Chapter 4-Arrangement of Electrons in Atoms
... 2. What is the relationship between wavelength and frequency? 3. Using the equation below, solve the following: v= c ...
... 2. What is the relationship between wavelength and frequency? 3. Using the equation below, solve the following: v= c ...
Chapter 5 Sec. 2 Bohr`s Model and the Quantum Mechanical Model
... He explained that electrons can act like _____________________________. He also showed that electrons on circular orbits can only have _____________________ numbers of wavelengths. o de Broglie predicted that all moving particles have wave characteristics. de Broglie knew that if an electron act ...
... He explained that electrons can act like _____________________________. He also showed that electrons on circular orbits can only have _____________________ numbers of wavelengths. o de Broglie predicted that all moving particles have wave characteristics. de Broglie knew that if an electron act ...
NIELS BOHR power point22222
... 2) Each orbit has an energy associated with it. For example the orbit closest to the nucleus has an energy E1, the next closest E2 and so on. 3) Light is emitted when an electron jumps from a higher orbit to a lower orbit and absorbed when it jumps from a lower to higher orbit. 4) The energy and fre ...
... 2) Each orbit has an energy associated with it. For example the orbit closest to the nucleus has an energy E1, the next closest E2 and so on. 3) Light is emitted when an electron jumps from a higher orbit to a lower orbit and absorbed when it jumps from a lower to higher orbit. 4) The energy and fre ...
Atom 2 - UF Physics
... Lack of dependence with light intensity of the photoelectric effect Lack of quantization of atomic emission and absorption spectra Collapse of atoms ...
... Lack of dependence with light intensity of the photoelectric effect Lack of quantization of atomic emission and absorption spectra Collapse of atoms ...
PHYSICS 4E QUIZ 4 SPRING QUARTER 2010 PROF. HIRSCH
... (c) Show that your result in (b) agrees with the prediction of the Bohr atom for this n and Z. Alternatively, if you didn't find n and Z in (a), use the fact that your result in (b) should agree with the Bohr atom prediction to deduce the values of n and Z. Problem 3 (10 pts) (a) Sodium has atomic n ...
... (c) Show that your result in (b) agrees with the prediction of the Bohr atom for this n and Z. Alternatively, if you didn't find n and Z in (a), use the fact that your result in (b) should agree with the Bohr atom prediction to deduce the values of n and Z. Problem 3 (10 pts) (a) Sodium has atomic n ...
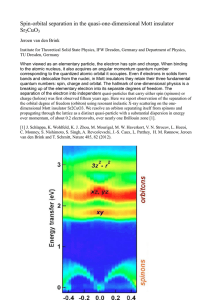
Spin-orbital separation in the quasi-one
... Institute for Theoretical Solid State Physics, IFW Dresden, Germany and Department of Physics, TU Dresden, Germany When viewed as an elementary particle, the electron has spin and charge. When binding to the atomic nucleus, it also acquires an angular momentum quantum number corresponding to the qua ...
... Institute for Theoretical Solid State Physics, IFW Dresden, Germany and Department of Physics, TU Dresden, Germany When viewed as an elementary particle, the electron has spin and charge. When binding to the atomic nucleus, it also acquires an angular momentum quantum number corresponding to the qua ...
The true nature of the atom?
... Three phenomena that could not be understood with Newtonian physics were investigated and explained by radical new ideas that would lead to a new type of physics, the field that would become quantum mechanics. ...
... Three phenomena that could not be understood with Newtonian physics were investigated and explained by radical new ideas that would lead to a new type of physics, the field that would become quantum mechanics. ...
CH1710 HW#7 (2017)-Quanta, electron config
... 3. Cobalt-60 is a radioactive isotope used to treat cancers of the brain and other tissues. A gamma ray emitted by an atom of the isotope has an energy of 1.33 MeV (million electron volts). a. If 1 eV= 1.602 x 10-19 J, what is the frequency (in Hz) of this gamma ray ? ...
