![Chapter7_1 - Department of Chemistry [FSU]](http://s1.studyres.com/store/data/016128835_1-aea3c1aec04363d6cbf538e8faf80e45-300x300.png)
Chapter7_1 - Department of Chemistry [FSU]
... The Nature of Light • electromagnetic radiation travels in waves • at the speed of light (in vacuum, c = "#) • intensity is either amplitude or number of photons/second • energy of light is quantized, E = h" • the energy of atom is also quantized, E = nh" h = 6.626$10-34 J•s ...
... The Nature of Light • electromagnetic radiation travels in waves • at the speed of light (in vacuum, c = "#) • intensity is either amplitude or number of photons/second • energy of light is quantized, E = h" • the energy of atom is also quantized, E = nh" h = 6.626$10-34 J•s ...
Lecture-2: Atomic Structure
... from a metal surface when light shines on it. The discovery of the photoelectric effect could not be explained by the electromagnetic theory of light. Albert Einstein developed the quantum theory of light in ...
... from a metal surface when light shines on it. The discovery of the photoelectric effect could not be explained by the electromagnetic theory of light. Albert Einstein developed the quantum theory of light in ...
科目名 Course Title Extreme Laser Physics [極限レーザー物理E] 講義
... Interactions between optical field and atomic, molecular, and materials system have been providing interesting issues in physics. This course covers the basics of ultrafast optics and atomic physics, necessary to understand rapidly growing research field in atomic, molecular, and optical physics, wi ...
... Interactions between optical field and atomic, molecular, and materials system have been providing interesting issues in physics. This course covers the basics of ultrafast optics and atomic physics, necessary to understand rapidly growing research field in atomic, molecular, and optical physics, wi ...
CHM1045 - Michael Blaber
... 5. An electron transitions from the n = 2 to the n = 4 quantum state in Bohr's model of the hydrogen atom. Is a photon absorbed or emitted for this electron transition? (3 points). What is the energy, frequency and wavelength of the associated photon? (9 points). Is this wavelength visible? (3 poin ...
... 5. An electron transitions from the n = 2 to the n = 4 quantum state in Bohr's model of the hydrogen atom. Is a photon absorbed or emitted for this electron transition? (3 points). What is the energy, frequency and wavelength of the associated photon? (9 points). Is this wavelength visible? (3 poin ...
Atomic Structure Origin of the elements Structure of atoms Periodic Trends
... ❧ nuclei are bombarded with a large neutron flux ❧ form highly unstable neutron rich nuclei ❧ rapidly decay to form stable neutron rich nuclei • P process ❧ Proton capture, not as common ...
... ❧ nuclei are bombarded with a large neutron flux ❧ form highly unstable neutron rich nuclei ❧ rapidly decay to form stable neutron rich nuclei • P process ❧ Proton capture, not as common ...
Chapter 4: Struct of Atom
... with their electrons stripped off, but we did not know that) at a very thin gold foil, and he observed what happened S Where did the α’s go ...
... with their electrons stripped off, but we did not know that) at a very thin gold foil, and he observed what happened S Where did the α’s go ...
Document
... in discrete packets, or quanta. The energy of these quanta is proportional to the frequency of the radiation. ...
... in discrete packets, or quanta. The energy of these quanta is proportional to the frequency of the radiation. ...
PowerPoint
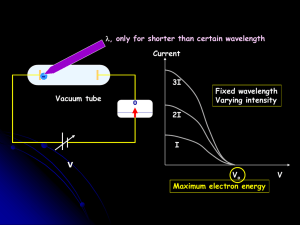
... Light consists of photons, each with a particular amount of energy, called a quantum of energy Upon collision, each photon can transfer its energy to a single electron The more photons strike the surface of the metal, the more electrons are liberated and the higher is the current ...
... Light consists of photons, each with a particular amount of energy, called a quantum of energy Upon collision, each photon can transfer its energy to a single electron The more photons strike the surface of the metal, the more electrons are liberated and the higher is the current ...
