Dr. Audrey Lugo`s AP Chemistry Course Syllabus
... These descriptive facts, including the chemistry involved in environmental and societal issues, should not be isolated from the principles being studied but should be taught throughout the course to illustrate and illuminate the principles. The following areas should be covered: 1. Chemical reactivi ...
... These descriptive facts, including the chemistry involved in environmental and societal issues, should not be isolated from the principles being studied but should be taught throughout the course to illustrate and illuminate the principles. The following areas should be covered: 1. Chemical reactivi ...
Chemistry - Gildredge House
... ‘facilitating subjects’, a subject considered to give students the most options. ...
... ‘facilitating subjects’, a subject considered to give students the most options. ...
Chemistry for Bio 11
... determined number of electrons (first holds two, 2nd and 3rd hold eight ...
... determined number of electrons (first holds two, 2nd and 3rd hold eight ...
Yearly Plan for MYP 1 Science
... Create an appropriate data table and collect data Analyze the data and draw an appropriate conclusion Write up a complete lab report. Evaluate the method, accuracy and reliability. Explain the concept of element and name an atom as the smallest piece of an element Describe the basic structure of an ...
... Create an appropriate data table and collect data Analyze the data and draw an appropriate conclusion Write up a complete lab report. Evaluate the method, accuracy and reliability. Explain the concept of element and name an atom as the smallest piece of an element Describe the basic structure of an ...
Chapter One Outline
... Identifying Matter: Physical Properties Physical properties can be observed and measured without changing the composition of a substance. Examples include temperature, mass, density, etc. Density is the ratio of an objects mass to its volume; D = m/v Chemical Properties A substances chemical propert ...
... Identifying Matter: Physical Properties Physical properties can be observed and measured without changing the composition of a substance. Examples include temperature, mass, density, etc. Density is the ratio of an objects mass to its volume; D = m/v Chemical Properties A substances chemical propert ...
AlBr3 E IO Ionic FU C O Cov Molec C IO Cov Molec Sn E N/A N/A
... combine in the ratio of small, whole numbers to form compounds. ...
... combine in the ratio of small, whole numbers to form compounds. ...
Introductory Chemistry Test Review
... 30. How much heat is absorbed when 50.00 g of carbon reacts in the presence of excess SO2(g) to produce CS2(l) and CO(g) according to the following chemical equation? 5 C(s) + 2 SO2(g) ...
... 30. How much heat is absorbed when 50.00 g of carbon reacts in the presence of excess SO2(g) to produce CS2(l) and CO(g) according to the following chemical equation? 5 C(s) + 2 SO2(g) ...
File - academia science
... Spontaneous generation • States that living organisms develop from nonliving matter • Belief proposed by Aristotle. During 2000 years people thought this was true • Van Helmont: scientist that made an experiment proving this theory to be true • 1870: Thomas Huxley came up with the term abiogenesis ...
... Spontaneous generation • States that living organisms develop from nonliving matter • Belief proposed by Aristotle. During 2000 years people thought this was true • Van Helmont: scientist that made an experiment proving this theory to be true • 1870: Thomas Huxley came up with the term abiogenesis ...
9.1-10.5 Organic Chemistry
... formulas for saturated and unsaturated aliphatic (including cyclic) • Containing up to 10 carbon atoms in the parent chain/cyclic structure • Containing only one type of a functional group or multiple bond • Using the IUPAC nomenclature guidelines 2) Identify types of compounds from the functional g ...
... formulas for saturated and unsaturated aliphatic (including cyclic) • Containing up to 10 carbon atoms in the parent chain/cyclic structure • Containing only one type of a functional group or multiple bond • Using the IUPAC nomenclature guidelines 2) Identify types of compounds from the functional g ...
Chemistry - Solutions
... • Solubility: the amount of a substance that will dissolve in a given amount of solvent to form a saturated solution at a given temperature • Solubility depends on RANDOM MOLECULAR MOTION, which is affected by temperature, pressure and surface ...
... • Solubility: the amount of a substance that will dissolve in a given amount of solvent to form a saturated solution at a given temperature • Solubility depends on RANDOM MOLECULAR MOTION, which is affected by temperature, pressure and surface ...
General Chemistry First Semester Review General
... Phase notations (solid, liquids, gases, aqueous) - aqueous (aq) is written if a solution is used - pure liquids (not a mixture of something) use (l) - solid: This could refer to a multitude of different substances: metals, flakes, crystals, and precipitates; use (s) - gas: Use (g), these are usually ...
