Proteins File
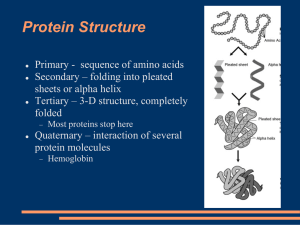
... Tertiary structure This is the overall arrangement of all the atoms in the protein, i.e., its overall shape. Every protein has a natural tertiary structure – most stable shape. It is active only in that shape. Tertiary structure is determined by primary structure. Tertiary structure is stabil ...
... Tertiary structure This is the overall arrangement of all the atoms in the protein, i.e., its overall shape. Every protein has a natural tertiary structure – most stable shape. It is active only in that shape. Tertiary structure is determined by primary structure. Tertiary structure is stabil ...
File - SMIC Nutrition Science
... 6. The making of protein from 20 amino acids was compared in the chapter to the use of the English alphabet (26 letters) to make words and speak the English language. Why was this such a fitting analogy? ...
... 6. The making of protein from 20 amino acids was compared in the chapter to the use of the English alphabet (26 letters) to make words and speak the English language. Why was this such a fitting analogy? ...
2016 N1 Week 4
... Warm UP Identify the following properties as either a carbohydrate or a lipid: 1. Does not dissolve in water. 2. The monomer is monosaccharide. 3. This molecule is not a true polymer. 4. The ratio of hydrogen to oxygen is 2:1. 5. Identify the following as a carb or lipid. B A ...
... Warm UP Identify the following properties as either a carbohydrate or a lipid: 1. Does not dissolve in water. 2. The monomer is monosaccharide. 3. This molecule is not a true polymer. 4. The ratio of hydrogen to oxygen is 2:1. 5. Identify the following as a carb or lipid. B A ...
ORGANIC CHEMISTRY SEMINAR Professor Jeff Kelly Biological and Chemical Approaches to Adapt
... proteome function by controlling ribosomal protein synthesis, chaperone and enzyme mediated protein folding, protein trafficking, proteindegradation and the like. Stress responsive signaling pathways match proteostasis network capacity with demand in each subcellular compartment to maintain cellular ...
... proteome function by controlling ribosomal protein synthesis, chaperone and enzyme mediated protein folding, protein trafficking, proteindegradation and the like. Stress responsive signaling pathways match proteostasis network capacity with demand in each subcellular compartment to maintain cellular ...
Ubiquitin
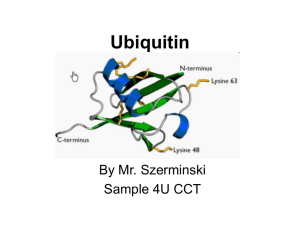
... Topics to be discussed • General info: - it is a regulatory protein that has been found in almost all tissues of eukaryotes - one of its functions: it directs protein recycling - can attach to proteins and label them for destruction. - discovery won the Nobel Prize for chemistry in 2004 ...
... Topics to be discussed • General info: - it is a regulatory protein that has been found in almost all tissues of eukaryotes - one of its functions: it directs protein recycling - can attach to proteins and label them for destruction. - discovery won the Nobel Prize for chemistry in 2004 ...
Macromolecules of life: Structure-function and Bioinformatics 356
... molecule DNA interactions. Chemical reactivity of amino acids. Domain structures of proteins and Ramachandran plots. Protein folding, sequence motifs and domains, higher order and supramolecular structure, self-assembly, conjugated proteins, post-translational modifications, conjugated proteins and ...
... molecule DNA interactions. Chemical reactivity of amino acids. Domain structures of proteins and Ramachandran plots. Protein folding, sequence motifs and domains, higher order and supramolecular structure, self-assembly, conjugated proteins, post-translational modifications, conjugated proteins and ...
proteins 2014-2015 net
... Single, spiral chain of amino acids stabilized by hydrogen bonds. Rolling up on the surface of an imaginary cylinder (+ or -). ...
... Single, spiral chain of amino acids stabilized by hydrogen bonds. Rolling up on the surface of an imaginary cylinder (+ or -). ...
Proteins for Growth and Repair
... manufacturing of hormones, enzymes, stimulation of the immune system and other compounds that provide fuel for your body. ...
... manufacturing of hormones, enzymes, stimulation of the immune system and other compounds that provide fuel for your body. ...
For complex multicellular organisms to function, individual
... Michael Hinczewski, Case Western Reserve University ...
... Michael Hinczewski, Case Western Reserve University ...
Protein Stability - Chemistry at Winthrop University
... 3. these domains interact to form the globular core of a protein. 4. The complex domains interact through surface contacts. ...
... 3. these domains interact to form the globular core of a protein. 4. The complex domains interact through surface contacts. ...
Document
... monoglucosylated oligosaccharide and hydrophobic segments of the unfolded glycoprotein (via their polypeptide binding or chaperone sites). Glycoprotein dissociation involves not only the action of glucosidase II to remove the terminal glucose residue but also a change in affinity of the polypeptide ...
... monoglucosylated oligosaccharide and hydrophobic segments of the unfolded glycoprotein (via their polypeptide binding or chaperone sites). Glycoprotein dissociation involves not only the action of glucosidase II to remove the terminal glucose residue but also a change in affinity of the polypeptide ...
Protein Structure
... MILK PROTEIN: “Curds” and “Whey” • Milk contains two major proteins: – “casein” found in the curds (cheese) – “whey” found in the liquid left over after cheese is made ...
... MILK PROTEIN: “Curds” and “Whey” • Milk contains two major proteins: – “casein” found in the curds (cheese) – “whey” found in the liquid left over after cheese is made ...
