Chapter 4.1
... Atomic theory is the study of the nature of atoms and how atoms combine to form all types of matter. ...
... Atomic theory is the study of the nature of atoms and how atoms combine to form all types of matter. ...
Nuclear Reactions
... Nuclear reactions The stability of isotopes is based on the ratio of neutrons and protons in its nucleus. Although most nuclei are stable, some are unstable and spontaneously decay, emitting radiation. Each radioactive isotope has a specific mode and rate of decay (half-life). A change in the nucleu ...
... Nuclear reactions The stability of isotopes is based on the ratio of neutrons and protons in its nucleus. Although most nuclei are stable, some are unstable and spontaneously decay, emitting radiation. Each radioactive isotope has a specific mode and rate of decay (half-life). A change in the nucleu ...
File
... flying apart. *Nuclear force = the force between nucleons i.e.: protons & neutrons Neutrons play a key role stabilizing the nucleus. Therefore, the ratio of neutrons to protons (n/p) is an important factor in determining the stability of nuclides. ...
... flying apart. *Nuclear force = the force between nucleons i.e.: protons & neutrons Neutrons play a key role stabilizing the nucleus. Therefore, the ratio of neutrons to protons (n/p) is an important factor in determining the stability of nuclides. ...
25.3 section summary
... Nuclear fission occurs when fissionable isotopes are bombarded with neutrons. The fissionable atom breaks into two fragments of about the same size, and in the process they release more neutrons and energy. Neutron moderation is the process that reduces the speed of neutrons. Sometimes water is used ...
... Nuclear fission occurs when fissionable isotopes are bombarded with neutrons. The fissionable atom breaks into two fragments of about the same size, and in the process they release more neutrons and energy. Neutron moderation is the process that reduces the speed of neutrons. Sometimes water is used ...
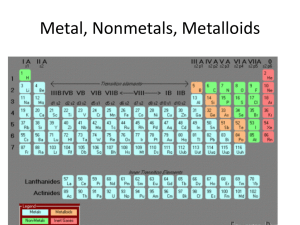
Atomic Theory and Structure
... • Isotopes are atoms that have the same number of protons, but different numbers of neutrons • Mass Number is the sum of protons and neutrons in an atom • Atomic Mass is the average mass of an element’s isotopes ...
... • Isotopes are atoms that have the same number of protons, but different numbers of neutrons • Mass Number is the sum of protons and neutrons in an atom • Atomic Mass is the average mass of an element’s isotopes ...
Nuclear Chemistry - Northwest ISD Moodle
... • PROTONS give the atom its identity • Held together by a very strong nuclear force o One of the four fundamental forces in our universe o Incredibly powerful o Releasing nuclear force results in a nuclear reaction ...
... • PROTONS give the atom its identity • Held together by a very strong nuclear force o One of the four fundamental forces in our universe o Incredibly powerful o Releasing nuclear force results in a nuclear reaction ...
Radioactivity - Teach Nuclear
... Produced when the nucleus of an atom is in an excited state and then releases energy, becoming more stable When a nucleus emits an or β particle, the daughter nucleus is sometimes left in an excited state. It can then jump down to a lower level by emitting a gamma ray ...
... Produced when the nucleus of an atom is in an excited state and then releases energy, becoming more stable When a nucleus emits an or β particle, the daughter nucleus is sometimes left in an excited state. It can then jump down to a lower level by emitting a gamma ray ...
Nuclear Chemistry - VCC Library
... neutrons and 6 protons. We can also express this isotope as “carbon-14” or “C-14” (element symbol followed by mass number). Two new terms are nucleons and nuclide. Nucleons are how we refer to protons and neutrons as a group. Nuclide is similar to isotope (atoms of the same element with different ma ...
... neutrons and 6 protons. We can also express this isotope as “carbon-14” or “C-14” (element symbol followed by mass number). Two new terms are nucleons and nuclide. Nucleons are how we refer to protons and neutrons as a group. Nuclide is similar to isotope (atoms of the same element with different ma ...
ib atomic and nuclear physics definitions and concepts
... RADIOACTIVE DECAY: A random and spontaneous process in which unstable nuclei emit a particle (disintegrate). The rate decreases exponentially with time. NATURAL RADIOACTIVE DECAY: The emission of an alpha or beta particle. NUCLEAR STRONG FORCE: The force that holds the particles of a nucleus togethe ...
... RADIOACTIVE DECAY: A random and spontaneous process in which unstable nuclei emit a particle (disintegrate). The rate decreases exponentially with time. NATURAL RADIOACTIVE DECAY: The emission of an alpha or beta particle. NUCLEAR STRONG FORCE: The force that holds the particles of a nucleus togethe ...
Chapter 4: The Structure of the Atom &
... ATOMIC MASS (not to be confused with the mass number) is the weighted average mass of the isotopes of that element. o Example: Element X has two isotopes X-6 (6.015 amu) and X-7 (7.016 amu). X-6 comprises 7.5% of all of element X. X-7 makes up the remaining 92.5%. What is the atomic mass? o NOTE: ...
