Background Information
... The Lucas reagent is an aqueous solution of strong acid (HCl) and zinc chloride. An insoluble layer, cloudiness, color change (red or orange) or an emulsion will form with 1°, 2°, 3° allylic, 3° alkyl and some 2 ° alcohols and constitutes a “positive” result. Students should compare his/her results ...
... The Lucas reagent is an aqueous solution of strong acid (HCl) and zinc chloride. An insoluble layer, cloudiness, color change (red or orange) or an emulsion will form with 1°, 2°, 3° allylic, 3° alkyl and some 2 ° alcohols and constitutes a “positive” result. Students should compare his/her results ...
20.4 Acid-Base Properties of Carboxylic Acids
... wish to separate. How might you take advantage of the acidity of one component in the mixture to accomplish the ...
... wish to separate. How might you take advantage of the acidity of one component in the mixture to accomplish the ...
Applications of Phosphorus, Sulfur, Silicon and Boron Chemistry:
... Predict the stereochemistry of the product(s) arising from reactions covered (see LO6, 7 and 8) using reaction mechanisms to explain the stereochemical outcome of the transformations. 10. Show how silyl ethers can be used as hydroxyl protecting groups in organic chemistry. These notes, self-study wo ...
... Predict the stereochemistry of the product(s) arising from reactions covered (see LO6, 7 and 8) using reaction mechanisms to explain the stereochemical outcome of the transformations. 10. Show how silyl ethers can be used as hydroxyl protecting groups in organic chemistry. These notes, self-study wo ...
aldehydesketonescarb..
... contain dipole-dipole forces and are able to hydrogen bond. • How does the carboxyl group affect the physical properties of these compounds? • They are weak acids that will ionize slightly in water. • The H on the –OH is the acidic hydrogen and leaves when the acid is ionized. ...
... contain dipole-dipole forces and are able to hydrogen bond. • How does the carboxyl group affect the physical properties of these compounds? • They are weak acids that will ionize slightly in water. • The H on the –OH is the acidic hydrogen and leaves when the acid is ionized. ...
Practice Exam 4 - BioChemWeb.net
... f. The acidity of benzoic acid (C6H5CO2H) is most affected by electron-withdrawing substituents in the: ortho position ...
... f. The acidity of benzoic acid (C6H5CO2H) is most affected by electron-withdrawing substituents in the: ortho position ...
nucleophilic addition
... mixed with another aldehyde that doesn’t have any alphahydrogens and conc. NaOH, all of the formaldehyde is oxidized and all of the other aldehyde is reduced. ...
... mixed with another aldehyde that doesn’t have any alphahydrogens and conc. NaOH, all of the formaldehyde is oxidized and all of the other aldehyde is reduced. ...
Substitution reactions of carbonyl compounds at the α
... – HO- and RO- bases are more useful for forming enolates from 1,3-dicarbonyl compounds or in reactions that involve both the enolate and carbonyl starting material. The enolate ion has two nonequivalent resonance forms (compare this with both the allyl anion and the carboxylate anion). The form wher ...
... – HO- and RO- bases are more useful for forming enolates from 1,3-dicarbonyl compounds or in reactions that involve both the enolate and carbonyl starting material. The enolate ion has two nonequivalent resonance forms (compare this with both the allyl anion and the carboxylate anion). The form wher ...
Chapter 22/23-Organic Chemistry
... reactions(if there are more then one possible set of products name at least two): a. Pentane is cracked b. Heptane is cracked c. Propane and butane undergo a reforming reaction d. Benzene burns in air ...
... reactions(if there are more then one possible set of products name at least two): a. Pentane is cracked b. Heptane is cracked c. Propane and butane undergo a reforming reaction d. Benzene burns in air ...
Exam 2 SOLUTION
... a) How many steps are in the mechanism to this reaction? There are 4 steps to this mechanism. b) Which step is the rate-determining step? The second step, with the highest activation barrier, is the RDS. c) Label ∆Hrxn and the activation energies on the graph. 4. Using curved-arrow notation, give th ...
... a) How many steps are in the mechanism to this reaction? There are 4 steps to this mechanism. b) Which step is the rate-determining step? The second step, with the highest activation barrier, is the RDS. c) Label ∆Hrxn and the activation energies on the graph. 4. Using curved-arrow notation, give th ...
Aldehydes and Ketones
... be a site that is attacked by nucleophiles. And, since the oxygen bears a partial negative charge, it is likely to be a site of electrophilic attack. Since ordinary carbanions (R: −) and hydride ions (H: −) are very poor leaving groups (unlike ...
... be a site that is attacked by nucleophiles. And, since the oxygen bears a partial negative charge, it is likely to be a site of electrophilic attack. Since ordinary carbanions (R: −) and hydride ions (H: −) are very poor leaving groups (unlike ...
