of specific peptide Inhibitors of Phospholipase A2
... before cleavage. In order to regulate the production of proinflammatory compounds, a specific peptide inhibitor of PLA2, Leu-Ala-Ile-Tyr-Ser has been designed. Phospholipase A2 from Daboia russelli pulchella (DPLA2) and peptide Leu-Ala-Ile-Tyr-Ser (LAIYS) have been co-crystallized. Diffraction data ...
... before cleavage. In order to regulate the production of proinflammatory compounds, a specific peptide inhibitor of PLA2, Leu-Ala-Ile-Tyr-Ser has been designed. Phospholipase A2 from Daboia russelli pulchella (DPLA2) and peptide Leu-Ala-Ile-Tyr-Ser (LAIYS) have been co-crystallized. Diffraction data ...
Signal Transduction
... Once a cell picks up a hormonal or sensory signal, it must transmit this information from the surface to the interior parts of the cell— Example, to the nucleus. This occurs via signal transduction pathways that are: very specific, both in their activation and in their ...
... Once a cell picks up a hormonal or sensory signal, it must transmit this information from the surface to the interior parts of the cell— Example, to the nucleus. This occurs via signal transduction pathways that are: very specific, both in their activation and in their ...
Proteome analysis of Arabidopsis thaliana mitochondrial proteins
... During the last decade, Arabidopsis thaliana, a small flowering weed, has developed into a model system for plant development, physiology, and genetics. Scientists all over the world are using new tools to investigate the plant’s proteome to discover the processes common to all plants. A. thaliana i ...
... During the last decade, Arabidopsis thaliana, a small flowering weed, has developed into a model system for plant development, physiology, and genetics. Scientists all over the world are using new tools to investigate the plant’s proteome to discover the processes common to all plants. A. thaliana i ...
Tertiary Protein Structure Prediction with Profile Analysis: A Case Study
... Profilemake, part of the Genetics Computing Group suite of programs, was used to make profiles from the files of known helices and a structure-correlated scoring matrix. In addition, leave-one-out profiles were made by omitting one sequence from the set of sequences used to create the profiles. The ...
... Profilemake, part of the Genetics Computing Group suite of programs, was used to make profiles from the files of known helices and a structure-correlated scoring matrix. In addition, leave-one-out profiles were made by omitting one sequence from the set of sequences used to create the profiles. The ...
Poster
... Within every cell, there exists a system known as the ubiquitin-proteasome system (UPS) that eliminates damaged, misfolded or excess proteins. Unwanted proteins are tagged with ubiquitin, a small protein that identifies other proteins as being ready for degradation. The process of activating and tra ...
... Within every cell, there exists a system known as the ubiquitin-proteasome system (UPS) that eliminates damaged, misfolded or excess proteins. Unwanted proteins are tagged with ubiquitin, a small protein that identifies other proteins as being ready for degradation. The process of activating and tra ...
Acetylation of Ribosomal Proteins in Regenerating Rat Liver
... lysosomal enzyme. Alkaline phosphatase (Fernley & Walker, 1965), mainly a membrane enzyme localized in the brush border, was also measured, along with lactate dehydrogenase (Wacker & Dorfman, 1962), which has been followed by other authors for similar purposes. These enzyme activities were compared ...
... lysosomal enzyme. Alkaline phosphatase (Fernley & Walker, 1965), mainly a membrane enzyme localized in the brush border, was also measured, along with lactate dehydrogenase (Wacker & Dorfman, 1962), which has been followed by other authors for similar purposes. These enzyme activities were compared ...
Exposure to the metalloid arsenic is a daily occurrence as a result of
... on these results, we plan to test our hypothesis and accomplish the overall objective of this proposal by pursuing the following two specific aims: 1. Crystallization and structural studies of human Cdc25B and Cdc25C reductase. (a) Crystallization trials of full length Cdc25B and Cdc25C. Human Cdc25 ...
... on these results, we plan to test our hypothesis and accomplish the overall objective of this proposal by pursuing the following two specific aims: 1. Crystallization and structural studies of human Cdc25B and Cdc25C reductase. (a) Crystallization trials of full length Cdc25B and Cdc25C. Human Cdc25 ...
