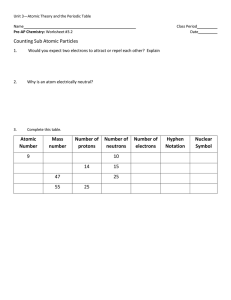
U2-LM2B-WS – FUNDAMENTAL PARTICLES OF ATOM 1. The
... The three fundamental subatomic particles are: ___________, the ______________and electron proton the _____________. The two particles that have equal but opposite charge are_______and neutron proton the _____________. The particles that reside in the nucleus are called nucleons and consist electron ...
... The three fundamental subatomic particles are: ___________, the ______________and electron proton the _____________. The two particles that have equal but opposite charge are_______and neutron proton the _____________. The particles that reside in the nucleus are called nucleons and consist electron ...
NUCLEAR CHEMISTRY
... by 2 protons + 2 neutrons) AZ My AZ X + 4 He 5. Illustrate beta emission using new isotope in step 4 with equation and marshmallows AZ Y + 0-1 β 6. Label the following decay sequence as α or β emission: 238 U 234 Th 234 Pa 230 Th 226 Ra 222 Rn ...
... by 2 protons + 2 neutrons) AZ My AZ X + 4 He 5. Illustrate beta emission using new isotope in step 4 with equation and marshmallows AZ Y + 0-1 β 6. Label the following decay sequence as α or β emission: 238 U 234 Th 234 Pa 230 Th 226 Ra 222 Rn ...
RAD 354 Chapt 3 Structure of Matter
... Varied #’s of mass# & atomic# • Isotope = same # of protons but different # of neutrons • Isobar = different # of protons and neutrons BUT same TOTAL number of nucleons • Isotones = same # of neutrons BUT different # of protons • Isomer = same atomic number and same atomic mass number- BUT exist at ...
... Varied #’s of mass# & atomic# • Isotope = same # of protons but different # of neutrons • Isobar = different # of protons and neutrons BUT same TOTAL number of nucleons • Isotones = same # of neutrons BUT different # of protons • Isomer = same atomic number and same atomic mass number- BUT exist at ...
Radioisotopes
... (Nuclides) of the same chemical element, each having a different atomic mass (mass number) • Isotopes of an element have nuclei with the same number of protons (the same atomic number) but different numbers of neutrons. • Therefore, isotopes have different mass numbers, which give the total number o ...
... (Nuclides) of the same chemical element, each having a different atomic mass (mass number) • Isotopes of an element have nuclei with the same number of protons (the same atomic number) but different numbers of neutrons. • Therefore, isotopes have different mass numbers, which give the total number o ...
File
... A neutral atom consists of a positively charged nucleus (composed of protons and neutrons) associated with orbital electrons. The atomic number (Z) is the number of protons in the nucleus The neutron number (N) is the number of neutrons in the nucleus. The mass number (A) is the sum of the protons a ...
... A neutral atom consists of a positively charged nucleus (composed of protons and neutrons) associated with orbital electrons. The atomic number (Z) is the number of protons in the nucleus The neutron number (N) is the number of neutrons in the nucleus. The mass number (A) is the sum of the protons a ...
answers_to_questions_on_pages_100
... Answers to Questions on Pages 100-101 5. A) Protons and neutrons are found in the nucleus while the electrons orbit the nucleus in their respective energy levels. B) The protons and neutrons make up the majority of the mass of an atom. C) CORRECTION – The electrons make up most of the space as their ...
... Answers to Questions on Pages 100-101 5. A) Protons and neutrons are found in the nucleus while the electrons orbit the nucleus in their respective energy levels. B) The protons and neutrons make up the majority of the mass of an atom. C) CORRECTION – The electrons make up most of the space as their ...
Nuclear Power Plant Notes
... travel many meters in air and many centimeters in human tissue. It readily penetrates most materials and is sometimes called "penetrating radiation." ...
... travel many meters in air and many centimeters in human tissue. It readily penetrates most materials and is sometimes called "penetrating radiation." ...
The New Alchemy
... fusion. Splitting of the nucleus of an atom into smaller parts The Search Went On Half life – time required for half the atoms of a radioactive material to decay Periodic Table Transition elements – an element that is found in the center of the periodic table that is a metal Lanthanides – Elements h ...
... fusion. Splitting of the nucleus of an atom into smaller parts The Search Went On Half life – time required for half the atoms of a radioactive material to decay Periodic Table Transition elements – an element that is found in the center of the periodic table that is a metal Lanthanides – Elements h ...
Modern Physics TEST
... the energy needed to remove one of the nucleons b. the average energy with which any nucleon is bound in the nucleus c. the energy released when nucleons bind together to form a stable nucleus d. the mass of the nucleus times c ____ 11. If the stable nuclei are plotted with neutron number versus pro ...
... the energy needed to remove one of the nucleons b. the average energy with which any nucleon is bound in the nucleus c. the energy released when nucleons bind together to form a stable nucleus d. the mass of the nucleus times c ____ 11. If the stable nuclei are plotted with neutron number versus pro ...
Proton Positively charged subatomic (smaller than an atom) particle
... Negativity charged subatomic (smaller than an atom) particle found orbiting the nucleus. Atomic Number- Represents the number of protons, which determines the element type. ...
... Negativity charged subatomic (smaller than an atom) particle found orbiting the nucleus. Atomic Number- Represents the number of protons, which determines the element type. ...
