Exam 3 Review Sheet
... The following questions have been pulled from various exams and review sheets from some of my previous classes. The review is not complete! Make sure you study the assigned homeworks, go through suggested problems and read through the Chapter Notes and your lecture notes. Topic Outline You should fe ...
... The following questions have been pulled from various exams and review sheets from some of my previous classes. The review is not complete! Make sure you study the assigned homeworks, go through suggested problems and read through the Chapter Notes and your lecture notes. Topic Outline You should fe ...
Chemistry Unit Test Review
... Students added liver to hydrogen peroxide. The mass of the substance after the reaction took place was less than the mass before. What might have accounted for the mass being different after? ...
... Students added liver to hydrogen peroxide. The mass of the substance after the reaction took place was less than the mass before. What might have accounted for the mass being different after? ...
Chapter 2 (Hill/Petrucci/McCreary/Perry This chapter deals with
... 2. in chemical reactions, atoms are neither created nor destroyed 3. atoms of each element have unique properties - all atoms of a given atom are identical and have identical masses and other properties 4. chemical reactions involve the uniting or the separation of atoms of different elements Dalton ...
... 2. in chemical reactions, atoms are neither created nor destroyed 3. atoms of each element have unique properties - all atoms of a given atom are identical and have identical masses and other properties 4. chemical reactions involve the uniting or the separation of atoms of different elements Dalton ...
Chapter 4 The Structure of Matter
... • 3. Use parentheses around them when more than one is needed in the formula. • a. (NH4)2SO4 - ammonium sulfate • b. NH4NO3 - ammonium nitrate ...
... • 3. Use parentheses around them when more than one is needed in the formula. • a. (NH4)2SO4 - ammonium sulfate • b. NH4NO3 - ammonium nitrate ...
7A SCIENCE FINAL REVIEW - MERRICK 7th SCIENCE REVIEW
... ___ Recognize elements from compounds if given the chemical symbol or a model. ___ Describe the difference between a chemical and physical property of matter, give examples of each. ___ Describe the difference between a chemical and physical change, list examples of each. ___ List evidence to show a ...
... ___ Recognize elements from compounds if given the chemical symbol or a model. ___ Describe the difference between a chemical and physical property of matter, give examples of each. ___ Describe the difference between a chemical and physical change, list examples of each. ___ List evidence to show a ...
Elements, Compounds and Chemical Reactions
... have both PHYSICAL and CHEMICAL properties. A property that can be observed, measured or changed without changing the substance itself is called a physical property. ...
... have both PHYSICAL and CHEMICAL properties. A property that can be observed, measured or changed without changing the substance itself is called a physical property. ...
Elements, basic principles, periodic table
... Ionization energy measures how easy or hard it is to remove an electron from an element or ion. Energies of filled electronic orbitals give rise to common oxidation states for individual elements. Electronic structure determines ionic charge and IE. ...
... Ionization energy measures how easy or hard it is to remove an electron from an element or ion. Energies of filled electronic orbitals give rise to common oxidation states for individual elements. Electronic structure determines ionic charge and IE. ...
Chapter 4
... Compute the amount of product produced or reactant consumed by two or more simultaneous reactions. ...
... Compute the amount of product produced or reactant consumed by two or more simultaneous reactions. ...
Lecture Resource ()
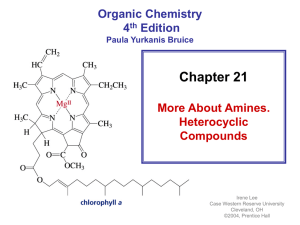
... • Most drugs, vitamins, and many other natural products are heterocycles • A natural product is a compound synthesized by a plant or an animal ...
... • Most drugs, vitamins, and many other natural products are heterocycles • A natural product is a compound synthesized by a plant or an animal ...
Unit 1 science of chemistry
... Elements are the simplest form of matter that has its unique set of properties. Ex. Gold is an element. All atoms of gold have the same properties. Elements are shown in the Periodic Table. There are more than 100 elements, most of them occur naturally. Elements are represented by one (a cap ...
