Atomic Structure and the Periodic Table
... • Electron cloud – cloud that surrounds the nucleus of an atom that describes the region in which an electron is most likely to be. – Example: students in a school ...
... • Electron cloud – cloud that surrounds the nucleus of an atom that describes the region in which an electron is most likely to be. – Example: students in a school ...
Topic 7 Atomic and Nuclear Physics
... Proton number (Z): The number of protons in a nucleus. Neutron number (N): The number of neutrons in a nucleus. Radioactive half-life: The time taken for half the original nuclei in a sample to decay. Artificial (induced) transmutation: The change of a nucleus from one form to another due to the bom ...
... Proton number (Z): The number of protons in a nucleus. Neutron number (N): The number of neutrons in a nucleus. Radioactive half-life: The time taken for half the original nuclei in a sample to decay. Artificial (induced) transmutation: The change of a nucleus from one form to another due to the bom ...
AP Chem
... even number of neutrons. The least stable situation is when both numbers are odd. There are only four (or five) stable odd/odd nuclei. Nuclides with a mass number over 200 usually undergo alpha decay. They emit a particle consisting of two protons and two neutrons. Nuclides with too many neutrons un ...
... even number of neutrons. The least stable situation is when both numbers are odd. There are only four (or five) stable odd/odd nuclei. Nuclides with a mass number over 200 usually undergo alpha decay. They emit a particle consisting of two protons and two neutrons. Nuclides with too many neutrons un ...
4.1 The Concepts of Force and Mass
... This gives rise to an energy quantization and to the appearance of the so-called magic numbers (analog of noble gases): 2, 8, 20, 28, ...
... This gives rise to an energy quantization and to the appearance of the so-called magic numbers (analog of noble gases): 2, 8, 20, 28, ...
30.1 Radioactivity The atom is the smallest unit of achemical
... It is found that nuclei with mass numbers greater than about 100 spontaneously decay into other types of nuclei. Such nuclei are said to be radioactive, and there are three main types of such decays. 1- Alpha (α) decay is the spontaneous emission of an alpha particle (identical to a helium nucleus) ...
... It is found that nuclei with mass numbers greater than about 100 spontaneously decay into other types of nuclei. Such nuclei are said to be radioactive, and there are three main types of such decays. 1- Alpha (α) decay is the spontaneous emission of an alpha particle (identical to a helium nucleus) ...
Chapter 21 Powerpoint: Nuclear Chemistry
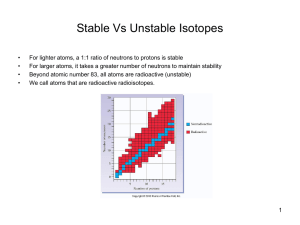
... gets and the further from a 1:1 ratio of protons and neutrons, the less stable the atom is After element 83 (Bismuth) the elements are naturally unstable and may emit decay particles ...
... gets and the further from a 1:1 ratio of protons and neutrons, the less stable the atom is After element 83 (Bismuth) the elements are naturally unstable and may emit decay particles ...
atoms - Groupfusion.net
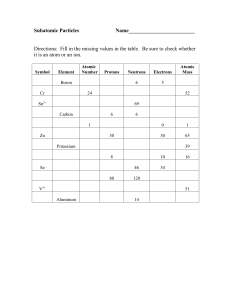
... Normally, this is the same for every atom of an element, therefore number of protons identifies the element. In a “normal”, neutral atom, number of protons = number of electrons Mass Number = the number of protons + number of neutrons = A Number of neutrons in an atom of an element can vary. These a ...
... Normally, this is the same for every atom of an element, therefore number of protons identifies the element. In a “normal”, neutral atom, number of protons = number of electrons Mass Number = the number of protons + number of neutrons = A Number of neutrons in an atom of an element can vary. These a ...
Nuclear radiation 4
... output of the reactor is 1·4GW. How many fission reactions are produced in one hour? ...
... output of the reactor is 1·4GW. How many fission reactions are produced in one hour? ...
Isotope Notes
... b. In order for a nucleus to be _____________________, there must be enough neutrons present between the protons to block the repulsive forces between the protons. c. If an isotope of an element is _________________________, it is radioactive and will undergo nuclear decay. ...
... b. In order for a nucleus to be _____________________, there must be enough neutrons present between the protons to block the repulsive forces between the protons. c. If an isotope of an element is _________________________, it is radioactive and will undergo nuclear decay. ...
Atomic Structure and Radioactivity
... It was found that magnetic field splits the radiation from radioactive materials into 3 parts. Alpha () particles positively charged Beta () particles negatively charged electrons Gamma () particles (rays) no charge - decay: the nucleus composition does not change. -decay occurs in larg ...
... It was found that magnetic field splits the radiation from radioactive materials into 3 parts. Alpha () particles positively charged Beta () particles negatively charged electrons Gamma () particles (rays) no charge - decay: the nucleus composition does not change. -decay occurs in larg ...
NUCLEAR CHEMISTRY
... intermediate mass 2. The mass of the products is less than the mass of the reactants. Missing mass is converted to energy a. Small amounts of missing mass are converted to HUGE amounts of energy (E = mc2) ...
