4.4 Formation of Esters from Carboxylic Acids and Alcohols
... + H2O (One might wonder how the ester gets formed in the first place, given that it is uphill from the carboxylic acid and alcohol molecules. In fact in biological systems the carboxylic acids are not the reactive molecule itself. The carboxylic acid is activated (energy level raised) by attaching ...
... + H2O (One might wonder how the ester gets formed in the first place, given that it is uphill from the carboxylic acid and alcohol molecules. In fact in biological systems the carboxylic acids are not the reactive molecule itself. The carboxylic acid is activated (energy level raised) by attaching ...
CARBONYL COMPOUNDS
... 6. Estimate the type and the extent of interactive forces between C2H6 (MR = 30), CH3OH (MR = 30) and HCHO (MR = 30) and put these compounds in order with respect to increasing boiling point. C2H6 CH3OH HCHO Methanal is a gas, other important carbonyl compounds are …………………... Early members are solub ...
... 6. Estimate the type and the extent of interactive forces between C2H6 (MR = 30), CH3OH (MR = 30) and HCHO (MR = 30) and put these compounds in order with respect to increasing boiling point. C2H6 CH3OH HCHO Methanal is a gas, other important carbonyl compounds are …………………... Early members are solub ...
nomenclature continued… - Turner Fenton Secondary School
... Are like aldehydes except the carbonyl group (C=O) is located somewhere in the middle of the chain (not at terminal end). Generally written as RR`C=O. Ketones like aldehydes do not exhibit hydrogen bonding. There are common names for ketones, for example, acetone. Acetone is the key ingredient in na ...
... Are like aldehydes except the carbonyl group (C=O) is located somewhere in the middle of the chain (not at terminal end). Generally written as RR`C=O. Ketones like aldehydes do not exhibit hydrogen bonding. There are common names for ketones, for example, acetone. Acetone is the key ingredient in na ...
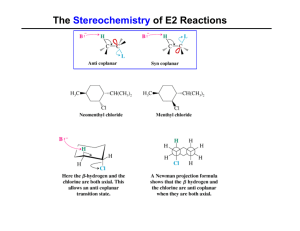
Regiochemistry of Eliminations
... 1) Show the mechanism for the formation of each of the three products expected in this experiment. Answer: ...
... 1) Show the mechanism for the formation of each of the three products expected in this experiment. Answer: ...
Going Bananas Over Isoamyl Acetate
... acid, as noted above. If either one is doubled, the theoretical yield increases to 85%. When one is tripled, it goes to 90%. But note that in the example cited the boiling point of the relatively nonpolar ester is only about 8° C higher than the boiling points of the polar acetic acid and 1-butanol, ...
... acid, as noted above. If either one is doubled, the theoretical yield increases to 85%. When one is tripled, it goes to 90%. But note that in the example cited the boiling point of the relatively nonpolar ester is only about 8° C higher than the boiling points of the polar acetic acid and 1-butanol, ...
Going Bananas Over Isoamyl Acetate
... acid, as noted above. If either one is doubled, the theoretical yield increases to 85%. When one is tripled, it goes to 90%. But note that in the example cited the boiling point of the relatively nonpolar ester is only about 8° C higher than the boiling points of the polar acetic acid and 1-butanol, ...
... acid, as noted above. If either one is doubled, the theoretical yield increases to 85%. When one is tripled, it goes to 90%. But note that in the example cited the boiling point of the relatively nonpolar ester is only about 8° C higher than the boiling points of the polar acetic acid and 1-butanol, ...
Practice problems for week 8 PDF
... This problem set includes questions covering material from chapters 7, 8 and 9 in your textbook. ...
... This problem set includes questions covering material from chapters 7, 8 and 9 in your textbook. ...
Bulent Terem - CH324 - Syllabus | Chaminade
... about the rates and regioselectivity of substituted aromatic compounds in electrophilic substitution reactions. Understand the basic principles of infrared and nuclear magnetic resonance spectroscoscopy, as well as mass spectrometry as tools to determine structures of organic molecules. Learn to ...
... about the rates and regioselectivity of substituted aromatic compounds in electrophilic substitution reactions. Understand the basic principles of infrared and nuclear magnetic resonance spectroscoscopy, as well as mass spectrometry as tools to determine structures of organic molecules. Learn to ...
esters - wellswaysciences
... to form an ester and water is an equilibrium reaction the back reaction of this is the hydrolysis of an ester to reform the acid and the alcohol. • The reaction uses an aqueous (dilute) acid catalyst and is refluxed as before. • As before the reaction does not go to completion so all reactants and p ...