Chapter 7
... • The slow step, RDS, is the second step, the formation of the carbocation • This explains the order of reactivity with the tertiary alcohol reacting easiest, due to the tertiary carbocation being the most stable. ...
... • The slow step, RDS, is the second step, the formation of the carbocation • This explains the order of reactivity with the tertiary alcohol reacting easiest, due to the tertiary carbocation being the most stable. ...
Organic Chemistry - Unit 2
... halocarbon – this is a hydrocarbon containing a halogen substituent. alkyl halide – this is an aliphatic hydrocarbon containing halogen substituents. aryl halide – this is a halocarbon in which there is a halogen substituent on an arene ring. (Substituted benzene) In all of these compounds, the halo ...
... halocarbon – this is a hydrocarbon containing a halogen substituent. alkyl halide – this is an aliphatic hydrocarbon containing halogen substituents. aryl halide – this is a halocarbon in which there is a halogen substituent on an arene ring. (Substituted benzene) In all of these compounds, the halo ...
Chem 3.5 Answers #7
... Aldehydes are produced by the oxidation of primary alcohols using acidified potassium dichromate solution. The aldehyde must be distilled off as it is made or it will oxidise further, up to the carboxylic acid. Ketones are made by the same oxidation reaction with secondary alcohols, but they do not ...
... Aldehydes are produced by the oxidation of primary alcohols using acidified potassium dichromate solution. The aldehyde must be distilled off as it is made or it will oxidise further, up to the carboxylic acid. Ketones are made by the same oxidation reaction with secondary alcohols, but they do not ...
Review sheet - Paws.wcu.edu.
... Cl2 or Br2 plus uv light – free radical addition to form a haloalkane SOCl2 and PBr3 (converts R-OH to R-X for 1° and 2° alcohols) HCl / HBr (R-OH to R-X for 3° alcohols) NBS (for allylic bromination) Alkene plus X2 or HX : addition to double bond (Markovnikov selectivity) Preparation of Alcohols Hy ...
... Cl2 or Br2 plus uv light – free radical addition to form a haloalkane SOCl2 and PBr3 (converts R-OH to R-X for 1° and 2° alcohols) HCl / HBr (R-OH to R-X for 3° alcohols) NBS (for allylic bromination) Alkene plus X2 or HX : addition to double bond (Markovnikov selectivity) Preparation of Alcohols Hy ...
Objective Reaction Type Structural Feature How to figure out how reactants react?
... chemical is an alkylating agent that reacts readily at nucleophilic sites, and sulfhydryl-containing enzymes such as those found in the eye are a prime target, the Army says. Effects of CS exposure include an extreme burning sensation in the eyes with a copious flow of tears, coughing, sneezing, a p ...
... chemical is an alkylating agent that reacts readily at nucleophilic sites, and sulfhydryl-containing enzymes such as those found in the eye are a prime target, the Army says. Effects of CS exposure include an extreme burning sensation in the eyes with a copious flow of tears, coughing, sneezing, a p ...
Nucleophilic Addition to Carbonyl Groups
... The reverse of acetal formation if acetal hydrolysis. This is achieved by excess water in the presence of an acid catalyst. ...
... The reverse of acetal formation if acetal hydrolysis. This is achieved by excess water in the presence of an acid catalyst. ...
Carbonyl Alpha-Substitution Reactions
... Constraints on Enolate Alkylation • SN2 reaction: the leaving group X can be chloride, bromide, iodide, or tosylate • R should be primary or methyl and preferably should be allylic or benzylic • Secondary halides react poorly, and tertiary halides don't react at all because of competing elimination ...
... Constraints on Enolate Alkylation • SN2 reaction: the leaving group X can be chloride, bromide, iodide, or tosylate • R should be primary or methyl and preferably should be allylic or benzylic • Secondary halides react poorly, and tertiary halides don't react at all because of competing elimination ...
Part (d) The Birch Reduction of Nitrogen
... As the constituents of poly amides (ie peptides) these functional groups are essential parts of biological systems. We can hydrolyse an amide bond in the laboratory, but require harsh acidic or basic conditions to do it ...
... As the constituents of poly amides (ie peptides) these functional groups are essential parts of biological systems. We can hydrolyse an amide bond in the laboratory, but require harsh acidic or basic conditions to do it ...
CHE-06 year 2004
... Outline ring bromination and sulphonation reactions of the following compounds. In each (7) case give the structure of the major reaction product(s). Also indicate whether the reaction would occur faster or slower than the corresponding reaction of benzene. ...
... Outline ring bromination and sulphonation reactions of the following compounds. In each (7) case give the structure of the major reaction product(s). Also indicate whether the reaction would occur faster or slower than the corresponding reaction of benzene. ...
