
PRESS-RELEASE Max Planck Institute of Quantum
... by Prof. Ferenc Krausz at the Max Planck Institute of Quantum Optics and the LudwigMaximilians-Universität in Munich, in collaborations with researchers from the United States and Saudi Arabia, have observed, for the first time, the quantum-mechanical behaviour occurring at the location in a noble g ...
... by Prof. Ferenc Krausz at the Max Planck Institute of Quantum Optics and the LudwigMaximilians-Universität in Munich, in collaborations with researchers from the United States and Saudi Arabia, have observed, for the first time, the quantum-mechanical behaviour occurring at the location in a noble g ...
pdf-file - Max Planck Institut für Quantenoptik
... by Prof. Ferenc Krausz at the Max Planck Institute of Quantum Optics and the LudwigMaximilians-Universität in Munich, in collaborations with researchers from the United States and Saudi Arabia, have observed, for the first time, the quantum-mechanical behaviour occurring at the location in a noble g ...
... by Prof. Ferenc Krausz at the Max Planck Institute of Quantum Optics and the LudwigMaximilians-Universität in Munich, in collaborations with researchers from the United States and Saudi Arabia, have observed, for the first time, the quantum-mechanical behaviour occurring at the location in a noble g ...
Document
... ACT: What about the radius? Z=3, n=1 1. larger than H atom 2. same as H atom 3. smaller than H atom ...
... ACT: What about the radius? Z=3, n=1 1. larger than H atom 2. same as H atom 3. smaller than H atom ...
Chapter 3 notes
... should gradually lose energy and spiral inward toward the nucleus. (BOOM! No more atom.) ...
... should gradually lose energy and spiral inward toward the nucleus. (BOOM! No more atom.) ...
cp351c04
... decays to lower states, with emission of photon (or other mechanism for energy transfer). Metastable State: “sort of stable” state state with a longer life time than ordinary excited states lifetime ~ 1E-3 s vs. 1E-8 s for ordinary states Three kinds of transitions h ...
... decays to lower states, with emission of photon (or other mechanism for energy transfer). Metastable State: “sort of stable” state state with a longer life time than ordinary excited states lifetime ~ 1E-3 s vs. 1E-8 s for ordinary states Three kinds of transitions h ...
NAME PERIOD ______ DATE Chapter 5 Sec. 2
... Calculate the wavelength of the photon emitted when the electron drops from the n = 6 orbit to the n = 1 orbit. The following values may be helpful: h = 6.626 x 10-34 J●s and c = 3.00 x 108 m/s. You must show your work to get the points. ...
... Calculate the wavelength of the photon emitted when the electron drops from the n = 6 orbit to the n = 1 orbit. The following values may be helpful: h = 6.626 x 10-34 J●s and c = 3.00 x 108 m/s. You must show your work to get the points. ...
1. Introduction - Departamento de Fisica/UFPB
... The quantity of an element which is separated is proportional to the quantity of charge transported in the process, and various elements are separated into equivalent weights by the same quantity of charge. From this, Faraday concluded: There are "atoms" of electricity - it was only after 70 years t ...
... The quantity of an element which is separated is proportional to the quantity of charge transported in the process, and various elements are separated into equivalent weights by the same quantity of charge. From this, Faraday concluded: There are "atoms" of electricity - it was only after 70 years t ...
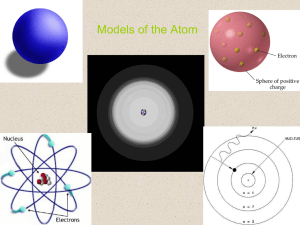
Models of the Atom
... the chemical properties of an element? No, to describe the chemical properties of an element we needed a model that better describes the behavior of electrons. ...
... the chemical properties of an element? No, to describe the chemical properties of an element we needed a model that better describes the behavior of electrons. ...
Early Modern Physics
... • already went over kinematics • Rutherford scattering can either be off a heavier object (nuclei) change in angle but little energy loss “multiple scattering” • or off light target (electrons) where can transfer energy but little angular change (energy loss due to ionization, also produces “del ...
