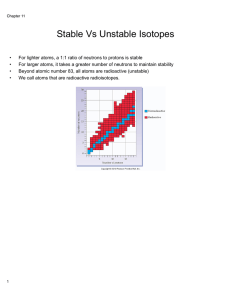
Isotopes of an atom have the same number of protons, but a different
... Above the atomic number 82, all elements are radioactive. The nucleons (P or N) are in a state of continual motion (natural isotopes) If additional N or a deficiency of N occurs, the atom attempts to regain its stability by giving off either a photon, such as gamma ray, or particle from the nu ...
... Above the atomic number 82, all elements are radioactive. The nucleons (P or N) are in a state of continual motion (natural isotopes) If additional N or a deficiency of N occurs, the atom attempts to regain its stability by giving off either a photon, such as gamma ray, or particle from the nu ...
Summative Assessment Review!
... • An alpha particle is simply a helium nuclei (He) which is ejected with high energy from an unstable nucleus ...
... • An alpha particle is simply a helium nuclei (He) which is ejected with high energy from an unstable nucleus ...
Radioactivity Revision Questions Decay – Nucleus
... Sometimes the nucleus of an atom is unstable. A change will occur in the nucleus to make it more stable. The change is called a decay 2. During Radioactive Decay, what can a Nucleus Emit? When a nucleus decays it will emit (give out) some particles or waves. Emitting particles or waves from the nucl ...
... Sometimes the nucleus of an atom is unstable. A change will occur in the nucleus to make it more stable. The change is called a decay 2. During Radioactive Decay, what can a Nucleus Emit? When a nucleus decays it will emit (give out) some particles or waves. Emitting particles or waves from the nucl ...
Atomic Theory and the Nuclear Atom
... ______________ are very involved in this force. 1. Atoms having low atomic numbers (up to about ___) are more stable when the neutron-proton ratio is ________. 2. Atoms having large atomic numbers are more stable when there are greater numbers of _______________ than ________________. 3. No stable n ...
... ______________ are very involved in this force. 1. Atoms having low atomic numbers (up to about ___) are more stable when the neutron-proton ratio is ________. 2. Atoms having large atomic numbers are more stable when there are greater numbers of _______________ than ________________. 3. No stable n ...
Beryllium isotopes in geochronology Cosmogenic Be and Be
... rocks at three sites on the Level 2 terrace in upper Holtwood Gorge, Pennsylvania, approximately 50 km upstream of Chesapeake Bay [38]. ...
... rocks at three sites on the Level 2 terrace in upper Holtwood Gorge, Pennsylvania, approximately 50 km upstream of Chesapeake Bay [38]. ...
Quiz: The Atom (Open Notes)
... 11. T or F A proton has a charge that is equal in force but opposite in charge to each electron. 12. T or F Protons and electrons are about equal in mass. 13. T or F The mass of an atom depends on the number of protons and neutrons in its nucleus. 14. The atomic number of an atom is determined by th ...
... 11. T or F A proton has a charge that is equal in force but opposite in charge to each electron. 12. T or F Protons and electrons are about equal in mass. 13. T or F The mass of an atom depends on the number of protons and neutrons in its nucleus. 14. The atomic number of an atom is determined by th ...
Nuclear Chemistry - Mrs. Carlyle`s Classroom
... 1. Neutron moderation – a moderator slows down the neutrons so that they do not hit the next uranium atom with as much force; therefore, reducing the number of uranium atoms being split. 2. Neutron absorption – control rods decreases the number of neutrons by absorption. This prevents too many urani ...
... 1. Neutron moderation – a moderator slows down the neutrons so that they do not hit the next uranium atom with as much force; therefore, reducing the number of uranium atoms being split. 2. Neutron absorption – control rods decreases the number of neutrons by absorption. This prevents too many urani ...
Example 27-3 The Binding Energy of 4He
... binding energy of the nucleus divided by the number of nucleons in the nucleus. ...
... binding energy of the nucleus divided by the number of nucleons in the nucleus. ...
A Z N Atomic Mass: A = Z + N Mass and Rest Energy m =
... because the nuclear force is a much stronger force than that of the electric force caused by repulsion. The nuclear force is sometimes called the strong force. It is the interaction that binds nucleons together in the nucleus. The force works with dimensions on the nuclear level that are MUCH smalle ...
... because the nuclear force is a much stronger force than that of the electric force caused by repulsion. The nuclear force is sometimes called the strong force. It is the interaction that binds nucleons together in the nucleus. The force works with dimensions on the nuclear level that are MUCH smalle ...
Physical Science: Nuclear Chemistry Study Guide
... 9. In this example, what are the chemical symbols of the products of the decay? a. Ra and Rn c. Rn and He b. Ra and He d. He is the only product. 10. During beta decay, a nucleus a. gives up two protons and two neutrons. b. maintains the same number of protons and neutrons. c. loses a proton and gai ...
... 9. In this example, what are the chemical symbols of the products of the decay? a. Ra and Rn c. Rn and He b. Ra and He d. He is the only product. 10. During beta decay, a nucleus a. gives up two protons and two neutrons. b. maintains the same number of protons and neutrons. c. loses a proton and gai ...
Elements!
... (center of atom) The electric charge is positive (+) Weighs 1 amu (atomic mass unit) Each element has a unique number of protons (All Oxygen atoms have 8 protons; all Iron have 26) ...