Membrane Proteins Integral membrane proteins often contain
... Integral membrane proteins often contain helical segments of appropriate length to span the lipid bilayer. In a protein that has a single segment that spans the membrane, the helix usually only contains hydrophobic residues and is called a single-span membrane protein. In transmembrane proteins with ...
... Integral membrane proteins often contain helical segments of appropriate length to span the lipid bilayer. In a protein that has a single segment that spans the membrane, the helix usually only contains hydrophobic residues and is called a single-span membrane protein. In transmembrane proteins with ...
Übung: Monte Carlo, Molecular Dynamics
... amino acids which are separated by one residue (i,i+2) and those separated by many residues. Why will this be a very bad approximation ? 8. I am working with a lattice model for a protein. Describe how I would use the Boltzmann relation to work out the absolute probability for a certain configuratio ...
... amino acids which are separated by one residue (i,i+2) and those separated by many residues. Why will this be a very bad approximation ? 8. I am working with a lattice model for a protein. Describe how I would use the Boltzmann relation to work out the absolute probability for a certain configuratio ...
Protein Structure and Folding
... 1. Use SCOP (Structural Classification Of Proteins) http://scop.mrc-lmb.cam.ac.uk/scop/ to classify PDB entry 1tml. 2. Name the fold of central domain of 1m6h and draw the corresponding topology diagram. 3. Classify the two domains of a metabolic regulator protein 1d66 from Baker’s yeast. 4. Use DAL ...
... 1. Use SCOP (Structural Classification Of Proteins) http://scop.mrc-lmb.cam.ac.uk/scop/ to classify PDB entry 1tml. 2. Name the fold of central domain of 1m6h and draw the corresponding topology diagram. 3. Classify the two domains of a metabolic regulator protein 1d66 from Baker’s yeast. 4. Use DAL ...
Protein Folding
... • Collapsed, with native-like 2º structure (far UV CD) • Weak or transient side-chain interactions (near UV CD) • Binds hydrophobic dyes • Many proteins form molten globules at low pH • Model for early stages of protein folding (hydrophobic collapse) ...
... • Collapsed, with native-like 2º structure (far UV CD) • Weak or transient side-chain interactions (near UV CD) • Binds hydrophobic dyes • Many proteins form molten globules at low pH • Model for early stages of protein folding (hydrophobic collapse) ...
051507
... – Side chains may have different pKas • pKa affected by charges on amino/carboxyl groups • pKa may be affected by interactions with other side chains in the larger molecule ...
... – Side chains may have different pKas • pKa affected by charges on amino/carboxyl groups • pKa may be affected by interactions with other side chains in the larger molecule ...
Biochemistry Homework
... Give the structural formula of the dipeptide formed by the reaction of alanine and glycine. State the other substance formed during this reaction. ...
... Give the structural formula of the dipeptide formed by the reaction of alanine and glycine. State the other substance formed during this reaction. ...
33-6-ET-V1-S1__biomi.. - e-Acharya Integrated E
... brain when searching for complex multifactor dependencies in data ...
... brain when searching for complex multifactor dependencies in data ...
Protein Synthesis
... Protein synthesis occurs at the ribosomes in the cytoplasm of a cell. But--DNA is in the nucleus. ...
... Protein synthesis occurs at the ribosomes in the cytoplasm of a cell. But--DNA is in the nucleus. ...
DOC
... Protein depletion by genetic means, in a very general sense including the use of RNA interference [1, 2] or CRISPR/Cas9-based methods, represents a central paradigm of modern biology to study protein functions in vivo. However, acting upstream the proteic level is a limiting factor if the turnover o ...
... Protein depletion by genetic means, in a very general sense including the use of RNA interference [1, 2] or CRISPR/Cas9-based methods, represents a central paradigm of modern biology to study protein functions in vivo. However, acting upstream the proteic level is a limiting factor if the turnover o ...
Essential amino acids and nutrition
... 1. Consider why protein is needed and what amino acids and proteins are used for in the body. Describe the symptoms you would expect a person with protein deficiency to have. ...
... 1. Consider why protein is needed and what amino acids and proteins are used for in the body. Describe the symptoms you would expect a person with protein deficiency to have. ...
Cardiff International School Dhaka (CISD) Lost Class Make Up
... Q1. a) The primary structure of a protein is the level of protein structure which refers to the specific sequence of amino acids. When two amino acids are in such a position that the carboxyl groups of each amino acid are adjacent to each other, they can be combined by undergoing a dehydration react ...
... Q1. a) The primary structure of a protein is the level of protein structure which refers to the specific sequence of amino acids. When two amino acids are in such a position that the carboxyl groups of each amino acid are adjacent to each other, they can be combined by undergoing a dehydration react ...
Protein folding

Protein folding is the process by which a protein structure assumes its functional shape or conformation. It is the physical process by which a polypeptide folds into its characteristic and functional three-dimensional structure from random coil.Each protein exists as an unfolded polypeptide or random coil when translated from a sequence of mRNA to a linear chain of amino acids. This polypeptide lacks any stable (long-lasting) three-dimensional structure (the left hand side of the first figure). Amino acids interact with each other to produce a well-defined three-dimensional structure, the folded protein (the right hand side of the figure), known as the native state. The resulting three-dimensional structure is determined by the amino acid sequence (Anfinsen's dogma). Experiments beginning in the 1980s indicate the codon for an amino acid can also influence protein structure.The correct three-dimensional structure is essential to function, although some parts of functional proteins may remain unfolded, so that protein dynamics is important. Failure to fold into native structure generally produces inactive proteins, but in some instances misfolded proteins have modified or toxic functionality. Several neurodegenerative and other diseases are believed to result from the accumulation of amyloid fibrils formed by misfolded proteins. Many allergies are caused by incorrect folding of some proteins, because the immune system does not produce antibodies for certain protein structures.