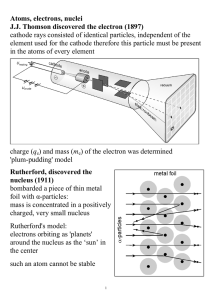
Atoms, electrons, nuclei J.J. Thomson discovered the electron (1897
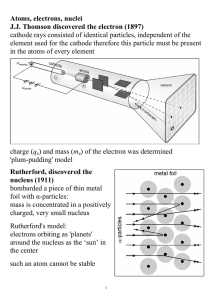
... the center such an atom cannot be stable ...
... the center such an atom cannot be stable ...
Chapter 31 Quantum Mechanics and Atomic Physics
... light as they undergo transitions from a higher to a lower atomic energy level. Our venture into the atomic world begins with the concept of the nuclear atom and continues with the Bohr model of the hydrogen atom. This model introduces many basic atomic features, including the notions of discrete en ...
... light as they undergo transitions from a higher to a lower atomic energy level. Our venture into the atomic world begins with the concept of the nuclear atom and continues with the Bohr model of the hydrogen atom. This model introduces many basic atomic features, including the notions of discrete en ...
Dr.Eman Zakaria Hegazy Quantum Mechanics and Statistical
... electron must be quantized according to L=mvr=nℏ ...
... electron must be quantized according to L=mvr=nℏ ...
Chapter 28 Atoms
... In 1926, the German physicist Erwin Schroedinger used de Brogli’s wave model to create a quantum theory of atom based on waves. The theory does not provide a simple planetary picture of an atom as in the Bohr model. In particular, the radius of the electron orbit is not like the radius of the orbit ...
... In 1926, the German physicist Erwin Schroedinger used de Brogli’s wave model to create a quantum theory of atom based on waves. The theory does not provide a simple planetary picture of an atom as in the Bohr model. In particular, the radius of the electron orbit is not like the radius of the orbit ...
Course Title (1) Brief Course Description Modern Physics involves
... Modern Physics involves the extremes of very small distances and velocities close to the speed of light. These extremes demanded new theories in the early part of the 20th century and yielded the weird and wonderful results of Einstein’s relativity theory and Schrodinger’s equation in quantum mechan ...
... Modern Physics involves the extremes of very small distances and velocities close to the speed of light. These extremes demanded new theories in the early part of the 20th century and yielded the weird and wonderful results of Einstein’s relativity theory and Schrodinger’s equation in quantum mechan ...
3. Represents an atom that has four valence electrons.
... The emission spectrum of gold shows a line of wavelength 2.676 x 10-7 m. How much energy is emitted during this transition? (A) 7.43 x 10-19 J (B) 5.30 x 10-20 J (C) 6.05 x 10-19 J (D) 3.60 x 10-20 J (E) 5.16 x 10-20 J 9. Which of the responses contains all the statements that are consistent with th ...
... The emission spectrum of gold shows a line of wavelength 2.676 x 10-7 m. How much energy is emitted during this transition? (A) 7.43 x 10-19 J (B) 5.30 x 10-20 J (C) 6.05 x 10-19 J (D) 3.60 x 10-20 J (E) 5.16 x 10-20 J 9. Which of the responses contains all the statements that are consistent with th ...
Modules to examine on the Arrangement of Electrons in Atoms website
... Modules to examine on the Arrangement of Electrons in Atoms website: (click to go directly to the different modules) EM Waves Evidence for EM Waves Catch the Wave Stadium Wave Electric Force Quantum Atom Spectral Lines Edvidence for Spectra Absorption Spectra Bohr's Atom Vibrating Charges rev. Adv B ...
... Modules to examine on the Arrangement of Electrons in Atoms website: (click to go directly to the different modules) EM Waves Evidence for EM Waves Catch the Wave Stadium Wave Electric Force Quantum Atom Spectral Lines Edvidence for Spectra Absorption Spectra Bohr's Atom Vibrating Charges rev. Adv B ...
PH469 Fall 2002
... 7) In a Stern-Gerlach type of experiment, the magnetic field varies with distance in the z direction according to dBz/dz = 1.4 T/m. The silver atoms travel a distance x = 3.5 cm. The most probable speed of the atoms emerging from the oven is v = 750 m/s. Find the separation of the two beams as they ...
... 7) In a Stern-Gerlach type of experiment, the magnetic field varies with distance in the z direction according to dBz/dz = 1.4 T/m. The silver atoms travel a distance x = 3.5 cm. The most probable speed of the atoms emerging from the oven is v = 750 m/s. Find the separation of the two beams as they ...