... to form an ester and water is an equilibrium reaction the back reaction of this is the hydrolysis of an ester to reform the acid and the alcohol. • The reaction uses an aqueous (dilute) acid catalyst and is refluxed as before. • As before the reaction does not go to completion so all reactants and p ...
Procedure Notes
... • Concentrated acids such as H2SO4 can cause burns. Wash immediately with water if spilled on the skin. • As always, be careful with the hot ...
... • Concentrated acids such as H2SO4 can cause burns. Wash immediately with water if spilled on the skin. • As always, be careful with the hot ...
Chapter 7
... • The slow step, RDS, is the second step, the formation of the carbocation • This explains the order of reactivity with the tertiary alcohol reacting easiest, due to the tertiary carbocation being the most stable. ...
... • The slow step, RDS, is the second step, the formation of the carbocation • This explains the order of reactivity with the tertiary alcohol reacting easiest, due to the tertiary carbocation being the most stable. ...
Answers
... forced back to the left? (Hint: What is the byproduct of the reaction?) This equilibrium is possible through the addition of acid to the electrophile, making it a better electrophile. This means it does not need as strong of a nucleophile to react. The reaction could be forced back to the left is wa ...
... forced back to the left? (Hint: What is the byproduct of the reaction?) This equilibrium is possible through the addition of acid to the electrophile, making it a better electrophile. This means it does not need as strong of a nucleophile to react. The reaction could be forced back to the left is wa ...
Chapter 7 - Alkenes and Alkynes I less substituted alkene due to
... mechanism - To favor E2, a secondary or tertiary alkyl halide should be used - If there is only a possibility for a primary alkyl halide, use a bulky base - Use a higher concentration of a strong and nonpolarizable base, like an alkoxide - EtONa/EtOH favors the more substituted double bond while t-E ...
... mechanism - To favor E2, a secondary or tertiary alkyl halide should be used - If there is only a possibility for a primary alkyl halide, use a bulky base - Use a higher concentration of a strong and nonpolarizable base, like an alkoxide - EtONa/EtOH favors the more substituted double bond while t-E ...
Relative Reactivity of Aldehydes and Ketones: Generally
... Formations of hydrates can occur under either acidic or basic conditions This reaction is a reversible reaction due to the nucleophile (either H2O or –OH) having an electronegative oxygen atom that can act as a “Leaving Group”. ...
... Formations of hydrates can occur under either acidic or basic conditions This reaction is a reversible reaction due to the nucleophile (either H2O or –OH) having an electronegative oxygen atom that can act as a “Leaving Group”. ...
10.5 Carbonyl Compounds (a) describe: (i) the
... Different carbonyl compounds (R, R’ differ in number of carbon/hydrogen atoms) have different melting points. This is a nucleophilic addition then elimination (of water) type of reaction to give the hydrazone. ...
... Different carbonyl compounds (R, R’ differ in number of carbon/hydrogen atoms) have different melting points. This is a nucleophilic addition then elimination (of water) type of reaction to give the hydrazone. ...
info
... i. NaBH4 will reduce an aldehyde, ketone, or acid chloride to the corresponding alcohol. It will not reduce an acid or an ester. ii. LiAlH4 will reduce an aldehyde, ketone, acid, or ester to the corresponding alcohol. iii. LiAlH(OtBu)3 will reduce an acid chloride to an aldehyde. iv. DIBAL will ...
... i. NaBH4 will reduce an aldehyde, ketone, or acid chloride to the corresponding alcohol. It will not reduce an acid or an ester. ii. LiAlH4 will reduce an aldehyde, ketone, acid, or ester to the corresponding alcohol. iii. LiAlH(OtBu)3 will reduce an acid chloride to an aldehyde. iv. DIBAL will ...
I (21 points) Complete the following reactions by providing starting
... A. (JOC, 2008, ASAP, Loh) Chemists have been studying the Barbier-Grignard reactions with the goal of affecting the carbon-carbon bond forming reaction in solvents like water. Recent developments include the use of indium metal catalysts that react through single electron transfer mechanisms. Show t ...