... Phase notations (solid, liquids, gases, aqueous) - aqueous (aq) is written if a solution is used - pure liquids (not a mixture of something) use (l) - solid: This could refer to a multitude of different substances: metals, flakes, crystals, and precipitates; use (s) - gas: Use (g), these are usually ...
Ch. 2 - Ltcconline.net
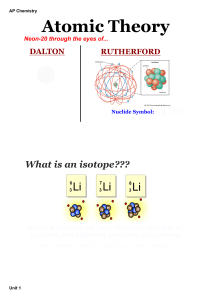
... 3. Define a compound and explain how compounds in living organisms are different from compounds in nonliving things. 4. Describe the structure of an atom. 5. Distinguish between atomic number and atomic weight or mass number of an atom. 6. Define an isotope and explain what makes some isotopes radio ...
... 3. Define a compound and explain how compounds in living organisms are different from compounds in nonliving things. 4. Describe the structure of an atom. 5. Distinguish between atomic number and atomic weight or mass number of an atom. 6. Define an isotope and explain what makes some isotopes radio ...
Spring 2017 - Ventura College Chemistry, Malia Rose-Seisa
... esters, amines, and biochemical amino acids using IUPAC and common systems of nomenclature, in addition to continued ChemV12A knowledge. 2.) Examine, evaluate, and formulate mechanisms for the reactions of aromatics, ketones, aldehydes, carboxylic acids, esters, and amines given reactants and reagen ...
... esters, amines, and biochemical amino acids using IUPAC and common systems of nomenclature, in addition to continued ChemV12A knowledge. 2.) Examine, evaluate, and formulate mechanisms for the reactions of aromatics, ketones, aldehydes, carboxylic acids, esters, and amines given reactants and reagen ...
Lecture 3 Chemistry
... Number of electrons in outer shell determines bonding properties chemical behavior ...
... Number of electrons in outer shell determines bonding properties chemical behavior ...
Science Outline NHPS: Chemistry
... formation and structure of carbon-based polymers, including synthetic polymers, such as polyethylene, and biopolymers, such as carbohydrate. Explain how simple chemical monomers can be combined to create linear, branched and/or cross-linked polymers. Be able to draw structural formulas and name orga ...
... formation and structure of carbon-based polymers, including synthetic polymers, such as polyethylene, and biopolymers, such as carbohydrate. Explain how simple chemical monomers can be combined to create linear, branched and/or cross-linked polymers. Be able to draw structural formulas and name orga ...
A Level Chemistry.pub
... • You will still be able to combine A Levels with other types of qualifications such as BTECs. • These changes are happening at different times for different subjects. • You’ll have lots of support from us before you have to make your final choice of subjects. Specifics for this subject: The first t ...
... • You will still be able to combine A Levels with other types of qualifications such as BTECs. • These changes are happening at different times for different subjects. • You’ll have lots of support from us before you have to make your final choice of subjects. Specifics for this subject: The first t ...
Organic Naming Notes
... Organic Chemistry Organic chemistry is the study of carbon based compounds - This field of chemistry is very important because all living things and many non-living material are organic 1-meth, 2-eth, 3-prop, 4-but, 5-pent, 6-hex, 7-hept, 8-oct, 9-non, 10-dec ...
... Organic Chemistry Organic chemistry is the study of carbon based compounds - This field of chemistry is very important because all living things and many non-living material are organic 1-meth, 2-eth, 3-prop, 4-but, 5-pent, 6-hex, 7-hept, 8-oct, 9-non, 10-dec ...
Basic Chemistry notes
... ______________________—two or more like atoms combined chemically ______________________—two or more different atoms combined chemically ...
... ______________________—two or more like atoms combined chemically ______________________—two or more different atoms combined chemically ...
Writing Formulas
... Remember the algebraic sum of the ions' oxidation numbers must equal zero. (Balance) Learn the polyatomic ions. Learn those ions with multiple oxidation numbers and use Roman numerals to indicate the charge. ...
... Remember the algebraic sum of the ions' oxidation numbers must equal zero. (Balance) Learn the polyatomic ions. Learn those ions with multiple oxidation numbers and use Roman numerals to indicate the charge. ...
