www.xtremepapers.net
... effective if electronegative elements such as chlorine are present. Thus the chloroethanoic acids become increasingly more acidic as more chlorine atoms are present in the molecule. The reagent of preference is SOCl2, since both by-products are gases. Other possibilities are PCl3 and PCl5. The react ...
... effective if electronegative elements such as chlorine are present. Thus the chloroethanoic acids become increasingly more acidic as more chlorine atoms are present in the molecule. The reagent of preference is SOCl2, since both by-products are gases. Other possibilities are PCl3 and PCl5. The react ...
www.xtremepapers.net
... effective if electronegative elements such as chlorine are present. Thus the chloroethanoic acids become increasingly more acidic as more chlorine atoms are present in the molecule. The reagent of preference is SOCl2, since both by-products are gases. Other possibilities are PCl3 and PCl5. The react ...
... effective if electronegative elements such as chlorine are present. Thus the chloroethanoic acids become increasingly more acidic as more chlorine atoms are present in the molecule. The reagent of preference is SOCl2, since both by-products are gases. Other possibilities are PCl3 and PCl5. The react ...
32 GRIGNARD REACTION Alkyl halides can react with magnesium
... reagent reacts with water to give hydrocarbons (benzene in this case). Mount a 50 mL round bottom flask with a condenser. Do not run water through your condenser. Place 0.7 g Mg turnings in the flask. Add 10 mL of dry ether, followed by 0.5 mL of bromobenzene. Your TA will then come around and add a ...
... reagent reacts with water to give hydrocarbons (benzene in this case). Mount a 50 mL round bottom flask with a condenser. Do not run water through your condenser. Place 0.7 g Mg turnings in the flask. Add 10 mL of dry ether, followed by 0.5 mL of bromobenzene. Your TA will then come around and add a ...
Study Guide for Exam 2-‐ Aldehydes and Ketones
... forming an oxaphosphetane intermediate. Thus, the electrophile is not H+ as in the previous examples but the phosphonium center. The intermediate undergoes a reverse 2+2 process to ...
... forming an oxaphosphetane intermediate. Thus, the electrophile is not H+ as in the previous examples but the phosphonium center. The intermediate undergoes a reverse 2+2 process to ...
Final Exam Review
... Hydrolysis of acid chlorides, anhydrides, esters, amides, and nitriles to carboxylic acids Preparation and reaction of carboxylic acids and their derivatives Amines Synthesis of amines: amide + LiAlH4 nitrile hydrolysis ammonia + R-X (SN2) Amino Acids and Proteins Amino acids, peptide bonds, and pro ...
... Hydrolysis of acid chlorides, anhydrides, esters, amides, and nitriles to carboxylic acids Preparation and reaction of carboxylic acids and their derivatives Amines Synthesis of amines: amide + LiAlH4 nitrile hydrolysis ammonia + R-X (SN2) Amino Acids and Proteins Amino acids, peptide bonds, and pro ...
Carboxylic acid
... exist primarily of dimers (two molecules held together by H-bonding) • Because of the above properties, carboxylic acids have high boiling points (higher than corresponding alcohols) • Those with less than 5 carbons are soluble in water - those with more than 5 C’s can be soluble when ionized O C ...
... exist primarily of dimers (two molecules held together by H-bonding) • Because of the above properties, carboxylic acids have high boiling points (higher than corresponding alcohols) • Those with less than 5 carbons are soluble in water - those with more than 5 C’s can be soluble when ionized O C ...
Alcohols - Miller, Jonathan
... (often larger molecular masses) formed when an alcohol reacts with a carboxylic acid in the presence of an acid catalyst (small amount of conc. H2SO4). It is an equilibrium reaction. ...
... (often larger molecular masses) formed when an alcohol reacts with a carboxylic acid in the presence of an acid catalyst (small amount of conc. H2SO4). It is an equilibrium reaction. ...
