* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Chapter 5 Sec. 2 Bohr`s Model and the Quantum Mechanical Model
Molecular Hamiltonian wikipedia , lookup
Wave function wikipedia , lookup
History of quantum field theory wikipedia , lookup
Schrödinger equation wikipedia , lookup
Renormalization group wikipedia , lookup
Double-slit experiment wikipedia , lookup
Hidden variable theory wikipedia , lookup
James Franck wikipedia , lookup
Relativistic quantum mechanics wikipedia , lookup
Quantum electrodynamics wikipedia , lookup
X-ray photoelectron spectroscopy wikipedia , lookup
Particle in a box wikipedia , lookup
Rutherford backscattering spectrometry wikipedia , lookup
Copenhagen interpretation wikipedia , lookup
X-ray fluorescence wikipedia , lookup
Electron scattering wikipedia , lookup
Atomic orbital wikipedia , lookup
Matter wave wikipedia , lookup
Bohr–Einstein debates wikipedia , lookup
Electron configuration wikipedia , lookup
Tight binding wikipedia , lookup
Theoretical and experimental justification for the Schrödinger equation wikipedia , lookup
Wave–particle duality wikipedia , lookup
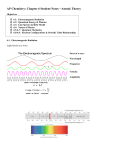
Chapter 5 Sec. 2 Bohr’s Model and the Quantum Mechanical Model NOTES Bohr’s Model of the Atom o Remember that the atomic emission spectra for hydrogen is discontinuous because it is made up of certain frequencies of light. Scientists wanted to know WHY this was. In 1913 the Danish physicist named Niels Bohr answered this question and correctly predicted the frequencies of the lines in hydrogen’s atomic emission spectra. o Energy States of Hydrogen Bohr proposed the following: The lowest allowable energy level of an atom is called its ___________________ _______________________. When an atom gains energy it is said to be in an __________________________ _______________________. Bohr assigned a number, ______________, called a quantum number, to each orbit. The first orbit, the one closest to the nucleus, has a quantum number of n=1. o The Hydrogen Line Spectrum When energy is added, the electron in an atom rises to an excited state. Equation used to calculate the change in energy between 2 energy levels: Equation used to calculate the energy of a quantum, the energy of a photon, or the energy difference between 2 levels: E= h = Planck’s constant = c = speed of light = λ= Ex #1) A green light of wavelength 489 nm is observed in the emission spectrum of hydrogen. Calculate the energy difference between the two energy states that are responsible for this line. Ex #2) The energy of one photon is 5.67 x 10-19 J. What is the length of the wavelength observed? Since only certain atomic energies are possible, only certain frequencies of electromagnetic radiation can be emitted. Keep in mind that the energy levels are not evenly spaced. o Drawings of Bohr’s Model of the Atom: IMPORTANT: Bohr’s model was good, but it only explained the atomic emission spectra of the hydrogen atom. The Quantum Mechanical Model of the Atom o Quantum Mechanical Model of the Atom: o In 1924, a French physics student named Louis de Broglie explained the fixed energy levels of Bohr’s model. He explained that electrons can act like _____________________________. He also showed that electrons on circular orbits can only have _____________________ numbers of wavelengths. o de Broglie predicted that all moving particles have wave characteristics. de Broglie knew that if an electron acted like a wave and was restricted to certain circular orbits that only certain wavelengths, frequencies, and energies are possible. de Broglie Equation/ Particle Electromagnetic-Wave Relationship: λ= h = Planck’s constant = m= v= o The Heisenberg Uncertainty Principle German physicist, Werner Heisenberg, showed that it is impossible to measure any object without disturbing that object. The Heisenberg Uncertainty Principles states: From the Heisenberg Uncertainty Principle we also understand that we can only know the probability for an electron to occupy a certain region around the nucleus. o The Schrodinger Wave Equation In 1926 the Austrian physicist Erwin Schrodinger came up with an equation that showed hydrogen atom’s electron acting like a wave. Each solution to the Schrodinger Wave Equation is called a wave function. The wave function predicts the 3D region around the nucleus, called an atomic orbital, where the electron is most likely to be found. o An electron is most likely to be found: The Bohr Model vs. The Quantum Mechanical Model of the Atom BOHR MODEL Scientist primarily responsible for it What electrons are considered to be Allowed energy states/values Electron pathways QUANTUM MECHANICAL MODEL