Study Guide Energy
... 2.____________ is given off. 3. In a battery __________________ energy is changed to electrical energy. 4. In which substance (solids, liquids, gases) do particles move the slowest? ____________ 5. In which substance (solids, liquids, gases) do particles move the fastest? ____________ 6. As a substa ...
... 2.____________ is given off. 3. In a battery __________________ energy is changed to electrical energy. 4. In which substance (solids, liquids, gases) do particles move the slowest? ____________ 5. In which substance (solids, liquids, gases) do particles move the fastest? ____________ 6. As a substa ...
Classifying Matter and the Periodic Table
... I Conservation of Energy: In an isolated system, the total amount of energy, including heat, is conserved. II Entropy or disorder Energy always goes from a more useful to a less useful form. ...
... I Conservation of Energy: In an isolated system, the total amount of energy, including heat, is conserved. II Entropy or disorder Energy always goes from a more useful to a less useful form. ...
Energy and its forms
... Energy can be conserved in Non-Mechanical forms The chemical energy in a battery transforms into electrical energy Any reaction where more energy is given off than is used to start it is Exogonic An Endogonic reaction absorbs energy and causes cooling ...
... Energy can be conserved in Non-Mechanical forms The chemical energy in a battery transforms into electrical energy Any reaction where more energy is given off than is used to start it is Exogonic An Endogonic reaction absorbs energy and causes cooling ...
Energy Unit Class Notes
... Energy – the ability to do work (joules) - kinetic and potential Types of Energy 1. Mechanical – the energy of movement or position (KE + PE) - sound is an example of mechanical energy 2. Thermal (heat) energy – total energy of the moving molecules within a substance Temperature – the average kineti ...
... Energy – the ability to do work (joules) - kinetic and potential Types of Energy 1. Mechanical – the energy of movement or position (KE + PE) - sound is an example of mechanical energy 2. Thermal (heat) energy – total energy of the moving molecules within a substance Temperature – the average kineti ...
Energy and Matter Notes
... 3. Weight: Force produced by _________________ acting on a mass. This is ____________ In different locations. ...
... 3. Weight: Force produced by _________________ acting on a mass. This is ____________ In different locations. ...
Name: Date: Pod: Name: Date: Pod: Name: Date: Pod: Do Now
... Which of the following explains the total amount of kinetic What is the rolling motion of boiling water an example of? energy contained in the particles of a substance? a. Conduction a. Temperature b. Convection b. Heat c. Radiation c. Thermal energy d. Insulation d. Kinetic energy Label the parts o ...
... Which of the following explains the total amount of kinetic What is the rolling motion of boiling water an example of? energy contained in the particles of a substance? a. Conduction a. Temperature b. Convection b. Heat c. Radiation c. Thermal energy d. Insulation d. Kinetic energy Label the parts o ...
Conservation of Energy
... Conservation of Energy • Energy will always be energy, no matter what form it is in • Energy can be transferred from one type of energy to another • Potential Energy to Kinetic Energy • Kinetic Energy to Heat Energy • Heat Energy to Light Energy etc….. Any examples? ...
... Conservation of Energy • Energy will always be energy, no matter what form it is in • Energy can be transferred from one type of energy to another • Potential Energy to Kinetic Energy • Kinetic Energy to Heat Energy • Heat Energy to Light Energy etc….. Any examples? ...
Sample outline for Cornell Notes
... An object’s gravitational potential energy depends on its weight and on its height relative to a reference point. GPE = Weight x Height Example; Skier with greater mass will have greater GPE if jumping from = heights B. Elastic Potential Energy 6) EPE is the energy of objects due to it being str ...
... An object’s gravitational potential energy depends on its weight and on its height relative to a reference point. GPE = Weight x Height Example; Skier with greater mass will have greater GPE if jumping from = heights B. Elastic Potential Energy 6) EPE is the energy of objects due to it being str ...
final1-executive-summary-harwin
... performance figures of U-value (0.4 W/m²/K) and VLT (75%) comparable or even superior to existing triple glazing can be achieved at almost 50% lower weight. The new extremely stiff and thermally insulating frame, based on polymer foam coreglass fiber reinforced polymer skin materials offers addition ...
