03 Atoms – Nuclides
... Radioactivity is the ability of an unstable atomic nucleus to transform into a stable product or another unstable product while emitting radiation. This transformation and emission of energy is called radioactive decay. A transformation from one element to another is known as a transmutation. The ra ...
... Radioactivity is the ability of an unstable atomic nucleus to transform into a stable product or another unstable product while emitting radiation. This transformation and emission of energy is called radioactive decay. A transformation from one element to another is known as a transmutation. The ra ...
Nuclear Chemistry
... nuclear forces that overcome the electromagnetic repulsion between the protons. b. Students know the energy release per gram of material is much larger in nuclear fusion or fission reactions than in chemical reactions. The change in mass (calculated by E = mc2) is small but significant in nuclear re ...
... nuclear forces that overcome the electromagnetic repulsion between the protons. b. Students know the energy release per gram of material is much larger in nuclear fusion or fission reactions than in chemical reactions. The change in mass (calculated by E = mc2) is small but significant in nuclear re ...
File
... • Gamma rays have no mass, no p+ and no no so they do not alter atomic mass or atomic number. • It is PURE ENERGY which explains the large penetrating power of rays. • They are often emitted along with alpha or beta particles 230 Th 226 Ra + 4 He + ...
... • Gamma rays have no mass, no p+ and no no so they do not alter atomic mass or atomic number. • It is PURE ENERGY which explains the large penetrating power of rays. • They are often emitted along with alpha or beta particles 230 Th 226 Ra + 4 He + ...
Notes - Science With Horne
... • Gamma rays have no mass, no p+ and no no so they do not alter atomic mass or atomic number. • It is PURE ENERGY which explains the large penetrating power of rays. • They are often emitted along with alpha or beta particles 230 Th 226 Ra + 4 He + ...
... • Gamma rays have no mass, no p+ and no no so they do not alter atomic mass or atomic number. • It is PURE ENERGY which explains the large penetrating power of rays. • They are often emitted along with alpha or beta particles 230 Th 226 Ra + 4 He + ...
Chapter 3 Chemical Foundations
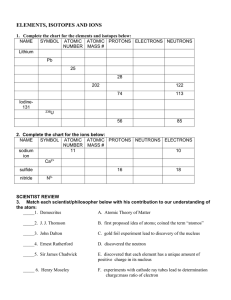
... Atomic Number, Mass Number, and Nucleus atomic number (Z) = mass number (A) = element symbol (X) = Note: mass number= Therefore …. mass number = ……. A= Z + number of neutrons ….. Number of neutrons = A-Z Note: For any given element on the periodic table: Number of protons = In order to symbolically ...
... Atomic Number, Mass Number, and Nucleus atomic number (Z) = mass number (A) = element symbol (X) = Note: mass number= Therefore …. mass number = ……. A= Z + number of neutrons ….. Number of neutrons = A-Z Note: For any given element on the periodic table: Number of protons = In order to symbolically ...
Physics: Principles and Applications, 6e Giancoli
... 5) Compared to the masses of its separate protons and neutrons, the total mass of a stable nucleus is always A) less. B) the same. C) greater. D) zero. 6) When nucleons join to form a stable nucleus, energy is A) destroyed. B) absorbed. C) released. D) not transferred. 7) The binding energy of a nuc ...
... 5) Compared to the masses of its separate protons and neutrons, the total mass of a stable nucleus is always A) less. B) the same. C) greater. D) zero. 6) When nucleons join to form a stable nucleus, energy is A) destroyed. B) absorbed. C) released. D) not transferred. 7) The binding energy of a nuc ...
Name
... 9. The conversion of an atomic nucleus of one element into an atomic nucleus of another element through a loss or gain in the number of protons. 10. High-energy radiation emitted by the nuclei of radioactive atoms. 11. Nuclear fusion produced by high temperature. Down 2. The force of interaction bet ...
... 9. The conversion of an atomic nucleus of one element into an atomic nucleus of another element through a loss or gain in the number of protons. 10. High-energy radiation emitted by the nuclei of radioactive atoms. 11. Nuclear fusion produced by high temperature. Down 2. The force of interaction bet ...
Radioactivity
... • Large nuclei tend to be unstable because the force is not strong enough to hold it together • These nuclei break apart and decay • All nuclei with 83 or more protons are radioactive • Almost all elements with more than 92 protons don’t exist naturally ...
... • Large nuclei tend to be unstable because the force is not strong enough to hold it together • These nuclei break apart and decay • All nuclei with 83 or more protons are radioactive • Almost all elements with more than 92 protons don’t exist naturally ...
printable version
... b. Beta emission occurs in isotopes that have too many neutrons ( more neutrons than protons in small atoms) like Cobalt – 60 ( 27 protons and 33 neutrons) c. Isotopes that have too few neutrons (like nitrogen-13) would attain stability by electron capture or positron emission ...
... b. Beta emission occurs in isotopes that have too many neutrons ( more neutrons than protons in small atoms) like Cobalt – 60 ( 27 protons and 33 neutrons) c. Isotopes that have too few neutrons (like nitrogen-13) would attain stability by electron capture or positron emission ...
Introduction to Radiation and Radioactivity
... Atomic number = Z the number of protons in the atom. Determines which element it is. Atomic mass (or Mass number) = A the total number of protons and neutrons. A = Z + N (# of neutrons) ...
... Atomic number = Z the number of protons in the atom. Determines which element it is. Atomic mass (or Mass number) = A the total number of protons and neutrons. A = Z + N (# of neutrons) ...