Bohr Model of the Atom
... Bohr Model of the Atom Fundamental postulates: The Danish physicist Niels Bohr, who first presented this model of the atom, based it on 3 fundamental postulates. (1) Electrons move around the nucleus in circular non-radiating orbits - called “stationary states”. However, they are not at rest! (2) An ...
... Bohr Model of the Atom Fundamental postulates: The Danish physicist Niels Bohr, who first presented this model of the atom, based it on 3 fundamental postulates. (1) Electrons move around the nucleus in circular non-radiating orbits - called “stationary states”. However, they are not at rest! (2) An ...
Atoms, electrons, nuclei J.J. Thomson discovered the electron (1897
... atoms radiate if an electron 'jumps' from one such orbit to another (Em > Ek) hf = Em – Ek where Em and Ek are the two energy levels for the given orbits, and h denotes the Planck constant For H atom: ...
... atoms radiate if an electron 'jumps' from one such orbit to another (Em > Ek) hf = Em – Ek where Em and Ek are the two energy levels for the given orbits, and h denotes the Planck constant For H atom: ...
No Slide Title
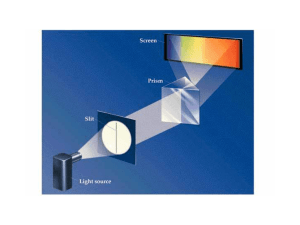
... The classical laws of physics do not explain the distribution of light emitted from hot objects. Max Planck solved the problem mathematically (in 1900) by assuming that the light can only be released in “chunks” of a discrete size (quantized like currency or the notes on a piano). l = wavelength (m) ...
... The classical laws of physics do not explain the distribution of light emitted from hot objects. Max Planck solved the problem mathematically (in 1900) by assuming that the light can only be released in “chunks” of a discrete size (quantized like currency or the notes on a piano). l = wavelength (m) ...
CHAPTER 4 TEST REVIEW GUIDE
... 15. Calculate the frequency and wavelength and energy of electromagnetic radiation and ...
... 15. Calculate the frequency and wavelength and energy of electromagnetic radiation and ...
Frank-Herze experiment with Neon
... elegantly supported Niels Bohr's model of the atom, with electrons orbiting the nucleus with specific, discrete energies. Franck and Hertz were awarded the Nobel Prize in Physics in 1925 for this work. In the early 20th century, experiments by Ernest Rutherford established that atoms consisted of a ...
... elegantly supported Niels Bohr's model of the atom, with electrons orbiting the nucleus with specific, discrete energies. Franck and Hertz were awarded the Nobel Prize in Physics in 1925 for this work. In the early 20th century, experiments by Ernest Rutherford established that atoms consisted of a ...
CHAPTER 4: Structure of the Atom
... In the present first part of the paper the mechanism of the binding of electrons by a positive nucleus is discussed in relation to Planck’s theory. It will be shown that it is possible from the point of view taken to account in a simple way for the law of the line spectrum of hydrogen. - Niels Bohr, ...
... In the present first part of the paper the mechanism of the binding of electrons by a positive nucleus is discussed in relation to Planck’s theory. It will be shown that it is possible from the point of view taken to account in a simple way for the law of the line spectrum of hydrogen. - Niels Bohr, ...
CHAPTER 4: Structure of the Atom
... In the present first part of the paper the mechanism of the binding of electrons by a positive nucleus is discussed in relation to Planck’s theory. It will be shown that it is possible from the point of view taken to account in a simple way for the law of the line spectrum of hydrogen. - Niels Bohr, ...
... In the present first part of the paper the mechanism of the binding of electrons by a positive nucleus is discussed in relation to Planck’s theory. It will be shown that it is possible from the point of view taken to account in a simple way for the law of the line spectrum of hydrogen. - Niels Bohr, ...
Atomic Structure
... Workshop Tutorials for Physics QR5: Atomic Structure A. Qualitative Questions: 1. The Bohr model of the atom was the first quantum mechanical model of the atom. a. Bohr postulated that a hydrogen atom could only exist without radiating in one of a set of stationary states. Explain what is meant by t ...
... Workshop Tutorials for Physics QR5: Atomic Structure A. Qualitative Questions: 1. The Bohr model of the atom was the first quantum mechanical model of the atom. a. Bohr postulated that a hydrogen atom could only exist without radiating in one of a set of stationary states. Explain what is meant by t ...
DARLLENWCH Y DARN ISOD AC ATEBWCH Y CWESTIYNAU SY
... in orbits of fixed size and energy. The energy of an electron depends on the size of the orbit and is lower for smaller orbits. Radiation can occur only when the electron jumps from one orbit to another. The atom will be completely stable in the state with the smallest orbit, since there is no orbit ...