The Photoelectric Effect
... Light consists of photons, each with a particular amount of energy, called a quantum of energy Upon collision, each photon can transfer its energy to a single electron The more photons strike the surface of the metal, the more electrons are liberated and the higher is the current ...
... Light consists of photons, each with a particular amount of energy, called a quantum of energy Upon collision, each photon can transfer its energy to a single electron The more photons strike the surface of the metal, the more electrons are liberated and the higher is the current ...
PS7 - Bergen.org
... (b) Draw an electronic energy level diagram for the hydrogen atom and indicate on it the transition corresponding to the line of lowest frequency in the Balmer series. (c) Account for the existence of several series of lines in the hydrogen spectrum. What distinguishes one series of lines from anoth ...
... (b) Draw an electronic energy level diagram for the hydrogen atom and indicate on it the transition corresponding to the line of lowest frequency in the Balmer series. (c) Account for the existence of several series of lines in the hydrogen spectrum. What distinguishes one series of lines from anoth ...
Problem set #1 - U.C.C. Physics Department
... 4) Let us assume that a light bulb emits a monochromatic yellow light, at wavelength λ = 600 nm. You are standing 3 meters away from a 60 W light bulb, and looks at it. Calculate the number of photons that enter your eyes each second. You can assume that the light bulb’s emission is spherical, and t ...
... 4) Let us assume that a light bulb emits a monochromatic yellow light, at wavelength λ = 600 nm. You are standing 3 meters away from a 60 W light bulb, and looks at it. Calculate the number of photons that enter your eyes each second. You can assume that the light bulb’s emission is spherical, and t ...
Lec-23_Strachan
... As a general rule, the order that electrons fill an atom’s subshell is: Once one subshell is filled, the next electron goes into the vacant subshell that is lowest in energy Otherwise, the electron would radiate energy until it reached the subshell with the lowest energy A subshell is filled when it ...
... As a general rule, the order that electrons fill an atom’s subshell is: Once one subshell is filled, the next electron goes into the vacant subshell that is lowest in energy Otherwise, the electron would radiate energy until it reached the subshell with the lowest energy A subshell is filled when it ...
Excitation of Quantum Jumps by Collisions
... Muonic atoms are objects of the nuclear physics research. ...
... Muonic atoms are objects of the nuclear physics research. ...
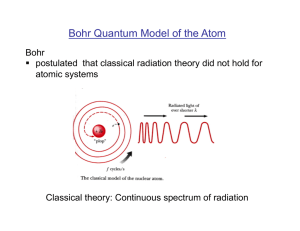
Bohr Quantum Model of the Atom
... § postulated that the electron orbital momentum is quantized Justification of Bohr’s postulates: comparison with experimental observations! ...
... § postulated that the electron orbital momentum is quantized Justification of Bohr’s postulates: comparison with experimental observations! ...
Transcript - the Cassiopeia Project
... energy state. The electron occupies a cloud instead of an orbit, but it spends most of its time at the radius predicted as an orbit by the Bohr model. AND it spends most of its time possessing the energy that an electron in that orbit would have. 410 Excited States But of course, the atom is not alw ...
... energy state. The electron occupies a cloud instead of an orbit, but it spends most of its time at the radius predicted as an orbit by the Bohr model. AND it spends most of its time possessing the energy that an electron in that orbit would have. 410 Excited States But of course, the atom is not alw ...
Frank-Hertz experiment with Neon
... electric discharges through various low-pressure gasses in evacuated glass tubes had shown that atoms will only emit light (that is, electromagnetic radiation) at certain discrete frequencies. To overcome this difficulty, Niels Bohr proposed in 1913 the now called Bohr model of the atom, which sugge ...
... electric discharges through various low-pressure gasses in evacuated glass tubes had shown that atoms will only emit light (that is, electromagnetic radiation) at certain discrete frequencies. To overcome this difficulty, Niels Bohr proposed in 1913 the now called Bohr model of the atom, which sugge ...
Bohr Model and Quantum Model
... RULE 1: Electrons can orbit only at certain allowed distances from the nucleus. RULE 2: Atoms radiate energy when an electron jumps from a higher-energy orbit to a lower-energy orbit. Also, an atom absorbs energy when an electron gets boosted from a low-energy orbit to a highenergy orbit. ...