... (center of atom) The electric charge is positive (+) Weighs 1 amu (atomic mass unit) Each element has a unique number of protons (All Oxygen atoms have 8 protons; all Iron have 26) ...
Ch 21 Nuclear - coolchemistrystuff
... Radioactive series (or nuclear disintegration series): a series of nuclear reactions that begins with an unstable nucleus and terminates with a stable one Magic numbers: nuclei with 2, 8, 20, 28, 50, or 82 protons or 2, 8, 20, 28, 50, 82, or 126 neutrons; results in very stable nuclei Nuclei wit ...
... Radioactive series (or nuclear disintegration series): a series of nuclear reactions that begins with an unstable nucleus and terminates with a stable one Magic numbers: nuclei with 2, 8, 20, 28, 50, or 82 protons or 2, 8, 20, 28, 50, 82, or 126 neutrons; results in very stable nuclei Nuclei wit ...
Chapter 7 - Bakersfield College
... A. For a given nucleus, the binding energy per nucleon is found by dividing the total binding energy of the nucleus by the number of nucleons (protons and neutrons) it contains; the greater the binding energy per nucleon, the more stable the nucleus. B. Nuclear fission is the splitting of a heavy nu ...
... A. For a given nucleus, the binding energy per nucleon is found by dividing the total binding energy of the nucleus by the number of nucleons (protons and neutrons) it contains; the greater the binding energy per nucleon, the more stable the nucleus. B. Nuclear fission is the splitting of a heavy nu ...
2.10 Basic Nuclear Chemistry
... A. This term refers to the nucleus of an atom. It is without the electron cloud. B. Nucleons 1. This term refers to the components of an atom’s nucleus – protons & neutrons. 2. Remember, each proton and neutron has a mass = 1 AMU or Dalton; protons carry a positive charge and neutrons carry no charg ...
... A. This term refers to the nucleus of an atom. It is without the electron cloud. B. Nucleons 1. This term refers to the components of an atom’s nucleus – protons & neutrons. 2. Remember, each proton and neutron has a mass = 1 AMU or Dalton; protons carry a positive charge and neutrons carry no charg ...
nuclear force
... • The electron will combine with a proton in the nucleus and form a neutron. • The atomic number decreases by one but the mass number stays the same. ...
... • The electron will combine with a proton in the nucleus and form a neutron. • The atomic number decreases by one but the mass number stays the same. ...
Review and Radioactivity
... A nucleus which is in an excited state may emit one or more photons (packets of electromagnetic radiation) of discrete energies. The emission of gamma rays does not alter the number of protons or neutrons in the nucleus but instead has the effect of moving the nucleus from a higher to a lower energy ...
... A nucleus which is in an excited state may emit one or more photons (packets of electromagnetic radiation) of discrete energies. The emission of gamma rays does not alter the number of protons or neutrons in the nucleus but instead has the effect of moving the nucleus from a higher to a lower energy ...
Unit 2: The Atom
... •Alpha decay is how elements greater than Bismuth try to become stable. •They will emit an alpha particle (2 neutrons and 2 protons) to try to become stable. •Alpha reactions will always have He on the right side! •To balance: write the upper and lower equations! ...
... •Alpha decay is how elements greater than Bismuth try to become stable. •They will emit an alpha particle (2 neutrons and 2 protons) to try to become stable. •Alpha reactions will always have He on the right side! •To balance: write the upper and lower equations! ...
Fission and Fusion of Atomic Nuclei
... hydrogen) are fused together at extremely high temperatures and pressures to form heavier elements (such as helium) • Fusion reactions release more energy than fission reactions • Requires extremely high energies to initiate and sustain • Sun is powered by fusion ...
... hydrogen) are fused together at extremely high temperatures and pressures to form heavier elements (such as helium) • Fusion reactions release more energy than fission reactions • Requires extremely high energies to initiate and sustain • Sun is powered by fusion ...
Isotopes of an atom have the same number of protons, but a different
... Above the atomic number 82, all elements are radioactive. The nucleons (P or N) are in a state of continual motion (natural isotopes) If additional N or a deficiency of N occurs, the atom attempts to regain its stability by giving off either a photon, such as gamma ray, or particle from the nu ...
... Above the atomic number 82, all elements are radioactive. The nucleons (P or N) are in a state of continual motion (natural isotopes) If additional N or a deficiency of N occurs, the atom attempts to regain its stability by giving off either a photon, such as gamma ray, or particle from the nu ...
Chapter 9: Nuclear Changes
... - ____________- the process by which an unstable nucleus emits one or more particles or energy in the form of electromagnetic radiation - _____ ________- process radioactive materials undergo - radioactive materials have an unstable nucleus - become stable by undergoing nuclear decay - results in a ...
... - ____________- the process by which an unstable nucleus emits one or more particles or energy in the form of electromagnetic radiation - _____ ________- process radioactive materials undergo - radioactive materials have an unstable nucleus - become stable by undergoing nuclear decay - results in a ...
BellWork 2/16/2015
... Mg2+ ClFe2+ O2These elements are written to describe what? Grab a textbook and look it up if needed. ...
... Mg2+ ClFe2+ O2These elements are written to describe what? Grab a textbook and look it up if needed. ...