Introduction to Subatomic Physics
... According to collision participants (photonuclear reactions, heavy ion reactions, proton induced reactions, neutron production reactions …) According to reaction energy (exothermic, endothermic reactions) According to energy of impinging particles (low energy, high energy, relativistic collision, ul ...
... According to collision participants (photonuclear reactions, heavy ion reactions, proton induced reactions, neutron production reactions …) According to reaction energy (exothermic, endothermic reactions) According to energy of impinging particles (low energy, high energy, relativistic collision, ul ...
summer learning G10
... 4. Jessica heated some bright blue copper(II) nitrate crystals in a test-tube. She noticed brown nitrogen dioxide gas being produced. When a glowing splint was held at the top of the test-tube, it relit, proving that oxygen gas was also produced. A fine black solid, copper(II) oxide, was left in the ...
... 4. Jessica heated some bright blue copper(II) nitrate crystals in a test-tube. She noticed brown nitrogen dioxide gas being produced. When a glowing splint was held at the top of the test-tube, it relit, proving that oxygen gas was also produced. A fine black solid, copper(II) oxide, was left in the ...
ch 2 matter and energy
... Radioactive isotope Radioactive decay occurs when nuclei of unstable isotopes spontaneously emit fast-moving chunks of matter (alpha particles or beta particles), highenergy radiation (gamma rays), or both at a fixed rate. A particular radioactive isotope may emit any one or a combination of the thr ...
... Radioactive isotope Radioactive decay occurs when nuclei of unstable isotopes spontaneously emit fast-moving chunks of matter (alpha particles or beta particles), highenergy radiation (gamma rays), or both at a fixed rate. A particular radioactive isotope may emit any one or a combination of the thr ...
Notes - Organization of Matter
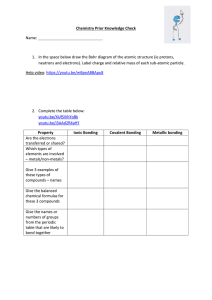
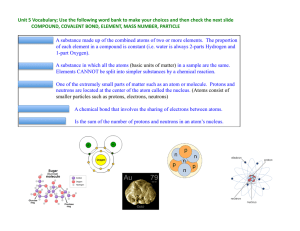
... • Compounds are pure substances that are composed of two or more atoms that are chemically combined • Compounds can only be changed into simpler substances called elements by chemical changes ...
... • Compounds are pure substances that are composed of two or more atoms that are chemically combined • Compounds can only be changed into simpler substances called elements by chemical changes ...
Adv review key
... B) Valence electrons- outer shell electrons C) Metals a. Lend valence electrons b. 1 – 4 valence electrons c. Form positive ions ( more protons than electrons) D) Nonmetals a. Borrow valence electrons b. 4 - 8 valence electrons c. Form negative ions (more electrons than protons) E) Metals lend and n ...
... B) Valence electrons- outer shell electrons C) Metals a. Lend valence electrons b. 1 – 4 valence electrons c. Form positive ions ( more protons than electrons) D) Nonmetals a. Borrow valence electrons b. 4 - 8 valence electrons c. Form negative ions (more electrons than protons) E) Metals lend and n ...
Midterm Review Answers
... sodium fluoride, and sodium hydroxide you need to separate the barium, mercury(II), and magnesium ions. How would you go about separating these ions? Discuss your experimental procedure and defend your answer. Using the solubility rules, chloride compounds are generally soluble, but mercury is an ex ...
... sodium fluoride, and sodium hydroxide you need to separate the barium, mercury(II), and magnesium ions. How would you go about separating these ions? Discuss your experimental procedure and defend your answer. Using the solubility rules, chloride compounds are generally soluble, but mercury is an ex ...
APS 1st semester exam review 2016
... B) Valence electrons- outer shell electrons C) Metals a. Lend valence electrons b. 1 – 4 valence electrons c. Form positive ions ( more protons than electrons) D) Nonmetals a. Borrow valence electrons b. 4 - 8 valence electrons c. Form negative ions (more electrons than protons) E) Metals lend and n ...
... B) Valence electrons- outer shell electrons C) Metals a. Lend valence electrons b. 1 – 4 valence electrons c. Form positive ions ( more protons than electrons) D) Nonmetals a. Borrow valence electrons b. 4 - 8 valence electrons c. Form negative ions (more electrons than protons) E) Metals lend and n ...
EOCT Physical Science Atomic Theory
... D. They must be found in the same group in the periodic table. 15.A chemical change occurs when a substance A. changes state. B. gains or loses electrons. C. reaches its boiling point. D. is ground into a powder. 16. Which of the following is a chemical change? A. Water is decomposed into hydrogen g ...
... D. They must be found in the same group in the periodic table. 15.A chemical change occurs when a substance A. changes state. B. gains or loses electrons. C. reaches its boiling point. D. is ground into a powder. 16. Which of the following is a chemical change? A. Water is decomposed into hydrogen g ...
Document
... a) A force acting on an object may cause a change in shape of the object. b) A force applied to an elastic object such as a spring will result in the object stretching and storing elastic potential energy. c) For an object that is able to recover its original shape, elastic potential energy is store ...
... a) A force acting on an object may cause a change in shape of the object. b) A force applied to an elastic object such as a spring will result in the object stretching and storing elastic potential energy. c) For an object that is able to recover its original shape, elastic potential energy is store ...