... ATOMIC MASS (not to be confused with the mass number) is the weighted average mass of the isotopes of that element. o Example: Element X has two isotopes X-6 (6.015 amu) and X-7 (7.016 amu). X-6 comprises 7.5% of all of element X. X-7 makes up the remaining 92.5%. What is the atomic mass? o NOTE: ...
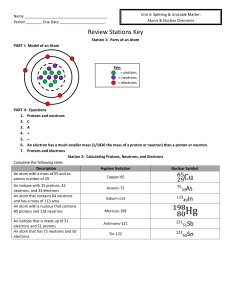
Name Period ______ Due Date Review Stations Key Station 1
... Stopped by paper, wood, cloth, etc. ...
... Stopped by paper, wood, cloth, etc. ...
Nuclear Physics and Radioactivity
... atomic mass number (A) - the number of protons and neutrons in the nucleus of an atom. atomic mass unit - the unit of mass equal to 1/12 the mass of a carbon-12 nucleus; the atomic mass rounded to the nearest whole number is called the mass number. atomic number (Z) - the number of protons in the nu ...
... atomic mass number (A) - the number of protons and neutrons in the nucleus of an atom. atomic mass unit - the unit of mass equal to 1/12 the mass of a carbon-12 nucleus; the atomic mass rounded to the nearest whole number is called the mass number. atomic number (Z) - the number of protons in the nu ...
Binding Energy1
... # protons = # neutrons Pauli Principle - neutrons and protons have spin like electron, and thus ms= 1/2. ...
... # protons = # neutrons Pauli Principle - neutrons and protons have spin like electron, and thus ms= 1/2. ...
notes ch 39 1st half Atomic Nucleus and Radioactivity
... • When the number of protons increases in a nucleus, then the number of neutrons needed to hold the nucleus together must get larger too. • At small atomic numbers, the number of neutrons may be equal or slightly more than the number of protons, but at higher atomic numbers, the number of neutrons m ...
... • When the number of protons increases in a nucleus, then the number of neutrons needed to hold the nucleus together must get larger too. • At small atomic numbers, the number of neutrons may be equal or slightly more than the number of protons, but at higher atomic numbers, the number of neutrons m ...
File - Chemistry with Mr. Patmos
... In one half-life, 40 mg of a radioisotope decays to 20 mg. After two half-lives, 10 mg of radioisotope ...
... In one half-life, 40 mg of a radioisotope decays to 20 mg. After two half-lives, 10 mg of radioisotope ...
Adobe Acrobat file ()
... The nuclear attractive force is stronger than the Coulomb repulsive force at the short ranges within the nucleus ...
... The nuclear attractive force is stronger than the Coulomb repulsive force at the short ranges within the nucleus ...
Topic Review: Nuclear Chemistry 1. The stability of an isotope
... 1. The stability of an isotope depends on the ratio of protons to neutrons in the nucleus. Most nuclei are stable, but some are unstable. These nuclei will spontaneously decay, emitting radiation. Stable isotopes of small atoms have a 1:1 ratio of protons and neutrons. Most radioactive isotopes ...
... 1. The stability of an isotope depends on the ratio of protons to neutrons in the nucleus. Most nuclei are stable, but some are unstable. These nuclei will spontaneously decay, emitting radiation. Stable isotopes of small atoms have a 1:1 ratio of protons and neutrons. Most radioactive isotopes ...
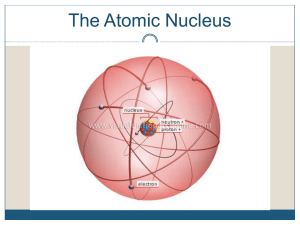
Structure of the Atom
... Note the following symbols: (they are not to scale) = proton (positive charge) = electron (negative charge) = neutron (no charge) The following three diagrams are hydrogen atoms: ...
... Note the following symbols: (they are not to scale) = proton (positive charge) = electron (negative charge) = neutron (no charge) The following three diagrams are hydrogen atoms: ...
nuclear powperpoint
... *Chemical reaction- atoms gain or lose electrons, change their electron configuration *Nuclear reaction- atoms gain or lose protons, neutrons, or energy; cannot be sped up, slowed down or stopped. ...
... *Chemical reaction- atoms gain or lose electrons, change their electron configuration *Nuclear reaction- atoms gain or lose protons, neutrons, or energy; cannot be sped up, slowed down or stopped. ...
Chemistry Standard 2A-Nucleus Section 20.1
... b. atomic number d. neutron-to-proton ratio 5. What is the process in which an unstable atomic nucleus emits charged particles or energy or both? Page answer is found ____________ a. radioactivity c. decomposition b. oxidation d. none of the above 6. When the ____ is not large enough to hold a nucle ...
... b. atomic number d. neutron-to-proton ratio 5. What is the process in which an unstable atomic nucleus emits charged particles or energy or both? Page answer is found ____________ a. radioactivity c. decomposition b. oxidation d. none of the above 6. When the ____ is not large enough to hold a nucle ...