... 3. Cobalt-60 is a radioactive isotope used to treat cancers of the brain and other tissues. A gamma ray emitted by an atom of the isotope has an energy of 1.33 MeV (million electron volts). a. If 1 eV= 1.602 x 10-19 J, what is the frequency (in Hz) of this gamma ray ? ...
Chemistry 354 - Homework Set IV
... mass of a hydrogen atom; the mass of Haystacks Calhoun (erstwhile professional wrestler); the distance between the n = 1 and n = 2 energy levels of an electron in a one-dimensional box of length 1 nm; and finally, the speed of light. Make a table of these numbers, and note that the “atomic-scale” qu ...
... mass of a hydrogen atom; the mass of Haystacks Calhoun (erstwhile professional wrestler); the distance between the n = 1 and n = 2 energy levels of an electron in a one-dimensional box of length 1 nm; and finally, the speed of light. Make a table of these numbers, and note that the “atomic-scale” qu ...
File
... h is Planck’s constant h= 6.63 x 10-34 J.s u is equal to the frequency of light. u = f = frequency ...
... h is Planck’s constant h= 6.63 x 10-34 J.s u is equal to the frequency of light. u = f = frequency ...
3.3 Review Name________________________________ Period_______Date_____________________
... ______ 15. Labeled each energy level in his atomic model with the principal quantum number, n. ______ 16. Wrote the equation E=h. ______ 17. The constant, h, is named for this scientist. ______ 18. Stated that you cannot observe or measure the “orbit” of an electron. Part IV: Drawing 19. Label the ...
... ______ 15. Labeled each energy level in his atomic model with the principal quantum number, n. ______ 16. Wrote the equation E=h. ______ 17. The constant, h, is named for this scientist. ______ 18. Stated that you cannot observe or measure the “orbit” of an electron. Part IV: Drawing 19. Label the ...
Chapter 13 – Electrons in Atoms
... Electron Arrangement in Atoms Physics and the Quantum Mechanical Model ...
... Electron Arrangement in Atoms Physics and the Quantum Mechanical Model ...
e - Purdue Physics - Purdue University
... of energy, the energy of the red-color photon. •The 656 nm emission line from H has a frequency f=4.57×1014 Hz. A photon of this color has an energy of hf= 3.03×10−19 J (1.89 eV). ...
... of energy, the energy of the red-color photon. •The 656 nm emission line from H has a frequency f=4.57×1014 Hz. A photon of this color has an energy of hf= 3.03×10−19 J (1.89 eV). ...
Modern Physics
... Calculate the location at which the radial probability density is a maximum for the 2-s state of the hydrogen atom. Then calculate the expectation value of the radial coordinate in this state. Which answer if either is consistent with the Bohr model ...
... Calculate the location at which the radial probability density is a maximum for the 2-s state of the hydrogen atom. Then calculate the expectation value of the radial coordinate in this state. Which answer if either is consistent with the Bohr model ...
LT1: Electron.NOTES - Simpson County Schools
... Why did the wave model of light need a revision? ________________________________________________________________________________________________________ ...
... Why did the wave model of light need a revision? ________________________________________________________________________________________________________ ...
Radiation and quantised orbits
... But in his model, Bohr directly assumed that the orbits in which electrons revolve are NON RADIATING. According to him, the orbits are quantised, but then what happened to classical theory of electrodynamics? ...
... But in his model, Bohr directly assumed that the orbits in which electrons revolve are NON RADIATING. According to him, the orbits are quantised, but then what happened to classical theory of electrodynamics? ...
Chapter 1 The Bohr Atom 1 Introduction
... You will find that these three definitions are used over and over again in atomic physics, so, these are worth remembering. For example, these definitions can be used to simply the calculation performed in Eq. 6. Finally, a third parameter that can be calculated using the Bohr model is the total ene ...
... You will find that these three definitions are used over and over again in atomic physics, so, these are worth remembering. For example, these definitions can be used to simply the calculation performed in Eq. 6. Finally, a third parameter that can be calculated using the Bohr model is the total ene ...