Translation - Protein Synthesis
... away from the ribosome, folding up into a protein as it goes. This continues for hundreds of amino acids until a stop codon is reached, when the ribosome falls apart, releasing the finished protein ...
... away from the ribosome, folding up into a protein as it goes. This continues for hundreds of amino acids until a stop codon is reached, when the ribosome falls apart, releasing the finished protein ...
Interactions, Tertiary Structures
... A. polypeptide chains held side by side by H bonds β-pleated sheet B. sequence of amino acids in a polypeptide chain primary C. corkscrew shape with H bonds between amino acids α-helix ...
... A. polypeptide chains held side by side by H bonds β-pleated sheet B. sequence of amino acids in a polypeptide chain primary C. corkscrew shape with H bonds between amino acids α-helix ...
Coevolution analysis of viral sequences: from HBV - LCQB
... protein and among proteins. Coevolving residues in a protein structure, possibly a complex, correspond to groups of residues whose mutations have arisen simultaneously during the evolution of different species, and this is due to several possible reasons involving the three-dimensional shape of th ...
... protein and among proteins. Coevolving residues in a protein structure, possibly a complex, correspond to groups of residues whose mutations have arisen simultaneously during the evolution of different species, and this is due to several possible reasons involving the three-dimensional shape of th ...
C.N.R. Short-term Mobility Program 2008 Gabriella Pocsfalvi
... in cell sorting and trafficking. Another putative non characterized protein in the same sample is SSO2749. This protein shows conserved domain of Linocin_M18 bacteriocin protein. Many Grampositive bacteria produce antimicrobial peptides, generally termed bacteriocins. These polypeptides usually has ...
... in cell sorting and trafficking. Another putative non characterized protein in the same sample is SSO2749. This protein shows conserved domain of Linocin_M18 bacteriocin protein. Many Grampositive bacteria produce antimicrobial peptides, generally termed bacteriocins. These polypeptides usually has ...
AP Biology Protein structure
... Ex. An enzyme must fit its substrate A hormone must fit its receptor ...
... Ex. An enzyme must fit its substrate A hormone must fit its receptor ...
Dreissena
... and protozoa from the water column which disrupts the food chain for native organisms and increases the clarity of the water, allowing photosynthesis to occur in deeper waters expanding the habitat of submergent aquatic macrophytes. The microbial loop is an important part of the aquatic food web tha ...
... and protozoa from the water column which disrupts the food chain for native organisms and increases the clarity of the water, allowing photosynthesis to occur in deeper waters expanding the habitat of submergent aquatic macrophytes. The microbial loop is an important part of the aquatic food web tha ...
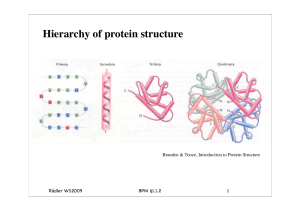
BPM§1.2_Protein Struktur.key
... This describes the overall shape of the domain structure as determined by the orientations of the secondary structures but ignores the connectivity between the secondary structures. ...
... This describes the overall shape of the domain structure as determined by the orientations of the secondary structures but ignores the connectivity between the secondary structures. ...
Protein domain

A protein domain is a conserved part of a given protein sequence and (tertiary) structure that can evolve, function, and exist independently of the rest of the protein chain. Each domain forms a compact three-dimensional structure and often can be independently stable and folded. Many proteins consist of several structural domains. One domain may appear in a variety of different proteins. Molecular evolution uses domains as building blocks and these may be recombined in different arrangements to create proteins with different functions. Domains vary in length from between about 25 amino acids up to 500 amino acids in length. The shortest domains such as zinc fingers are stabilized by metal ions or disulfide bridges. Domains often form functional units, such as the calcium-binding EF hand domain of calmodulin. Because they are independently stable, domains can be ""swapped"" by genetic engineering between one protein and another to make chimeric proteins.














![[Fe 4 S 4 Cys 4 ] 1](http://s1.studyres.com/store/data/008100934_1-bb8d7235eb07199e709035eea64be997-300x300.png)