Ester Lab / Adobe Acrobat Document
... amber dichromate ion to the green chromium(III) can be quantitatively interpreted to determine blood alcohol (ethanol) levels. Second generation breathalyzer tests measure the amount of water produced in the reaction, for a more accurate measure. Secondary alcohols (OH attached to a non-terminal car ...
... amber dichromate ion to the green chromium(III) can be quantitatively interpreted to determine blood alcohol (ethanol) levels. Second generation breathalyzer tests measure the amount of water produced in the reaction, for a more accurate measure. Secondary alcohols (OH attached to a non-terminal car ...
aldehyde ketone
... Due to the polarity of the C=O bond, a permanent dipole moment exists in aldehydes and ketones (dipole-dipole forces). Thus, the MP/BP of aldehydes and ketones is mid-range – higher than that of alkanes or alkenes (London forces) but lower than that of alcohols (hydrogen bonds). Nomenclature – IUPAC ...
... Due to the polarity of the C=O bond, a permanent dipole moment exists in aldehydes and ketones (dipole-dipole forces). Thus, the MP/BP of aldehydes and ketones is mid-range – higher than that of alkanes or alkenes (London forces) but lower than that of alcohols (hydrogen bonds). Nomenclature – IUPAC ...
Part B: Short Written Response - bourre-chem-11
... _______36. Which statements are true about the oil refining process? a) Fractions that have higher boiling points settle at the bottom, and fractions with lower boiling points settle to the bottom. b) Fractions that have lower boiling points settle at the bottom, and fractions with higher boiling po ...
... _______36. Which statements are true about the oil refining process? a) Fractions that have higher boiling points settle at the bottom, and fractions with lower boiling points settle to the bottom. b) Fractions that have lower boiling points settle at the bottom, and fractions with higher boiling po ...
BSc-Chemistry-II
... Mannich reaction. Use of acetals as protecting group. Oxidation of aldehydes, BaeyerVilliger oxidation of ketones, Cannizzaro reaction. MPV, Clemmensen, WolffKishner, LiAIH4 and NaBH4. UNIT IV Carboxylic Acid ...
... Mannich reaction. Use of acetals as protecting group. Oxidation of aldehydes, BaeyerVilliger oxidation of ketones, Cannizzaro reaction. MPV, Clemmensen, WolffKishner, LiAIH4 and NaBH4. UNIT IV Carboxylic Acid ...
Carbon-Carbon Bond Forming Reactions
... - both symmetrical & mixed Claisen condensation reactions are known - excellent method for preparation of beta ketoesters - intramolecular Claisen known as a Dieckman condensation ...
... - both symmetrical & mixed Claisen condensation reactions are known - excellent method for preparation of beta ketoesters - intramolecular Claisen known as a Dieckman condensation ...
Application of IBX Method for the Synthesis of Ketones from
... One of the most common reactions in chemistry is the synthesis of ketones from acids1−4 . First, the acid is changed into its chloride. Then the acid chloride reacts with an organometallic reagent or gives a FriedelCrafts type reaction in the presence of Lewis acids. These methods are very useful fo ...
... One of the most common reactions in chemistry is the synthesis of ketones from acids1−4 . First, the acid is changed into its chloride. Then the acid chloride reacts with an organometallic reagent or gives a FriedelCrafts type reaction in the presence of Lewis acids. These methods are very useful fo ...
Erythro and Threo
... Conversion of all -OH groups to -OR, • Modified Williamson synthesis • After converting sugar to acetal, stable in base. • Helps to purify by recrystallization from water. ...
... Conversion of all -OH groups to -OR, • Modified Williamson synthesis • After converting sugar to acetal, stable in base. • Helps to purify by recrystallization from water. ...
Carbonyl Compounds
... Of the three compound listed, ethanamide has the highest boiling point because it has the potential t form multiple H-bonds between molecules, all the bonds must be broken before the compound can pass to vapour phase. ethanoic acid, however, has fewer H-bonds compared with ethanamide. Propanone has ...