... Elements are the simplest form of matter that has its unique set of properties. Ex. Gold is an element. All atoms of gold have the same properties. Elements are shown in the Periodic Table. There are more than 100 elements, most of them occur naturally. Elements are represented by one (a cap ...
DALTON`S ATOMIC THEORY - 1808: Publication of Dalton`s "A New
... - Dalton's theory sets LIMITS on what can be done with chemistry. For example: Chemistry can't convert lead (an element) into gold (another element). Sorry, alchemists! You can't have a compound form in a chemical reaction that contains an element that was not in your starting materials. You can onl ...
... - Dalton's theory sets LIMITS on what can be done with chemistry. For example: Chemistry can't convert lead (an element) into gold (another element). Sorry, alchemists! You can't have a compound form in a chemical reaction that contains an element that was not in your starting materials. You can onl ...
SAMPLE QUESTION PAPER-II Chemistry (Theory) Class-XII
... Neetu and Asha look organic compound synthesis as their chemistry project. They prepare benzene diazonium chloride and stored it at room temperature. Due to holiday, they start preparing azodye but it cannot be prepared. Then their friend Reena told them to prepare benzene diazonium chloride again a ...
... Neetu and Asha look organic compound synthesis as their chemistry project. They prepare benzene diazonium chloride and stored it at room temperature. Due to holiday, they start preparing azodye but it cannot be prepared. Then their friend Reena told them to prepare benzene diazonium chloride again a ...
Chemistry to Remember
... Electrovalence is the transferring of electrons to form a compound and the creation of ions that are electrically charged particles with properties totally different from the atom from which they came. Covalence is sharing pairs of electrons via a single, double, or triple bond. Radicals are cluster ...
... Electrovalence is the transferring of electrons to form a compound and the creation of ions that are electrically charged particles with properties totally different from the atom from which they came. Covalence is sharing pairs of electrons via a single, double, or triple bond. Radicals are cluster ...
Class Notes
... Materials are made of one or more substances. A single substance material is a pure substance, while a material composed of several substances is classified as a mixture. ...
... Materials are made of one or more substances. A single substance material is a pure substance, while a material composed of several substances is classified as a mixture. ...
2.2 WATER POLLUTION Definition: Any alteration in physical
... Oxygen Demand” (BOD) and “Chemical Oxygen Demand” (COD). BOD of sewage is defined as amount of free oxygen required by the bacteria, under aerobic condition, for the decomposition of organic matter at 20oC in 5 days. The unit of BOD is mg/l or ppm. The average BOD of sewages is between 100 to 150 mg ...
... Oxygen Demand” (BOD) and “Chemical Oxygen Demand” (COD). BOD of sewage is defined as amount of free oxygen required by the bacteria, under aerobic condition, for the decomposition of organic matter at 20oC in 5 days. The unit of BOD is mg/l or ppm. The average BOD of sewages is between 100 to 150 mg ...
Chemical Equations Balancing Chemical Equations Try One…
... (the starting substances) and the products (what is made), the arrow is the same as an “equals sign” (=) in math for the number of each type of atom must be equal on both sides (“yields” or “produces”) ...
... (the starting substances) and the products (what is made), the arrow is the same as an “equals sign” (=) in math for the number of each type of atom must be equal on both sides (“yields” or “produces”) ...
2011-2012 Summer Packet - Tenafly Public Schools
... D Physical & chemical properties: A physical property is a characteristic of matter that can be determined without changing the chemical formula of the substance. These properties include density, freezing point, or melting point (same point) boiling point, hardness, electrical conductivity, malleab ...
... D Physical & chemical properties: A physical property is a characteristic of matter that can be determined without changing the chemical formula of the substance. These properties include density, freezing point, or melting point (same point) boiling point, hardness, electrical conductivity, malleab ...
Unit 6 Jeopardy review - Fort Thomas Independent Schools
... DAILY DOUBLE! Chemicals that act as biological catalysts by speeding up reactions in living things. ...
