Practice Questions for Chapters 1-8 CHEM 4000A
... In the second step, the nucleophilic carbanion attacks the electrophilic carbon of the thioester. A tetrahedral intermediate is formed, and EnzS– is the eventual leaving group. (It’s a good leaving group since S is large so the charge density is fairly low. Also, S is a reasonably electronegative at ...
... In the second step, the nucleophilic carbanion attacks the electrophilic carbon of the thioester. A tetrahedral intermediate is formed, and EnzS– is the eventual leaving group. (It’s a good leaving group since S is large so the charge density is fairly low. Also, S is a reasonably electronegative at ...
C h e m g u id e –... ACID ANHYDRIDES: REACTIONS WITH WATER, ALCOHOLS AND PHENOLS
... and so the top group in your target molecule must come from an acid anhydride, but a bigger one than ethanoic anhydride. You can ignore the other group on the ring as just a distraction. You haven’t come across any reaction which would attach a group like this to a benzene ring, so it must have been ...
... and so the top group in your target molecule must come from an acid anhydride, but a bigger one than ethanoic anhydride. You can ignore the other group on the ring as just a distraction. You haven’t come across any reaction which would attach a group like this to a benzene ring, so it must have been ...
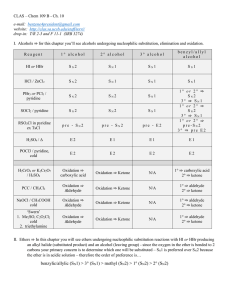
Chem 263 Notes March 2, 2006 Preparation of Aldehydes and
... Use of stronger hydride donors (such as LiAlH4) leads to overreduction of the aldehyde or ketone, right down to the alcohol (aldehydes reduce 1o alcohol and ketones reduce to 2o alcohols). The two hydride donors shown above are not as reactive as LiAlH4 since they are sterically more bulky. This ma ...
... Use of stronger hydride donors (such as LiAlH4) leads to overreduction of the aldehyde or ketone, right down to the alcohol (aldehydes reduce 1o alcohol and ketones reduce to 2o alcohols). The two hydride donors shown above are not as reactive as LiAlH4 since they are sterically more bulky. This ma ...
Microsoft Word
... alcohol 33, which was then converted to the ketone-1,3-dithio acetal 34 (Scheme8). However, further oxidation of the dithio acetal 34 yielded the acid 35 in low yields and hence an alterate synthetic route was adopted. (Scheme-9) Figure In the scheme-9, allylic secondary alcohol 38, was prepared by ...
... alcohol 33, which was then converted to the ketone-1,3-dithio acetal 34 (Scheme8). However, further oxidation of the dithio acetal 34 yielded the acid 35 in low yields and hence an alterate synthetic route was adopted. (Scheme-9) Figure In the scheme-9, allylic secondary alcohol 38, was prepared by ...
Organic Chemistry I Mario Lintz 1st Year MD/PhD Candidate Mario
... o 3) remove the protecting group Alcohol behaves as the nucleophile. (As is often the case) OH easily transfer H to a basic reagent, a problem in some reactions. Conversion of the OH to a removable functional group without an acidic proton protects the alcohol One common method of alcohol pr ...
... o 3) remove the protecting group Alcohol behaves as the nucleophile. (As is often the case) OH easily transfer H to a basic reagent, a problem in some reactions. Conversion of the OH to a removable functional group without an acidic proton protects the alcohol One common method of alcohol pr ...
top 5 organic - No Brain Too Small
... Lose the -Cl of acid chloride and -H of alcohol as HCl(g) Preparation method (alc + c.acid); heat under reflux; add carbonate to neutralise acids, separate ester from impurities by distillation Breaking – reaction with H2O - hydrolysis (break where you make – i.e between O and C=O) ...
... Lose the -Cl of acid chloride and -H of alcohol as HCl(g) Preparation method (alc + c.acid); heat under reflux; add carbonate to neutralise acids, separate ester from impurities by distillation Breaking – reaction with H2O - hydrolysis (break where you make – i.e between O and C=O) ...
67 Preview of Carbonyl Chemistry Kinds of carbonyls 1. Aldehydes
... with α,β-unsaturated ketones; however, they also undergo direct addition to non-conjugated ketones (1,2-additions), aldehydes and will react with alkyl halides and tosylates, and epoxides Mechanism of conjugate addition by organocopper reagents is complex ...
... with α,β-unsaturated ketones; however, they also undergo direct addition to non-conjugated ketones (1,2-additions), aldehydes and will react with alkyl halides and tosylates, and epoxides Mechanism of conjugate addition by organocopper reagents is complex ...
Chapter 16 Aldehydes and Ketones I. Nucleophilic Addition to the
... The carbonyl p electrons shift to oxygen to give the alkoxide The carbonyl carbon changes from trigonal planar to tetrahedral ...
... The carbonyl p electrons shift to oxygen to give the alkoxide The carbonyl carbon changes from trigonal planar to tetrahedral ...