Nuclear Chemistry
... study of the nucleus and reactions between nuclei. Remember that virtually all of the mass of an atom resides in the nucleus, as does all of the positive charge. Nuclear energy is a much greater form of energy than bond energy. ...
... study of the nucleus and reactions between nuclei. Remember that virtually all of the mass of an atom resides in the nucleus, as does all of the positive charge. Nuclear energy is a much greater form of energy than bond energy. ...
Atom and Nucleus. Radioactivity. Nuclear Energy.
... in 1932). They both are referred to as nucleons. Atoms of different chemical elements have different number of protons (atomic number) Atoms with the same number of protons and different number of neutrons are called isotopes A nucleus with a particular composition is called a nuclide. 1 H – hydroge ...
... in 1932). They both are referred to as nucleons. Atoms of different chemical elements have different number of protons (atomic number) Atoms with the same number of protons and different number of neutrons are called isotopes A nucleus with a particular composition is called a nuclide. 1 H – hydroge ...
Chapter1
... Z: the number of protons (same as atomic number since the number of protons dictates the chemical properties of the atom) A: Mass number (N + Z) M: Atomic Mass, I: Neutron excess number (I = N – Z) Isotopes have the same number of protons but different numbers of neutrons Isobars have the same mass ...
... Z: the number of protons (same as atomic number since the number of protons dictates the chemical properties of the atom) A: Mass number (N + Z) M: Atomic Mass, I: Neutron excess number (I = N – Z) Isotopes have the same number of protons but different numbers of neutrons Isobars have the same mass ...
Download: Worksheet - New York Science Teacher
... A. Nuclear reactions – the energy released during nuclear reactions is much greater than the energy released during chemical reactions 1. Radioactive decay a.) The stability of an isotope is based on the ratio of neutrons to protons in its nucleus or the binding energy per nucleon. b.) Nuclei that a ...
... A. Nuclear reactions – the energy released during nuclear reactions is much greater than the energy released during chemical reactions 1. Radioactive decay a.) The stability of an isotope is based on the ratio of neutrons to protons in its nucleus or the binding energy per nucleon. b.) Nuclei that a ...
Chapter 21 - Richsingiser.com
... sum of the mass numbers and atomic numbers on the reactant and product sides of the equation must be equal. ...
... sum of the mass numbers and atomic numbers on the reactant and product sides of the equation must be equal. ...
Document
... • Per unit volume, an atom bomb may be millions or billions of times more powerful than TNT. • Nuclear reactions (rxn) occur: neutrons r fired @ closely packed atoms w/ heavy nuclei (uranium or plutonium isotopes). ...
... • Per unit volume, an atom bomb may be millions or billions of times more powerful than TNT. • Nuclear reactions (rxn) occur: neutrons r fired @ closely packed atoms w/ heavy nuclei (uranium or plutonium isotopes). ...
Concept Review 3.1 Introduction to Nuclear
... Electrostatic Force – Like charges repel (+, + and -, -) and opposite charges attract (-, +). As the number of protons in a nucleus increases, the repulsive electrostatic force between protons increases faster than the nuclear force. More neutrons are required to increase the nuclear force and sta ...
... Electrostatic Force – Like charges repel (+, + and -, -) and opposite charges attract (-, +). As the number of protons in a nucleus increases, the repulsive electrostatic force between protons increases faster than the nuclear force. More neutrons are required to increase the nuclear force and sta ...
Nuclear Chemistry Test Topics
... first to perform fission experiments. Radiation consists of particles and energy being given off by unstable nuclei. It cannot be seen or felt, but it is harmful to living things. An isotope of an element differs in the number of neutrons it contains. To determine the mass number of an atom, add the ...
... first to perform fission experiments. Radiation consists of particles and energy being given off by unstable nuclei. It cannot be seen or felt, but it is harmful to living things. An isotope of an element differs in the number of neutrons it contains. To determine the mass number of an atom, add the ...
Chapter 25
... 1. What causes a transmutation of the nucleus to occur? 2. How are nuclear decay reaction equations balanced? 3. Do all radionuclides decay at the same rate? ...
... 1. What causes a transmutation of the nucleus to occur? 2. How are nuclear decay reaction equations balanced? 3. Do all radionuclides decay at the same rate? ...
CHM 123-Chapter 2.7
... has at least one radioactive isotope. • Hydrogen is the only element whose most abundant stable isotope, hydrogen-1, contains more protons (1) than neutrons (0). • The ratio of neutrons to protons gradually increases for elements heavier than calcium. • All isotopes heavier than bismuth209 are radio ...
... has at least one radioactive isotope. • Hydrogen is the only element whose most abundant stable isotope, hydrogen-1, contains more protons (1) than neutrons (0). • The ratio of neutrons to protons gradually increases for elements heavier than calcium. • All isotopes heavier than bismuth209 are radio ...
have shown no evidence
... • Stability is favoured by even numbers of protons and neutrons • Not usually equal numbers • Plotting neutron number (A) against proton number (Z) for all known nuclei, shows area of stability • For very light elements N ≈ Z gives stable elements • 1:1 up to 4020Ca • Ratio gradually rises (A>Z) unt ...
... • Stability is favoured by even numbers of protons and neutrons • Not usually equal numbers • Plotting neutron number (A) against proton number (Z) for all known nuclei, shows area of stability • For very light elements N ≈ Z gives stable elements • 1:1 up to 4020Ca • Ratio gradually rises (A>Z) unt ...