... performance figures of U-value (0.4 W/m²/K) and VLT (75%) comparable or even superior to existing triple glazing can be achieved at almost 50% lower weight. The new extremely stiff and thermally insulating frame, based on polymer foam coreglass fiber reinforced polymer skin materials offers addition ...
Motion Along a Straight Line at Constant
... Gain in kinetic energy is related to the loss in potential energy Take care! System maybe perfect or energy may be lost to the work done against air resistance and friction etc ...
... Gain in kinetic energy is related to the loss in potential energy Take care! System maybe perfect or energy may be lost to the work done against air resistance and friction etc ...
Motion Along a Straight Line at Constant
... Gain in kinetic energy is related to the loss in potential energy Take care! System maybe perfect or energy may be lost to the work done against air resistance and friction etc ...
... Gain in kinetic energy is related to the loss in potential energy Take care! System maybe perfect or energy may be lost to the work done against air resistance and friction etc ...
Chapter-9-Energy-notes
... The unit of work is Newton-meters or Nm is called ______________________. ...
... The unit of work is Newton-meters or Nm is called ______________________. ...
Chemistry of Life
... • Oxygen and Hydrogen are bonded together by ________ electrons, but the Oxygen atom _________the shared electrons closer to it, creating negative and positive sides of the water molecules. Water has a partial negative charge due to the extra unshared e- that Oxygen and a partial + charge near the h ...
... • Oxygen and Hydrogen are bonded together by ________ electrons, but the Oxygen atom _________the shared electrons closer to it, creating negative and positive sides of the water molecules. Water has a partial negative charge due to the extra unshared e- that Oxygen and a partial + charge near the h ...
$doc.title
... Colloidal quantum dots (QD), also known as semiconductor nanocrystals, are a promising material platform for solution-‐processable optoelectronic devices, such as solar cells, light-‐ emitting diodes, thermoelectric ...
... Colloidal quantum dots (QD), also known as semiconductor nanocrystals, are a promising material platform for solution-‐processable optoelectronic devices, such as solar cells, light-‐ emitting diodes, thermoelectric ...
Chapter 10: Heat Energy
... HOW IS ENERGY RELATED TO MOTION? • The words hot, warm, cool, and cold are words used to describe the temperature of something. ...
... HOW IS ENERGY RELATED TO MOTION? • The words hot, warm, cool, and cold are words used to describe the temperature of something. ...
Vocabulary for Energy Unit
... the movement of heated particles from one location to another Density- mass divided by volume. How much matter is in a volume Dependent variable - the thing in the experiment that changes because of what you did. Elastic Potential energyElectrical energy –energy in the form of a moving charged parti ...
... the movement of heated particles from one location to another Density- mass divided by volume. How much matter is in a volume Dependent variable - the thing in the experiment that changes because of what you did. Elastic Potential energyElectrical energy –energy in the form of a moving charged parti ...
L29_AS2_2008_09_KE_GPE_Efficiency
... Gain in kinetic energy is related to the loss in potential energy Take care! System maybe perfect or energy may be lost to the work done against air resistance and friction etc ...
... Gain in kinetic energy is related to the loss in potential energy Take care! System maybe perfect or energy may be lost to the work done against air resistance and friction etc ...
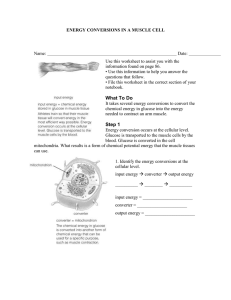
Mechanical & Thermal Energy Energy
... The sum of all kinetic energies of all the particles comprising an object is thermal energy. (most matter expands as its thermal energy increases) The faster molecules are moving, the more thermal energy they have; which is why balls go farther in warm weather than cold. ...
... The sum of all kinetic energies of all the particles comprising an object is thermal energy. (most matter expands as its thermal energy increases) The faster molecules are moving, the more thermal energy they have; which is why balls go farther in warm weather than cold. ...
Chapter 7: Quantum Mechanical Model of Atom
... ground state (lowest energy level) into higher energy levels (excited states). – Electrons prefer ground state (more stable). ...
... ground state (lowest energy level) into higher energy levels (excited states). – Electrons prefer ground state (more stable). ...