Reactions of Alcohols
... SN2 reaction between R-X and R-OWE NEED TO CONSIDER STERIC HINDERANCE. This might lead to E2! ...
... SN2 reaction between R-X and R-OWE NEED TO CONSIDER STERIC HINDERANCE. This might lead to E2! ...
Chapter 18 Reactions of aromatics
... Mechanism of NBS (Radical) Reaction • Abstraction of a benzylic hydrogen atom generates an intermediate benzylic radical • Reacts with Br2 to yield product • Br· radical cycles back into reaction to carry chain • Br2 produced from reaction of HBr with NBS ...
... Mechanism of NBS (Radical) Reaction • Abstraction of a benzylic hydrogen atom generates an intermediate benzylic radical • Reacts with Br2 to yield product • Br· radical cycles back into reaction to carry chain • Br2 produced from reaction of HBr with NBS ...
Exam 1 - Winona State University
... min. Exams must be turned in immediately upon my call of time up. Grading will be on the basis of a highest possible score of 100 points. I. Multiple Choice – 2 points each, 20 points total II. Nomenclature/Structure – 2 points each, 20 points total III. Synthesis – 5 points each, 15 points total IV ...
... min. Exams must be turned in immediately upon my call of time up. Grading will be on the basis of a highest possible score of 100 points. I. Multiple Choice – 2 points each, 20 points total II. Nomenclature/Structure – 2 points each, 20 points total III. Synthesis – 5 points each, 15 points total IV ...
Carboxylic Acids
... Electron withdrawing groups can increase acid strength by weakening the OH bond and stabilizing the acid anion. The positive inductive effect of E-groups is very small through more than two or three carbon-carbon bonds. Electron donating groups reduce the partially positive charge of carboxyl carbon ...
... Electron withdrawing groups can increase acid strength by weakening the OH bond and stabilizing the acid anion. The positive inductive effect of E-groups is very small through more than two or three carbon-carbon bonds. Electron donating groups reduce the partially positive charge of carboxyl carbon ...
Synthesis of Isobutyl Propionate via Esterification
... we can, by Le Chatelier's principle, increase the concentration of either the alcohol or acid, as noted above. If either one is doubled, the theoretical yield increases to 85%. When one is tripled, it goes to 90%. But note that in the example cited the boiling point of the relatively nonpolar ester ...
... we can, by Le Chatelier's principle, increase the concentration of either the alcohol or acid, as noted above. If either one is doubled, the theoretical yield increases to 85%. When one is tripled, it goes to 90%. But note that in the example cited the boiling point of the relatively nonpolar ester ...
ppt
... • There will be two possible Wittig routes to an alkene. • Analyze the structure retrosynthetically, i.e., work the synthesis out backworks • Disconnect (break the bond of the target that can be formed by a known reaction) the doubly bonded carbons. One becomes the aldehyde or ketone, the other the ...
... • There will be two possible Wittig routes to an alkene. • Analyze the structure retrosynthetically, i.e., work the synthesis out backworks • Disconnect (break the bond of the target that can be formed by a known reaction) the doubly bonded carbons. One becomes the aldehyde or ketone, the other the ...
Exp`t 73
... Dehydration of 2-methylcyclohexanol, 1 (B.P. = 163-166°C) with 85% phosphoric acid yields a mixture of three products: the main product overall (75-80%) is 1-methyl-1-cyclohexene, 2 (B.P. = 110-111 °C); also present are 3-methyl-1-cyclohexene, 3 (B.P.=104 °C) and methylenecyclohexane, 4. The relativ ...
... Dehydration of 2-methylcyclohexanol, 1 (B.P. = 163-166°C) with 85% phosphoric acid yields a mixture of three products: the main product overall (75-80%) is 1-methyl-1-cyclohexene, 2 (B.P. = 110-111 °C); also present are 3-methyl-1-cyclohexene, 3 (B.P.=104 °C) and methylenecyclohexane, 4. The relativ ...
Exam 1 Review Sheet Chapter 15 Chemistry 110b
... Esters and lactones: mechanism of the Fischer esterification; ester syntheses using SN2 chemistry; ester syntheses using activated carbonyl groups. Mechanism of basic saponification of esters, use of isotopes to support mechanisms of acyl vs. alkyl attack, be aware of an example where alkyl attack i ...
... Esters and lactones: mechanism of the Fischer esterification; ester syntheses using SN2 chemistry; ester syntheses using activated carbonyl groups. Mechanism of basic saponification of esters, use of isotopes to support mechanisms of acyl vs. alkyl attack, be aware of an example where alkyl attack i ...
Classification of Halogen Derivatives
... Like KCN, KNO2 form R-ONO while AgNO2 produces R-NO2 as product. Vinyl chloride is less reactive towards nucleophilic substitution reactions due to resonance. Nucleophilic substitution reactions are of two types (a) SN1 type (Unimolecular nucleophilic reactions proceed in two steps: ...