... DAILY DOUBLE! Chemicals that act as biological catalysts by speeding up reactions in living things. ...
Chapter 2 Study Guides
... ______ 7. atoms held together by covalent bonds ______ 8. composed of different types of atoms ______ 9. composed of one type of atom ...
... ______ 7. atoms held together by covalent bonds ______ 8. composed of different types of atoms ______ 9. composed of one type of atom ...
can be determined without changing the identity of matter
... We classify PROPERTIES of substances by whether or not you must change the identity of a substance to obtain information about the property ...
... We classify PROPERTIES of substances by whether or not you must change the identity of a substance to obtain information about the property ...
Chemical Reactions Chemistry - is the study of matter, its properties
... Chemical Property - is a characteristic behavior that occurs when a substance changes to a new substance. It may determine the transformation which is about to take place. The change, itself, which occurs is called the chemical change. The starting materials in such a change are called reactants and ...
... Chemical Property - is a characteristic behavior that occurs when a substance changes to a new substance. It may determine the transformation which is about to take place. The change, itself, which occurs is called the chemical change. The starting materials in such a change are called reactants and ...
matter and its reactivity. Objects in the universe are composed of
... 3.3a All matter is made up of atoms. Atoms are far too small to see with a light microscope. 3.3c Atoms may join together in well-defined molecules or may be arranged in regular geometric patterns. 3.3d Interactions among atoms and/or molecules result in chemical reactions. 3.3e The atoms of any on ...
... 3.3a All matter is made up of atoms. Atoms are far too small to see with a light microscope. 3.3c Atoms may join together in well-defined molecules or may be arranged in regular geometric patterns. 3.3d Interactions among atoms and/or molecules result in chemical reactions. 3.3e The atoms of any on ...
Chemical Reaction Basics
... Requirements for Chemical Equations 1. Must represent all known facts (states of reactants and products, etc.) *Assume reactions take place at room temperature unless noted otherwise* 2. Must contain correctly written formulas 3. Must satisfy the Law of Conservation of Mass (*Balanced*) ...
... Requirements for Chemical Equations 1. Must represent all known facts (states of reactants and products, etc.) *Assume reactions take place at room temperature unless noted otherwise* 2. Must contain correctly written formulas 3. Must satisfy the Law of Conservation of Mass (*Balanced*) ...
Syllabus
... basic or universal science. There are hundreds of different fields and subfields of chemistry with very specific journals dedicated to each. Finally, chemistry is a practical science that can be applied in everyday life. For example, you use chemistry when you clean your house and when you read the ...
... basic or universal science. There are hundreds of different fields and subfields of chemistry with very specific journals dedicated to each. Finally, chemistry is a practical science that can be applied in everyday life. For example, you use chemistry when you clean your house and when you read the ...
Organic chemistry

Organic chemistry is a chemistry subdiscipline involving the scientific study of the structure, properties, and reactions of organic compounds and organic materials, i.e., matter in its various forms that contain carbon atoms. Study of structure includes many physical and chemical methods to determine the chemical composition and the chemical constitution of organic compounds and materials. Study of properties includes both physical properties and chemical properties, and uses similar methods as well as methods to evaluate chemical reactivity, with the aim to understand the behavior of the organic matter in its pure form (when possible), but also in solutions, mixtures, and fabricated forms. The study of organic reactions includes probing their scope through use in preparation of target compounds (e.g., natural products, drugs, polymers, etc.) by chemical synthesis, as well as the focused study of the reactivities of individual organic molecules, both in the laboratory and via theoretical (in silico) study.The range of chemicals studied in organic chemistry include hydrocarbons (compounds containing only carbon and hydrogen), as well as myriad compositions based always on carbon, but also containing other elements, especially oxygen, nitrogen, sulfur, phosphorus (these, included in many organic chemicals in biology) and the radiostable elements of the halogens.In the modern era, the range extends further into the periodic table, with main group elements, including:