Document
... Molarity, or moles per liter (M) A mole of an element or compound is equal to its atomic or molecular weight (sum of atomic weights) in grams One mole of any substance contains exactly the same number of solute particles (6.02 x 1023) 37. Colloids and Suspensions Colloids, or emulsions, are heteroge ...
... Molarity, or moles per liter (M) A mole of an element or compound is equal to its atomic or molecular weight (sum of atomic weights) in grams One mole of any substance contains exactly the same number of solute particles (6.02 x 1023) 37. Colloids and Suspensions Colloids, or emulsions, are heteroge ...
How are Molecules Depicted? - Belle Vernon Area School District
... E level of an atom and determines the chemical properties Lewis Structure = a structure in which e- are represented by dots: dot pairs or dashes b/t 2 atomic symbols represents pairs in covalent bonds ...
... E level of an atom and determines the chemical properties Lewis Structure = a structure in which e- are represented by dots: dot pairs or dashes b/t 2 atomic symbols represents pairs in covalent bonds ...
Programma Inglese XXXII Scuola Corbella
... Back to the “iron age”: recent developments in iron-catalysed chemical transformations ...
... Back to the “iron age”: recent developments in iron-catalysed chemical transformations ...
Organic and Bio-Molecular Chemistry
... by microorganisms. Organic and Bio-Molecular Chemistry concerns in particular, as indicated by the name, the wide variety of compounds that constitute the living organisms; it includes however also structurally related compounds that have been mass-produced in laboratories and by industry in the 20t ...
... by microorganisms. Organic and Bio-Molecular Chemistry concerns in particular, as indicated by the name, the wide variety of compounds that constitute the living organisms; it includes however also structurally related compounds that have been mass-produced in laboratories and by industry in the 20t ...
Branches of Chemistry
... Inorganic chemists study the chemistry of all the elements and their compounds, except for those compounds that contain mainly carbon and hydrogen. Nuclear chemists investigate changes that happen in atomic nuclei. Organic chemists study hydrocarbons – compounds of carbon and hydrogen – and other re ...
... Inorganic chemists study the chemistry of all the elements and their compounds, except for those compounds that contain mainly carbon and hydrogen. Nuclear chemists investigate changes that happen in atomic nuclei. Organic chemists study hydrocarbons – compounds of carbon and hydrogen – and other re ...
Organic chemistry

Organic chemistry is a chemistry subdiscipline involving the scientific study of the structure, properties, and reactions of organic compounds and organic materials, i.e., matter in its various forms that contain carbon atoms. Study of structure includes many physical and chemical methods to determine the chemical composition and the chemical constitution of organic compounds and materials. Study of properties includes both physical properties and chemical properties, and uses similar methods as well as methods to evaluate chemical reactivity, with the aim to understand the behavior of the organic matter in its pure form (when possible), but also in solutions, mixtures, and fabricated forms. The study of organic reactions includes probing their scope through use in preparation of target compounds (e.g., natural products, drugs, polymers, etc.) by chemical synthesis, as well as the focused study of the reactivities of individual organic molecules, both in the laboratory and via theoretical (in silico) study.The range of chemicals studied in organic chemistry include hydrocarbons (compounds containing only carbon and hydrogen), as well as myriad compositions based always on carbon, but also containing other elements, especially oxygen, nitrogen, sulfur, phosphorus (these, included in many organic chemicals in biology) and the radiostable elements of the halogens.In the modern era, the range extends further into the periodic table, with main group elements, including:Group 1 and 2 organometallic compounds, i.e., involving alkali (e.g., lithium, sodium, and potassium) or alkaline earth metals (e.g., magnesium)Metalloids (e.g., boron and silicon) or other metals (e.g., aluminium and tin)In addition, much modern research focuses on organic chemistry involving further organometallics, including the lanthanides, but especially the transition metals; (e.g., zinc, copper, palladium, nickel, cobalt, titanium and chromium)Finally, organic compounds form the basis of all earthly life and constitute a significant part of human endeavors in chemistry. The bonding patterns open to carbon, with its valence of four—formal single, double, and triple bonds, as well as various structures with delocalized electrons—make the array of organic compounds structurally diverse, and their range of applications enormous. They either form the basis of, or are important constituents of, many commercial products including pharmaceuticals; petrochemicals and products made from them (including lubricants, solvents, etc.); plastics; fuels and explosives; etc. As indicated, the study of organic chemistry overlaps with organometallic chemistry and biochemistry, but also with medicinal chemistry, polymer chemistry, as well as many aspects of materials science.