Chemical Reactions and Equations
... in one substance on each side of the equation, make sure that each side of the equation has an equal # of that element. Proceed with all elements. Remember that changing the # of one element may alter elements that have already been ...
... in one substance on each side of the equation, make sure that each side of the equation has an equal # of that element. Proceed with all elements. Remember that changing the # of one element may alter elements that have already been ...
Atoms, compounds and elements - Mrs. Tes de Luna`s Science Class
... ◦ The first part of his theory states that all matter is made of atoms, which are indivisible. ◦ The second part of the theory says all atoms of a given element are identical in mass and properties. ◦ The third part says compounds are combinations of two or more different types of atoms. ◦ The fourt ...
... ◦ The first part of his theory states that all matter is made of atoms, which are indivisible. ◦ The second part of the theory says all atoms of a given element are identical in mass and properties. ◦ The third part says compounds are combinations of two or more different types of atoms. ◦ The fourt ...
Fundamentals of Chemistry
... atoms in which reactant compounds are converted into product compounds. • During a chem rxn, chemical bonds in the reactants are broken and chemical bonds in the products are created. • A rxn is accompanied by a change in energy (i.e. heat can be absorbed or given off), color, state of matter, etc. ...
... atoms in which reactant compounds are converted into product compounds. • During a chem rxn, chemical bonds in the reactants are broken and chemical bonds in the products are created. • A rxn is accompanied by a change in energy (i.e. heat can be absorbed or given off), color, state of matter, etc. ...
Chapter 2 – Chemical Composition of the Body
... • Electronegativity is the property that describes an atom’s attraction for a shared pair of e-. • If two atoms with different electronegativity values share e-, i.e. form a covalent bond, one of the atoms will have a “larger share” of the e-. • This produces a molecule with differently charged en ...
... • Electronegativity is the property that describes an atom’s attraction for a shared pair of e-. • If two atoms with different electronegativity values share e-, i.e. form a covalent bond, one of the atoms will have a “larger share” of the e-. • This produces a molecule with differently charged en ...
Review Sheet for Chemistry* First Semester Final
... Put the following into a graphic organizer/flowchart and define each: Matter Pure substances – elements and compounds Mixtures – homogeneous (solutions) and heterogeneous Identify the following as pure substances, homogeneous mixtures or heterogeneous mixtures: a. copper ____________________ ...
... Put the following into a graphic organizer/flowchart and define each: Matter Pure substances – elements and compounds Mixtures – homogeneous (solutions) and heterogeneous Identify the following as pure substances, homogeneous mixtures or heterogeneous mixtures: a. copper ____________________ ...
Lecture 1: RDCH 710 Introduction
... Variation of Np behavior based on redox * Need to understand redox kinetics * Reduction of Np(VI) by a range of compounds Back extraction of Np(V) can be used to separate from Pu and U * Controlled Np(VI) reduction in presence of Pu(III) ...
... Variation of Np behavior based on redox * Need to understand redox kinetics * Reduction of Np(VI) by a range of compounds Back extraction of Np(V) can be used to separate from Pu and U * Controlled Np(VI) reduction in presence of Pu(III) ...
Atoms, Elements, Compounds File
... SOL 6.4 Atoms, Elements, compounds The student will investigate and understand that all matter is made up of atoms. Key concepts include ...
... SOL 6.4 Atoms, Elements, compounds The student will investigate and understand that all matter is made up of atoms. Key concepts include ...
Cosmetology Learning Module 12
... matter and how matter changes under different chemical conditions Organic Chemistry – is the study of substances that contain carbon All living things are made up of compounds that contain carbon Organic compounds will burn ...
... matter and how matter changes under different chemical conditions Organic Chemistry – is the study of substances that contain carbon All living things are made up of compounds that contain carbon Organic compounds will burn ...
Compound Name
... Balance chemical equations by adding coefficients and not by changing compound subscripts; ...
... Balance chemical equations by adding coefficients and not by changing compound subscripts; ...
chapter 6 sec 2 resonance structure
... H is 2.1 and O is 3.5. 3.5 – 2.1 = 1.4 so the bond between H and O is a polar covalent bond. By definition a neutral group of atoms held together by covalent bonds is a molecule. So, the H2O particle is a molecule H2O is a molecule which makes H2O a molecular compound and a molecular formula. But H2 ...
... H is 2.1 and O is 3.5. 3.5 – 2.1 = 1.4 so the bond between H and O is a polar covalent bond. By definition a neutral group of atoms held together by covalent bonds is a molecule. So, the H2O particle is a molecule H2O is a molecule which makes H2O a molecular compound and a molecular formula. But H2 ...
CHM 2045C - State College of Florida
... (5 Credit Hours) (A.A.) Three hours lecture, three hours laboratory per week. Prerequisites: Completion of MAC 1105. Completion of CHM 1025C with a grade of “C” or better or one year of high school college preparatory honors or AP chemistry within last three ...
... (5 Credit Hours) (A.A.) Three hours lecture, three hours laboratory per week. Prerequisites: Completion of MAC 1105. Completion of CHM 1025C with a grade of “C” or better or one year of high school college preparatory honors or AP chemistry within last three ...
In organic chemistry, we studied a lot about the essential elements
... the technology nowadays, Some Alkaloid can be useful in Medical field, especially in pharmacology. Phosphorus: is an organic phosphate. It is very important for life. It is a component of DNA, RNA, in bonding with sugar Ribose to form Sugar-Phosphate backbone . The ATP molecule is also has Phosphate ...