... • already went over kinematics • Rutherford scattering can either be off a heavier object (nuclei) change in angle but little energy loss “multiple scattering” • or off light target (electrons) where can transfer energy but little angular change (energy loss due to ionization, also produces “del ...
Bohr`s atomic model
... of the hydrogen atom, but there was no clear physical reasoning to support his assumption of angular momentum quantization. 9. “New” quantum theory considers the electron to be a wave. It discards the notion of orbits and replaces it with the notion of orbitals. Orbitals are stationary (standing) wa ...
... of the hydrogen atom, but there was no clear physical reasoning to support his assumption of angular momentum quantization. 9. “New” quantum theory considers the electron to be a wave. It discards the notion of orbits and replaces it with the notion of orbitals. Orbitals are stationary (standing) wa ...
Electronic structure (download)
... The Bohr model for the electrons Electronic structure – how the electrons are arranged inside the atom Applying the quantum principle of energy ...
... The Bohr model for the electrons Electronic structure – how the electrons are arranged inside the atom Applying the quantum principle of energy ...
PPT
... • Predicts available energy states agreeing with Bohr. • Don’t have definite electron position, only a probability function. • Each orbital can have 0 angular momentum! • Each electron state labeled by 4 numbers: n = principal quantum number (1, 2, 3, …) l = angular momentum (0, 1, 2, … n-1) Coming ...
... • Predicts available energy states agreeing with Bohr. • Don’t have definite electron position, only a probability function. • Each orbital can have 0 angular momentum! • Each electron state labeled by 4 numbers: n = principal quantum number (1, 2, 3, …) l = angular momentum (0, 1, 2, … n-1) Coming ...
The de Broglie-Bohr Model for the Hydrogen Atom
... (centripetal and electron-nucleus) on an electron executing a circular orbit of radius R about the nucleus, and then arbitarily quantizing its angular momentum. Finally by fiat he declared that the electron was in a non-radiating stationary state because an orbiting (accelerating) charge radiates en ...
... (centripetal and electron-nucleus) on an electron executing a circular orbit of radius R about the nucleus, and then arbitarily quantizing its angular momentum. Finally by fiat he declared that the electron was in a non-radiating stationary state because an orbiting (accelerating) charge radiates en ...
Bohr Revisited: Model and spectral lines of helium
... a model of excited helium where one electron is in ground state and the other is excited, in which some shielding will take effect, changing the energy emitted as the excited electron transitions to a lower state. One of the most influential factors in why the Bohr and Bohrlike models were abandoned ...
... a model of excited helium where one electron is in ground state and the other is excited, in which some shielding will take effect, changing the energy emitted as the excited electron transitions to a lower state. One of the most influential factors in why the Bohr and Bohrlike models were abandoned ...
Early Modern Physics
... • Late 19th Century: try to derive Wien and StefanBoltzman Laws and shape of observed light spectra • used Statistical Mechanics (we’ll do later in 461) to determine relative probability for any wavelength l • need::number of states (“nodes”) for any l - energy of any state probability versus energy ...
... • Late 19th Century: try to derive Wien and StefanBoltzman Laws and shape of observed light spectra • used Statistical Mechanics (we’ll do later in 461) to determine relative probability for any wavelength l • need::number of states (“nodes”) for any l - energy of any state probability versus energy ...
Chapter7 - FSU Chemistry
... Practice! Problems *7.76. A ground state H atom absorbs a photon of wavelength 94.91 nm, and its electron attains a higher energy level. The atom then emits two photons: one of wavelength 1281 nm to reach an intermediate level, and a second to return to the ground state. (a) What level did the elec ...
... Practice! Problems *7.76. A ground state H atom absorbs a photon of wavelength 94.91 nm, and its electron attains a higher energy level. The atom then emits two photons: one of wavelength 1281 nm to reach an intermediate level, and a second to return to the ground state. (a) What level did the elec ...
Kvantfysik Lecture Notes No. 4x
... predictions for certain experiments using multi-electron atoms. In particular, it leads to beautiful predictions for the x-ray spectra of atoms with Z > 15. For now we make the following assumptions 1. The electrons are in discrete energy shells labeled by n = 1, 2, 3 . . .. 2. The lowest energy she ...