... Of the three compound listed, ethanamide has the highest boiling point because it has the potential t form multiple H-bonds between molecules, all the bonds must be broken before the compound can pass to vapour phase. ethanoic acid, however, has fewer H-bonds compared with ethanamide. Propanone has ...
SCH4U Unit Test Name
... 2. When two alcohols undergo a self condensation, what is formed? a. liquid alcohol d. an aldehyde b. a ketone e. an ether c. an ester ...
... 2. When two alcohols undergo a self condensation, what is formed? a. liquid alcohol d. an aldehyde b. a ketone e. an ether c. an ester ...
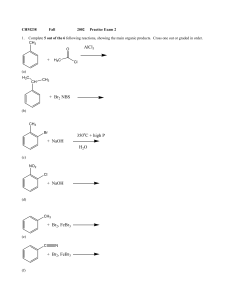
Exam 2
... worth studying. Also problems assigned for the text may also be helpful. Chap 9 &10-- Sn2, Sn1, E2 and E1 reactions. -Know the definitions of Sn2, Sn1, E2 and E1. -Be able to depict the reaction coordinate diagrams of each reaction -Be able to draw the mechanism, products and the stereochemical resu ...
... worth studying. Also problems assigned for the text may also be helpful. Chap 9 &10-- Sn2, Sn1, E2 and E1 reactions. -Know the definitions of Sn2, Sn1, E2 and E1. -Be able to depict the reaction coordinate diagrams of each reaction -Be able to draw the mechanism, products and the stereochemical resu ...
answer
... • Threonine (Y) is an amino acid. On the structure of (Y) below, identify all stereocentres in threonine with an asterisk (*). ...
... • Threonine (Y) is an amino acid. On the structure of (Y) below, identify all stereocentres in threonine with an asterisk (*). ...
Chapter 7 Alkenes and Alkynes I
... Rearrangements of carbocations occur if a more stable carbocation can be obtained Example ...
... Rearrangements of carbocations occur if a more stable carbocation can be obtained Example ...
Wolff rearrangement

The Wolff rearrangement is a reaction in organic chemistry in which an α-diazocarbonyl compound is converted into a ketene by loss of dinitrogen with accompanying 1,2-rearrangement. The Wolff rearrangement yields a ketene as an intermediate product, which can undergo nucleophilic attack with weakly acidic nucleophiles such as water, alcohols, and amines, to generate carboxylic acid derivatives or undergo [2+2] cycloaddition reactions to form four-membered rings. The mechanism of the Wolff rearrangement has been the subject of debate since its first use. No single mechanism sufficiently describes the reaction, and there are often competing concerted and carbene-mediated pathways; for simplicity, only the textbook, concerted mechanism is shown below. The reaction was discovered by Ludwig Wolff in 1902. The Wolff rearrangement has great synthetic utility due to the accessibility of α-diazocarbonyl compounds, variety of reactions from the ketene intermediate, and stereochemical retention of the migrating group. However, the Wolff rearrangement has limitations due to the highly reactive nature of α-diazocarbonyl compounds, which can undergo a variety of competing reactions.The Wolff rearrangement can be induced via thermolysis, photolysis, or transition metal catalysis. In this last case, the reaction is sensitive to the transition metal; silver (I) oxide or other Ag(I) catalysts work well and are generally used. The Wolff rearrangement has been used in many total syntheses; the most common use is trapping the ketene intermediate with nucleophiles to form carboxylic acid derivatives. The Arndt-Eistert homologation is a specific example of this use, wherein a carboxylic acid may be elongated by a methylene unit. Another common use is in ring-contraction methods; if the α-diazo ketone is cyclic, the Wolff rearrangement results in a ring-contracted product. The Wolff rearrangement works well in generating ring-strained systems, where other reactions may fail.