... intermediate mass 2. The mass of the products is less than the mass of the reactants. Missing mass is converted to energy a. Small amounts of missing mass are converted to HUGE amounts of energy (E = mc2) ...
The Band of Stability
... distances. If there are too few neutrons, or too many neutrons, the nucleus becomes unstable. If an atom has more than 82 protons in the nucleus, there is no arrangement of neutrons that can produce more attractive forces than repulsive forces. Therefore, all isotopes of elements beyond lead are rad ...
... distances. If there are too few neutrons, or too many neutrons, the nucleus becomes unstable. If an atom has more than 82 protons in the nucleus, there is no arrangement of neutrons that can produce more attractive forces than repulsive forces. Therefore, all isotopes of elements beyond lead are rad ...
Nuclear Notes
... A crucial factor in the stability of a nucleus is the ratio of neutron number to proton number. Nuclides with more the 20 protons require a larger number of neutrons than protons to moderate the effect of increasing proton repulsions. (Nuclides with less that 20 protons tend to have an equal number ...
... A crucial factor in the stability of a nucleus is the ratio of neutron number to proton number. Nuclides with more the 20 protons require a larger number of neutrons than protons to moderate the effect of increasing proton repulsions. (Nuclides with less that 20 protons tend to have an equal number ...
nuclear chemistry
... If a system loses mass, it loses energy( exothermic) If a system gains mass, it gains energy ( endothermic) Since c2 is a very large number, small mass changes create large energy changes NUCLEAR BINDING ENERGY The mass of a nucleus is less than the mass of its nucleons This is termed mas ...
... If a system loses mass, it loses energy( exothermic) If a system gains mass, it gains energy ( endothermic) Since c2 is a very large number, small mass changes create large energy changes NUCLEAR BINDING ENERGY The mass of a nucleus is less than the mass of its nucleons This is termed mas ...
Nuclear Chemistry
... • Unstable nuclei are naturally “built wrong” and “fall apart” • An unstable nucleus undergoes transmutation, changing from one element into another – the nucleus changes # of protons! ...
... • Unstable nuclei are naturally “built wrong” and “fall apart” • An unstable nucleus undergoes transmutation, changing from one element into another – the nucleus changes # of protons! ...
Document
... The atomic number, Z, equals the number of protons in the nucleus. The neutron number, N, is the number of neutrons in the nucleus. The mass number, A, is the number of nucleons in the nucleus. A=Z+N “Nucleon” is a generic term used to refer to either a proton or a neutron. The mass number is not th ...
... The atomic number, Z, equals the number of protons in the nucleus. The neutron number, N, is the number of neutrons in the nucleus. The mass number, A, is the number of nucleons in the nucleus. A=Z+N “Nucleon” is a generic term used to refer to either a proton or a neutron. The mass number is not th ...
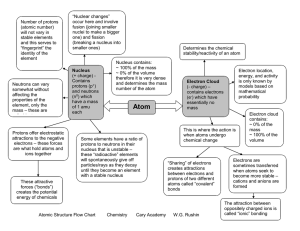
Atomic Structure Flow Chart Chemistry Cary Academy W.G. Rushin
... therefore it is very dense and determines the mass number of the atom ...
... therefore it is very dense and determines the mass number of the atom ...
nuclear chemistry - La Salle High School
... A. The nucleus of an atom is stable if it does NOT change into another nuclide without adding outside energy. B. Look at each element, determine what nuclides of the elements are stable, plot stable nuclides on graph. No. of protons x axis No. of neutrons y-axis ...
... A. The nucleus of an atom is stable if it does NOT change into another nuclide without adding outside energy. B. Look at each element, determine what nuclides of the elements are stable, plot stable nuclides on graph. No. of protons x axis No. of neutrons y-axis ...
Topic 12- Nuclear Chem Reg Rev
... *The reaction converts mass to energy *Einstein’s eq. E=mc2 describes the relationship between energy and matter ...
... *The reaction converts mass to energy *Einstein’s eq. E=mc2 describes the relationship between energy and matter ...
Chapter 21: Nuclear Chemistry
... We plot # of neutrons v. number of protons: n/p ratio where stable nuclei reside is called the 'belt of stability' belt of stability ends at atomic number 83: all nuclei with atomic number 84 are radioactive ...
... We plot # of neutrons v. number of protons: n/p ratio where stable nuclei reside is called the 'belt of stability' belt of stability ends at atomic number 83: all nuclei with atomic number 84 are radioactive ...
Nuclear Stability
... The Nucleus: A Chemist’s View • All nuclides with 84 or more protons are unstable with respect to radio active decay. • Light nuclides are stable when neutron/proton = 1. For heavier elements the neutron /proton ratio required for stability is greater than 1 and increases with Z. • Nuclides with eve ...
... The Nucleus: A Chemist’s View • All nuclides with 84 or more protons are unstable with respect to radio active decay. • Light nuclides are stable when neutron/proton = 1. For heavier elements the neutron /proton ratio required for stability is greater than 1 and increases with Z. • Nuclides with eve ...