LEP 5.1.03 -15 Franck-Hertz experiment with Ne-tube
... among those states producing radiation whose frequency is determined by the energy differences between states can be derived from the quantum mechanics which replaced classical mechanics when dealing with structures as small as atoms. It seems reasonable from the Bohr model that just as electrons ma ...
... among those states producing radiation whose frequency is determined by the energy differences between states can be derived from the quantum mechanics which replaced classical mechanics when dealing with structures as small as atoms. It seems reasonable from the Bohr model that just as electrons ma ...
(Bohr Model And X-Rays) Part-1
... Neils Bohr gave forward his model of atom with answeres most of the flaws that Rutherford model had. This model introduced quantization principal and this model is regarded as first quantum mechanical model. We will explore in detail, this structure of atom. We will also look into X - ray formation ...
... Neils Bohr gave forward his model of atom with answeres most of the flaws that Rutherford model had. This model introduced quantization principal and this model is regarded as first quantum mechanical model. We will explore in detail, this structure of atom. We will also look into X - ray formation ...
Document
... If an electron falls from the third orbit to the first, how much energy does it lose? ...
... If an electron falls from the third orbit to the first, how much energy does it lose? ...
Bohr Model of the Hydrogen Atom
... (c) What is the energy of the ground state, measured in eV (electron volts)? As you likely know from high school physics and/or chemistry courses, the Bohr model predicts the energies of the hydrogen atom incredibly accurately: in fact, not until the introduction of relativistic quantum mechanics wa ...
... (c) What is the energy of the ground state, measured in eV (electron volts)? As you likely know from high school physics and/or chemistry courses, the Bohr model predicts the energies of the hydrogen atom incredibly accurately: in fact, not until the introduction of relativistic quantum mechanics wa ...
Problem Set 05
... rotational energies of the molecule. [Result: n 2 2 /md 2 ] b) The wavelength of the photon needed to excite the molecule from the n to the n+1 rotational state. [Result: λ=2πcmd 2/(2n+1)] c) The wavelength of the photon need to excite € a nitrogen molecule (N2) from its ground state (n=0) to the ...
... rotational energies of the molecule. [Result: n 2 2 /md 2 ] b) The wavelength of the photon needed to excite the molecule from the n to the n+1 rotational state. [Result: λ=2πcmd 2/(2n+1)] c) The wavelength of the photon need to excite € a nitrogen molecule (N2) from its ground state (n=0) to the ...
James Franck

James Franck (26 August 1882 – 21 May 1964) was a German physicist who won the 1925 Nobel Prize for Physics with Gustav Hertz ""for their discovery of the laws governing the impact of an electron upon an atom"". He completed his doctorate in 1906 and his habilitation in 1911 at the Frederick William University in Berlin, where he lectured and taught until 1918, having reached the position of professor extraordinarius. He served as a volunteer in the German Army during World War I. He was seriously injured in 1917 in a gas attack and was awarded the Iron Cross 1st Class.Franck became the Head of the Physics Division of the Kaiser Wilhelm Gesellschaft for Physical Chemistry. In 1920, Franck became professor ordinarius of experimental physics and Director of the Second Institute for Experimental Physics at the University of Göttingen. While there he worked on quantum physics with Max Born, who was Director of the Institute of Theoretical Physics. His work included the Franck–Hertz experiment, an important confirmation of the Bohr model of the atom. He promoted the careers of women in physics, notably Lise Meitner, Hertha Sponer and Hilde Levi.After the NSDAP came to power in Germany in 1933, Franck resigned his post in protest against the dismissal of fellow academics. He assisted Frederick Lindemann in helping dismissed Jewish scientists find work overseas, before he left Germany in November 1933. After a year at the Niels Bohr Institute in Denmark, he moved to the United States, where he worked at Johns Hopkins University in Baltimore and then the University of Chicago. During this period he became interested in photosynthesis.Franck participated in the Manhattan Project during World War II as Director of the Chemistry Division of the Metallurgical Laboratory. He was also the chairman of the Committee on Political and Social Problems regarding the atomic bomb, which is best known for the compilation of the Franck Report, which recommended that the atomic bombs not be used on the Japanese cities without warning.