... Like KCN, KNO2 form R-ONO while AgNO2 produces R-NO2 as product. Vinyl chloride is less reactive towards nucleophilic substitution reactions due to resonance. Nucleophilic substitution reactions are of two types (a) SN1 type (Unimolecular nucleophilic reactions proceed in two steps: ...
Dehydration of Cyclohexanol
... carbocations derived from certain 2°alcohols may undergo rearrangement to form more stable carbocations. This can result in the formation of rearranged isomeric alkenes. Both 2° and 3° alcohols primarily undergo the E1 reaction under these conditions, whereas for 1° alcohols and methyl alcohol, symm ...
... carbocations derived from certain 2°alcohols may undergo rearrangement to form more stable carbocations. This can result in the formation of rearranged isomeric alkenes. Both 2° and 3° alcohols primarily undergo the E1 reaction under these conditions, whereas for 1° alcohols and methyl alcohol, symm ...
Benzocaine Synthesis via Esterification
... pipette to transfer the acid. A precipitate is expected to form. Attach a reflux condenser to the flask, secure the apparatus with clamps, and heat the mixture at a gentle reflux for 60-75 minutes using a heating mantle and Variac power controller. The solid should dissolve as it undergoes reaction. ...
... pipette to transfer the acid. A precipitate is expected to form. Attach a reflux condenser to the flask, secure the apparatus with clamps, and heat the mixture at a gentle reflux for 60-75 minutes using a heating mantle and Variac power controller. The solid should dissolve as it undergoes reaction. ...
Chapter 10 - UCSB CLAS
... as epoxides in basic/neutral conditions or rearrangement reactions to produce phenol (primary focus) – since there’s a carbocation formed in the rearrangement reaction this will determine which carbon gets the OH – any additional substituents that can stabilize a carbocation (electron donating) will ...
... as epoxides in basic/neutral conditions or rearrangement reactions to produce phenol (primary focus) – since there’s a carbocation formed in the rearrangement reaction this will determine which carbon gets the OH – any additional substituents that can stabilize a carbocation (electron donating) will ...
Ch 16 Aldehydes and Ketones I
... Nucleophilic Addition to the CarbonOxygen Double Bond • Many nucleophilic additions to the carbonyl are reversible • The results of the reaction depend on the equilibrium • Relative Reactivity – In general, aldehydes are more reactive than ketones – This is due to both steric and electronic factors ...
... Nucleophilic Addition to the CarbonOxygen Double Bond • Many nucleophilic additions to the carbonyl are reversible • The results of the reaction depend on the equilibrium • Relative Reactivity – In general, aldehydes are more reactive than ketones – This is due to both steric and electronic factors ...
Chemistry of Nitrogen-containing Organic
... 1. Find out what an acyl chloride is and draw the general structure. 2. Draw a reaction mechanism for an acyl chloride with 3 carbon atoms reacting with ethylamine. 3. What is this type of reaction called? 4. What type of organic compound is the product? ...
... 1. Find out what an acyl chloride is and draw the general structure. 2. Draw a reaction mechanism for an acyl chloride with 3 carbon atoms reacting with ethylamine. 3. What is this type of reaction called? 4. What type of organic compound is the product? ...
Wolff rearrangement

The Wolff rearrangement is a reaction in organic chemistry in which an α-diazocarbonyl compound is converted into a ketene by loss of dinitrogen with accompanying 1,2-rearrangement. The Wolff rearrangement yields a ketene as an intermediate product, which can undergo nucleophilic attack with weakly acidic nucleophiles such as water, alcohols, and amines, to generate carboxylic acid derivatives or undergo [2+2] cycloaddition reactions to form four-membered rings. The mechanism of the Wolff rearrangement has been the subject of debate since its first use. No single mechanism sufficiently describes the reaction, and there are often competing concerted and carbene-mediated pathways; for simplicity, only the textbook, concerted mechanism is shown below. The reaction was discovered by Ludwig Wolff in 1902. The Wolff rearrangement has great synthetic utility due to the accessibility of α-diazocarbonyl compounds, variety of reactions from the ketene intermediate, and stereochemical retention of the migrating group. However, the Wolff rearrangement has limitations due to the highly reactive nature of α-diazocarbonyl compounds, which can undergo a variety of competing reactions.The Wolff rearrangement can be induced via thermolysis, photolysis, or transition metal catalysis. In this last case, the reaction is sensitive to the transition metal; silver (I) oxide or other Ag(I) catalysts work well and are generally used. The Wolff rearrangement has been used in many total syntheses; the most common use is trapping the ketene intermediate with nucleophiles to form carboxylic acid derivatives. The Arndt-Eistert homologation is a specific example of this use, wherein a carboxylic acid may be elongated by a methylene unit. Another common use is in ring-contraction methods; if the α-diazo ketone is cyclic, the Wolff rearrangement results in a ring-contracted product. The Wolff rearrangement works well in generating ring-strained systems, where other reactions may fail.