... in orbits of fixed size and energy. The energy of an electron depends on the size of the orbit and is lower for smaller orbits. Radiation can occur only when the electron jumps from one orbit to another. The atom will be completely stable in the state with the smallest orbit, since there is no orbit ...
Corso di Fisica Moderna
... possible for an electron to move in an orbit for which its orbital angular momentum L is and integral mulAple of h, Planck’s constant. 3) Despite the fact that it is constantly acceleraAng, an ...
... possible for an electron to move in an orbit for which its orbital angular momentum L is and integral mulAple of h, Planck’s constant. 3) Despite the fact that it is constantly acceleraAng, an ...
Lesson 2 - The Bohr and Quantum Mechanical Model of the Atom
... Schrodinger: Energy of an electron can be represented by a wave function 1. Electrons can exist in different orbitals by absorbing or emitting quanta of energy ...
... Schrodinger: Energy of an electron can be represented by a wave function 1. Electrons can exist in different orbitals by absorbing or emitting quanta of energy ...
Chapter 2: Data Analysis
... The light coming out of the excited atomic entities is very specific to ...
... The light coming out of the excited atomic entities is very specific to ...
4. Structure of the Atom
... Never express yourself more clearly than you are able to think. Prediction is very difficult, especially about the future. - Niels Bohr ...
... Never express yourself more clearly than you are able to think. Prediction is very difficult, especially about the future. - Niels Bohr ...
The Franck-Hertz Experiment with Neon tube
... one year after Bohr published his theory of the hydrogen atom with its concept of ...
... one year after Bohr published his theory of the hydrogen atom with its concept of ...
Chapter 5 Electrons In Atoms 5.1 Models of the Atom The
... The model for the atom consisted of protons and neutrons making up a nucleus surrounded by electrons. The Bohr Model Bohr proposed that an ______________ is found only in specific circular paths, or orbits, around the nucleus. The fixed energies an electron can have are called __________________ lev ...
... The model for the atom consisted of protons and neutrons making up a nucleus surrounded by electrons. The Bohr Model Bohr proposed that an ______________ is found only in specific circular paths, or orbits, around the nucleus. The fixed energies an electron can have are called __________________ lev ...
energy levels
... Each atom has a set of energy levels. The line spectrum of an element results from the emission or absorption of photons when the atom makes a transition between different energy levels. ...
... Each atom has a set of energy levels. The line spectrum of an element results from the emission or absorption of photons when the atom makes a transition between different energy levels. ...
In 1913 Bohr proposed his quantized shell model of the atom to
... move in orbits of fixed size and energy. The energy of an electron depends on the size of the orbit and is lower for smaller orbits. Radiation can occur only when the electron jumps from one orbit to another. The atom will be completely stable in the state with the smallest orbit, since there is no ...
... move in orbits of fixed size and energy. The energy of an electron depends on the size of the orbit and is lower for smaller orbits. Radiation can occur only when the electron jumps from one orbit to another. The atom will be completely stable in the state with the smallest orbit, since there is no ...
James Franck

James Franck (26 August 1882 – 21 May 1964) was a German physicist who won the 1925 Nobel Prize for Physics with Gustav Hertz ""for their discovery of the laws governing the impact of an electron upon an atom"". He completed his doctorate in 1906 and his habilitation in 1911 at the Frederick William University in Berlin, where he lectured and taught until 1918, having reached the position of professor extraordinarius. He served as a volunteer in the German Army during World War I. He was seriously injured in 1917 in a gas attack and was awarded the Iron Cross 1st Class.Franck became the Head of the Physics Division of the Kaiser Wilhelm Gesellschaft for Physical Chemistry. In 1920, Franck became professor ordinarius of experimental physics and Director of the Second Institute for Experimental Physics at the University of Göttingen. While there he worked on quantum physics with Max Born, who was Director of the Institute of Theoretical Physics. His work included the Franck–Hertz experiment, an important confirmation of the Bohr model of the atom. He promoted the careers of women in physics, notably Lise Meitner, Hertha Sponer and Hilde Levi.After the NSDAP came to power in Germany in 1933, Franck resigned his post in protest against the dismissal of fellow academics. He assisted Frederick Lindemann in helping dismissed Jewish scientists find work overseas, before he left Germany in November 1933. After a year at the Niels Bohr Institute in Denmark, he moved to the United States, where he worked at Johns Hopkins University in Baltimore and then the University of Chicago. During this period he became interested in photosynthesis.Franck participated in the Manhattan Project during World War II as Director of the Chemistry Division of the Metallurgical Laboratory. He was also the chairman of the Committee on Political and Social Problems regarding the atomic bomb, which is best known for the compilation of the Franck Report, which recommended that the atomic bombs not be used on the Japanese cities without warning.