... the technology nowadays, Some Alkaloid can be useful in Medical field, especially in pharmacology. Phosphorus: is an organic phosphate. It is very important for life. It is a component of DNA, RNA, in bonding with sugar Ribose to form Sugar-Phosphate backbone . The ATP molecule is also has Phosphate ...
Chapter 5 – Chemical Reactions
... concentrated acid will react faster than a dilute acid) Add a catalyst – a catalyst is a chemical that speeds up a reaction but does not get used up by the reaction ...
... concentrated acid will react faster than a dilute acid) Add a catalyst – a catalyst is a chemical that speeds up a reaction but does not get used up by the reaction ...
Chemical reaction
... atoms of 2 or more elements • A molecule is the simplest part of a substance that retains all of the properties of the substance ...
... atoms of 2 or more elements • A molecule is the simplest part of a substance that retains all of the properties of the substance ...
Electron Arrangement
... A fraction is a group of compounds with boiling points within a given range. Fractions can be separated by distillation because they have different boiling points so evaporate at different times when crude oil is heated. They then cool down to be collected as liquids. The fractions from crude oil c ...
... A fraction is a group of compounds with boiling points within a given range. Fractions can be separated by distillation because they have different boiling points so evaporate at different times when crude oil is heated. They then cool down to be collected as liquids. The fractions from crude oil c ...
Chemistry of life
... Why is chemistry important to biology? • Many of our everyday experiences depend upon chemistry. – For example: Making kool aid or sweet tea. ...
... Why is chemistry important to biology? • Many of our everyday experiences depend upon chemistry. – For example: Making kool aid or sweet tea. ...
Synthesis/Decomposition Reactions
... 5. In terms of reactants and products, describe how you would recognize a reaction as being a decomposition reaction: ...
... 5. In terms of reactants and products, describe how you would recognize a reaction as being a decomposition reaction: ...
Chapter 10
... Predicting Products of Synthesis Reactions For Synthesis Reactions For metals that only form one cation, determine the charge on the ion of each element (metallic and nonmetallic) and form a compound from the two ions. If one of the elements forms more than one cation or 2 nonmetals are combined ...
... Predicting Products of Synthesis Reactions For Synthesis Reactions For metals that only form one cation, determine the charge on the ion of each element (metallic and nonmetallic) and form a compound from the two ions. If one of the elements forms more than one cation or 2 nonmetals are combined ...
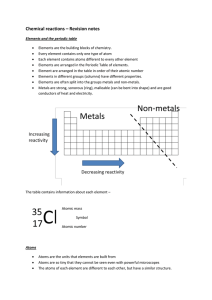
Chemical reactions revision
... Elements are the building blocks of chemistry. Every element contains only one type of atom Each element contains atoms different to every other element Elements are arranged in the Periodic Table of elements. Element are arranged in the table in order of their atomic number Elements in different gr ...
... Elements are the building blocks of chemistry. Every element contains only one type of atom Each element contains atoms different to every other element Elements are arranged in the Periodic Table of elements. Element are arranged in the table in order of their atomic number Elements in different gr ...
Chapter 2
... electrons is shared Double covalent bond – 2 pairs of electrons shared Triple covalent bond – 3 pairs of electrons shared ...
... electrons is shared Double covalent bond – 2 pairs of electrons shared Triple covalent bond – 3 pairs of electrons shared ...
Organic chemistry

Organic chemistry is a chemistry subdiscipline involving the scientific study of the structure, properties, and reactions of organic compounds and organic materials, i.e., matter in its various forms that contain carbon atoms. Study of structure includes many physical and chemical methods to determine the chemical composition and the chemical constitution of organic compounds and materials. Study of properties includes both physical properties and chemical properties, and uses similar methods as well as methods to evaluate chemical reactivity, with the aim to understand the behavior of the organic matter in its pure form (when possible), but also in solutions, mixtures, and fabricated forms. The study of organic reactions includes probing their scope through use in preparation of target compounds (e.g., natural products, drugs, polymers, etc.) by chemical synthesis, as well as the focused study of the reactivities of individual organic molecules, both in the laboratory and via theoretical (in silico) study.The range of chemicals studied in organic chemistry include hydrocarbons (compounds containing only carbon and hydrogen), as well as myriad compositions based always on carbon, but also containing other elements, especially oxygen, nitrogen, sulfur, phosphorus (these, included in many organic chemicals in biology) and the radiostable elements of the halogens.In the modern era, the range extends further into the periodic table, with main group elements, including:Group 1 and 2 organometallic compounds, i.e., involving alkali (e.g., lithium, sodium, and potassium) or alkaline earth metals (e.g., magnesium)Metalloids (e.g., boron and silicon) or other metals (e.g., aluminium and tin)In addition, much modern research focuses on organic chemistry involving further organometallics, including the lanthanides, but especially the transition metals; (e.g., zinc, copper, palladium, nickel, cobalt, titanium and chromium)Finally, organic compounds form the basis of all earthly life and constitute a significant part of human endeavors in chemistry. The bonding patterns open to carbon, with its valence of four—formal single, double, and triple bonds, as well as various structures with delocalized electrons—make the array of organic compounds structurally diverse, and their range of applications enormous. They either form the basis of, or are important constituents of, many commercial products including pharmaceuticals; petrochemicals and products made from them (including lubricants, solvents, etc.); plastics; fuels and explosives; etc. As indicated, the study of organic chemistry overlaps with organometallic chemistry and biochemistry, but also with medicinal chemistry, polymer chemistry, as well as many aspects of materials science.