... A. (JOC, 2008, ASAP, Loh) Chemists have been studying the Barbier-Grignard reactions with the goal of affecting the carbon-carbon bond forming reaction in solvents like water. Recent developments include the use of indium metal catalysts that react through single electron transfer mechanisms. Show t ...
DEHYDRATION - ALKENE TEST EXERCISES
... DEHYDRATION - ALKENE TEST EXERCISES 1. Give a detailed mechanism for the acid-catalyzed dehydration of cyclohexanol to cyclohexene. ...
... DEHYDRATION - ALKENE TEST EXERCISES 1. Give a detailed mechanism for the acid-catalyzed dehydration of cyclohexanol to cyclohexene. ...
organic lab questions
... this reaction. Present your ranking in the form of simplified organic structures separated by greater than signs. Show the degree of each alcohol below each structure. ...
... this reaction. Present your ranking in the form of simplified organic structures separated by greater than signs. Show the degree of each alcohol below each structure. ...
Alcohols and Phenols
... with moisture or acids Spin–spin splitting is observed between protons on the oxygen-bearing carbon and other neighbors ...
... with moisture or acids Spin–spin splitting is observed between protons on the oxygen-bearing carbon and other neighbors ...
Alkenes from Alcohols
... Alkenes from Alcohols 2-Methyl-1-butene and 2-Methyl-2-butene INTRODUCTION The dilute sulfuric acid catalyzed dehydration of 2-methyl-2-butanol (t-amyl alcohol) proceeds readily to give a mixture of alkenes. The mechanism of this reaction involves the intermediate formation of the relatively stable ...
... Alkenes from Alcohols 2-Methyl-1-butene and 2-Methyl-2-butene INTRODUCTION The dilute sulfuric acid catalyzed dehydration of 2-methyl-2-butanol (t-amyl alcohol) proceeds readily to give a mixture of alkenes. The mechanism of this reaction involves the intermediate formation of the relatively stable ...
1 Carbonyl Condensation Reactions (Conjugate Addition) If we look
... We have seen that “hard” nucleophiles, such as present in RMgBr and RLi reagents, add to the carbonyl. A way to force only conjugate addition with an organometallic reagent is to form the copper salt - in this case, a dialkylcuprate. We’ve already seen these are used to prepare ketones from acid chl ...
... We have seen that “hard” nucleophiles, such as present in RMgBr and RLi reagents, add to the carbonyl. A way to force only conjugate addition with an organometallic reagent is to form the copper salt - in this case, a dialkylcuprate. We’ve already seen these are used to prepare ketones from acid chl ...
Wolff rearrangement

The Wolff rearrangement is a reaction in organic chemistry in which an α-diazocarbonyl compound is converted into a ketene by loss of dinitrogen with accompanying 1,2-rearrangement. The Wolff rearrangement yields a ketene as an intermediate product, which can undergo nucleophilic attack with weakly acidic nucleophiles such as water, alcohols, and amines, to generate carboxylic acid derivatives or undergo [2+2] cycloaddition reactions to form four-membered rings. The mechanism of the Wolff rearrangement has been the subject of debate since its first use. No single mechanism sufficiently describes the reaction, and there are often competing concerted and carbene-mediated pathways; for simplicity, only the textbook, concerted mechanism is shown below. The reaction was discovered by Ludwig Wolff in 1902. The Wolff rearrangement has great synthetic utility due to the accessibility of α-diazocarbonyl compounds, variety of reactions from the ketene intermediate, and stereochemical retention of the migrating group. However, the Wolff rearrangement has limitations due to the highly reactive nature of α-diazocarbonyl compounds, which can undergo a variety of competing reactions.The Wolff rearrangement can be induced via thermolysis, photolysis, or transition metal catalysis. In this last case, the reaction is sensitive to the transition metal; silver (I) oxide or other Ag(I) catalysts work well and are generally used. The Wolff rearrangement has been used in many total syntheses; the most common use is trapping the ketene intermediate with nucleophiles to form carboxylic acid derivatives. The Arndt-Eistert homologation is a specific example of this use, wherein a carboxylic acid may be elongated by a methylene unit. Another common use is in ring-contraction methods; if the α-diazo ketone is cyclic, the Wolff rearrangement results in a ring-contracted product. The Wolff rearrangement works well in generating ring-strained systems, where other reactions may fail.