... RULE 1: Electrons can orbit only at certain allowed distances from the nucleus. RULE 2: Atoms radiate energy when an electron jumps from a higher-energy orbit to a lower-energy orbit. Also, an atom absorbs energy when an electron gets boosted from a low-energy orbit to a highenergy orbit. ...
- BUGS McGill
... 7. For a rotor with masses m1 and m2 located at distances r1 and r2 from the center of mass show that I (moment of inertia) can be represented as a reduced mass rotating at a distance r about a fixed point. (r = the distance between m1 and m2 ). ...
... 7. For a rotor with masses m1 and m2 located at distances r1 and r2 from the center of mass show that I (moment of inertia) can be represented as a reduced mass rotating at a distance r about a fixed point. (r = the distance between m1 and m2 ). ...
Transcript - the Cassiopeia Project
... by stating that “it just didn’t happen” (even great scientists cheat sometimes!) He postulated that inside an atom, electrons only radiate energy when they jump from one allowable orbit to another, and the energy of this radiation, reveals the allowable orbits. The wavelengths of light absorbed by h ...
... by stating that “it just didn’t happen” (even great scientists cheat sometimes!) He postulated that inside an atom, electrons only radiate energy when they jump from one allowable orbit to another, and the energy of this radiation, reveals the allowable orbits. The wavelengths of light absorbed by h ...
03-02BohrAtom
... What is the energy of the n = 5 orbital? What is the energy of the n = 3 orbital? What is the wavelength of the photon released from a 5 to 3 transition? (Hydrogen atom) ...
... What is the energy of the n = 5 orbital? What is the energy of the n = 3 orbital? What is the wavelength of the photon released from a 5 to 3 transition? (Hydrogen atom) ...
IntroQuantumNuclearp..
... Eincoming photon = Eenergy e- needs to break away from atom + Escattered electron + Eenergy scattered photon ...
... Eincoming photon = Eenergy e- needs to break away from atom + Escattered electron + Eenergy scattered photon ...
James Franck

James Franck (26 August 1882 – 21 May 1964) was a German physicist who won the 1925 Nobel Prize for Physics with Gustav Hertz ""for their discovery of the laws governing the impact of an electron upon an atom"". He completed his doctorate in 1906 and his habilitation in 1911 at the Frederick William University in Berlin, where he lectured and taught until 1918, having reached the position of professor extraordinarius. He served as a volunteer in the German Army during World War I. He was seriously injured in 1917 in a gas attack and was awarded the Iron Cross 1st Class.Franck became the Head of the Physics Division of the Kaiser Wilhelm Gesellschaft for Physical Chemistry. In 1920, Franck became professor ordinarius of experimental physics and Director of the Second Institute for Experimental Physics at the University of Göttingen. While there he worked on quantum physics with Max Born, who was Director of the Institute of Theoretical Physics. His work included the Franck–Hertz experiment, an important confirmation of the Bohr model of the atom. He promoted the careers of women in physics, notably Lise Meitner, Hertha Sponer and Hilde Levi.After the NSDAP came to power in Germany in 1933, Franck resigned his post in protest against the dismissal of fellow academics. He assisted Frederick Lindemann in helping dismissed Jewish scientists find work overseas, before he left Germany in November 1933. After a year at the Niels Bohr Institute in Denmark, he moved to the United States, where he worked at Johns Hopkins University in Baltimore and then the University of Chicago. During this period he became interested in photosynthesis.Franck participated in the Manhattan Project during World War II as Director of the Chemistry Division of the Metallurgical Laboratory. He was also the chairman of the Committee on Political and Social Problems regarding the atomic bomb, which is best known for the compilation of the Franck Report, which recommended that the atomic bombs not be used on the Japanese cities without warning.

![科目名 Course Title Extreme Laser Physics [極限レーザー物理E] 講義](http://s1.studyres.com/store/data/003538965_1-4c9ae3641327c1116053c260a01760fe-300x300.png)