... predictions for certain experiments using multi-electron atoms. In particular, it leads to beautiful predictions for the x-ray spectra of atoms with Z > 15. For now we make the following assumptions 1. The electrons are in discrete energy shells labeled by n = 1, 2, 3 . . .. 2. The lowest energy she ...
Topic 7: Atomic and nuclear physics 7.1 The atom
... So what holds it together? • Shortly after the discovery of the neutron, Hideki Yukawa, a Japanese physicist, postulated a strong force of attraction between nucleons that overcomes the Coulomb repulsion between protons. The existence of the force postulated by Yukawa is now well established and is ...
... So what holds it together? • Shortly after the discovery of the neutron, Hideki Yukawa, a Japanese physicist, postulated a strong force of attraction between nucleons that overcomes the Coulomb repulsion between protons. The existence of the force postulated by Yukawa is now well established and is ...
ELECTRONS IN ATOMS
... It is called a quantum. 4. Circle the letter of the term that completes the sentence correctly. A quantum of energy is the amount of energy required to a. move an electron from its present energy level to the next lower one b. maintain an electron in its present energy level c. move an electron from ...
... It is called a quantum. 4. Circle the letter of the term that completes the sentence correctly. A quantum of energy is the amount of energy required to a. move an electron from its present energy level to the next lower one b. maintain an electron in its present energy level c. move an electron from ...
Set #4
... 1. How is the quantization of the energy in the hydrogen atom similar to the quantization of the systems discussed in the 1-D infinite quantum well? How is it different? Do the quantizations originate from similar causes? (Krane, Q8, pg. 201) 2. In both the Rutherford theory and the Bohr theory, we ...
... 1. How is the quantization of the energy in the hydrogen atom similar to the quantization of the systems discussed in the 1-D infinite quantum well? How is it different? Do the quantizations originate from similar causes? (Krane, Q8, pg. 201) 2. In both the Rutherford theory and the Bohr theory, we ...
L 34 Modern Physics [1]
... • Light is a particle called a photon packets of energy moving at the speed of light! • A beam of light is thought of as a beam of photons. ...
... • Light is a particle called a photon packets of energy moving at the speed of light! • A beam of light is thought of as a beam of photons. ...
James Franck

James Franck (26 August 1882 – 21 May 1964) was a German physicist who won the 1925 Nobel Prize for Physics with Gustav Hertz ""for their discovery of the laws governing the impact of an electron upon an atom"". He completed his doctorate in 1906 and his habilitation in 1911 at the Frederick William University in Berlin, where he lectured and taught until 1918, having reached the position of professor extraordinarius. He served as a volunteer in the German Army during World War I. He was seriously injured in 1917 in a gas attack and was awarded the Iron Cross 1st Class.Franck became the Head of the Physics Division of the Kaiser Wilhelm Gesellschaft for Physical Chemistry. In 1920, Franck became professor ordinarius of experimental physics and Director of the Second Institute for Experimental Physics at the University of Göttingen. While there he worked on quantum physics with Max Born, who was Director of the Institute of Theoretical Physics. His work included the Franck–Hertz experiment, an important confirmation of the Bohr model of the atom. He promoted the careers of women in physics, notably Lise Meitner, Hertha Sponer and Hilde Levi.After the NSDAP came to power in Germany in 1933, Franck resigned his post in protest against the dismissal of fellow academics. He assisted Frederick Lindemann in helping dismissed Jewish scientists find work overseas, before he left Germany in November 1933. After a year at the Niels Bohr Institute in Denmark, he moved to the United States, where he worked at Johns Hopkins University in Baltimore and then the University of Chicago. During this period he became interested in photosynthesis.Franck participated in the Manhattan Project during World War II as Director of the Chemistry Division of the Metallurgical Laboratory. He was also the chairman of the Committee on Political and Social Problems regarding the atomic bomb, which is best known for the compilation of the Franck Report, which recommended that the atomic bombs not be used on the Japanese cities without warning.






















![L 34 Modern Physics [1]](http://s1.studyres.com/store/data/001537103_1-dca58a96feb57d01fab60ba8bdd791ec-300x300.png)