Concept Lecture Outline – Radioactivity and Nuclear Reactions
... b. Either 2 or 3 neutrons are also produced with the products. c. These neutrons act as "bullets" to ram into other unstable nuclei and split them, resulting in a chain reaction. d. All nuclear reactors in the world are fission reactors. 1) They cannot "blow up" like an atom bomb. 2) They produce ra ...
... b. Either 2 or 3 neutrons are also produced with the products. c. These neutrons act as "bullets" to ram into other unstable nuclei and split them, resulting in a chain reaction. d. All nuclear reactors in the world are fission reactors. 1) They cannot "blow up" like an atom bomb. 2) They produce ra ...
Unit 2 Notes - School City of Hobart
... • For a nuclide to be considered radioactive, it must have an unstable nucleus due to an imbalance between # of protons and neutrons ~1:1 ratio = stable *The further an isotope is from a 1:1 ratio, the more likely it is to be radioactive (Figure 21.2, p.881) • Types of Nuclear Decay (aka Radioactivi ...
... • For a nuclide to be considered radioactive, it must have an unstable nucleus due to an imbalance between # of protons and neutrons ~1:1 ratio = stable *The further an isotope is from a 1:1 ratio, the more likely it is to be radioactive (Figure 21.2, p.881) • Types of Nuclear Decay (aka Radioactivi ...
Critical Thinking Questions 2
... (a) α decay: the nucleus loses an α particle ( !!He!! ) (b) β decay: a neutron in the nucleus is converted into a proton and an electron. The electron is ejected from the nucleus. (c) Positron or β+ emission: a proton in the nucleus is converted into a neutron and a positron. The positron is ejected ...
... (a) α decay: the nucleus loses an α particle ( !!He!! ) (b) β decay: a neutron in the nucleus is converted into a proton and an electron. The electron is ejected from the nucleus. (c) Positron or β+ emission: a proton in the nucleus is converted into a neutron and a positron. The positron is ejected ...
Nuclear Reactions
... • differ in their number of neutrons in the nucleus • This gives them a different atomic mass. • The nucleus of an isotope with a certain atomic number and mass is called a nuclide. • Radiation energy is given off from unstable (large) nuclides. – Radioactive decay ...
... • differ in their number of neutrons in the nucleus • This gives them a different atomic mass. • The nucleus of an isotope with a certain atomic number and mass is called a nuclide. • Radiation energy is given off from unstable (large) nuclides. – Radioactive decay ...
IB-ATOMIC-AND-NUCLEAR-PHYSICS-DEFINITIONS
... RADIOACTIVE DECAY: A random and spontaneous process in which unstable nuclei emit a particle (disintegrate). The rate decreases exponentially with time. NATURAL RADIOACTIVE DECAY: The emission of an alpha or beta particle. NUCLEAR STRONG FORCE: The force that holds the particles of a nucleus togethe ...
... RADIOACTIVE DECAY: A random and spontaneous process in which unstable nuclei emit a particle (disintegrate). The rate decreases exponentially with time. NATURAL RADIOACTIVE DECAY: The emission of an alpha or beta particle. NUCLEAR STRONG FORCE: The force that holds the particles of a nucleus togethe ...
Beyond Element 83 are very unstable (radioactive)
... unstable (radioactive) • No amount neutrons can hold nucleus together once it has 83+ protons • All Elements 83 and above on PT are radioactive • Other elements may have radioactive isotopes applet ...
... unstable (radioactive) • No amount neutrons can hold nucleus together once it has 83+ protons • All Elements 83 and above on PT are radioactive • Other elements may have radioactive isotopes applet ...
Chapter 32 Nuclear Physics
... nuclei, particularly those of the heavier elements, are unstable and spontaneous break apart, or disintegrate, into other nuclei. This spontaneous disintegration is called radioactive decay, and it is accompanied by the emission of certain types of particles and high-energy photons. While these part ...
... nuclei, particularly those of the heavier elements, are unstable and spontaneous break apart, or disintegrate, into other nuclei. This spontaneous disintegration is called radioactive decay, and it is accompanied by the emission of certain types of particles and high-energy photons. While these part ...
Serway_PSE_quick_ch45
... have too many neutrons for the nucleus to be stable. Beta decay in which electrons are ejected decreases the number of neutrons and increases the number of protons in order to stabilize the nucleus. ...
... have too many neutrons for the nucleus to be stable. Beta decay in which electrons are ejected decreases the number of neutrons and increases the number of protons in order to stabilize the nucleus. ...
3-2 Radioactivity and the nucleus
... He concluded that most of the atom is made of ‘empty space’, and that the core of the atom carried a positive charge (he called the core nucleus and the positive charges the protons). ...
... He concluded that most of the atom is made of ‘empty space’, and that the core of the atom carried a positive charge (he called the core nucleus and the positive charges the protons). ...
10.2
... ‘empty space’, and that the core of the atom carried a positive charge (he called the core nucleus and the positive charges the protons). • He also suggested that electrons orbit the nucleus like planets orbit the Sun (Fig.3 p.281). ...
... ‘empty space’, and that the core of the atom carried a positive charge (he called the core nucleus and the positive charges the protons). • He also suggested that electrons orbit the nucleus like planets orbit the Sun (Fig.3 p.281). ...