Taylor`s Organic Reactions Summary Sheet
... Hydrolysis Reaction: A reaction in which a bond is broken by the addition of the components of water, with the formation of two or more products. ...
... Hydrolysis Reaction: A reaction in which a bond is broken by the addition of the components of water, with the formation of two or more products. ...
U. of Kentucky Chemistry 535 Synthetic Organic Chemistry Spring
... retrosynthetic analysis that leaves no doubt for the reader that you can make the molecule. You may start with molecules containing no less than eight carbon atoms. ...
... retrosynthetic analysis that leaves no doubt for the reader that you can make the molecule. You may start with molecules containing no less than eight carbon atoms. ...
Slide 1
... Given samples of various alcohols and carboxylic acids Establish some of their physical and chemical properties & perform various reactions HO HO ethanol ...
... Given samples of various alcohols and carboxylic acids Establish some of their physical and chemical properties & perform various reactions HO HO ethanol ...
The SN2 Reaction: 1
... Introduction In this experiment 1-butanol is converted to 1-bromobutane by an SN2 reaction. In general, a primary alkyl bromide can be prepared by heating the corresponding alcohol with (1) constantboiling hydrobromic acid (47% HBr), (2) an aqueous solution of sodium bromide and excess sulfuric acid ...
... Introduction In this experiment 1-butanol is converted to 1-bromobutane by an SN2 reaction. In general, a primary alkyl bromide can be prepared by heating the corresponding alcohol with (1) constantboiling hydrobromic acid (47% HBr), (2) an aqueous solution of sodium bromide and excess sulfuric acid ...
reactions of organic compounds
... • Determines which isomer will be most prominent (only a small amount of other isomer will be produced). • MARKOVNIKOV’s RULE: the halogen atom or OH group in an addition reaction is usually added to the carbon bonded to the most carbon atoms. ...
... • Determines which isomer will be most prominent (only a small amount of other isomer will be produced). • MARKOVNIKOV’s RULE: the halogen atom or OH group in an addition reaction is usually added to the carbon bonded to the most carbon atoms. ...
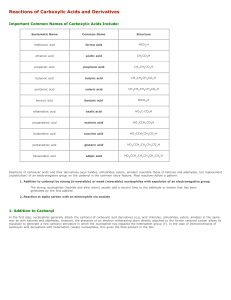
Reactions of Carboxylic Acids and Derivatives
... In the first step, nucleophiles generally attack the carbonyl of carboxylic acid derivatives (e.g. acid chlorides, anhydrides, esters, amides) in the same way as with ketones and aldehydes. However, the presence of an electron withdrawing atom directly attached to the former carbonyl carbon allows i ...
... In the first step, nucleophiles generally attack the carbonyl of carboxylic acid derivatives (e.g. acid chlorides, anhydrides, esters, amides) in the same way as with ketones and aldehydes. However, the presence of an electron withdrawing atom directly attached to the former carbonyl carbon allows i ...
Wolff rearrangement

The Wolff rearrangement is a reaction in organic chemistry in which an α-diazocarbonyl compound is converted into a ketene by loss of dinitrogen with accompanying 1,2-rearrangement. The Wolff rearrangement yields a ketene as an intermediate product, which can undergo nucleophilic attack with weakly acidic nucleophiles such as water, alcohols, and amines, to generate carboxylic acid derivatives or undergo [2+2] cycloaddition reactions to form four-membered rings. The mechanism of the Wolff rearrangement has been the subject of debate since its first use. No single mechanism sufficiently describes the reaction, and there are often competing concerted and carbene-mediated pathways; for simplicity, only the textbook, concerted mechanism is shown below. The reaction was discovered by Ludwig Wolff in 1902. The Wolff rearrangement has great synthetic utility due to the accessibility of α-diazocarbonyl compounds, variety of reactions from the ketene intermediate, and stereochemical retention of the migrating group. However, the Wolff rearrangement has limitations due to the highly reactive nature of α-diazocarbonyl compounds, which can undergo a variety of competing reactions.The Wolff rearrangement can be induced via thermolysis, photolysis, or transition metal catalysis. In this last case, the reaction is sensitive to the transition metal; silver (I) oxide or other Ag(I) catalysts work well and are generally used. The Wolff rearrangement has been used in many total syntheses; the most common use is trapping the ketene intermediate with nucleophiles to form carboxylic acid derivatives. The Arndt-Eistert homologation is a specific example of this use, wherein a carboxylic acid may be elongated by a methylene unit. Another common use is in ring-contraction methods; if the α-diazo ketone is cyclic, the Wolff rearrangement results in a ring-contracted product. The Wolff rearrangement works well in generating ring-strained systems, where other reactions may fail.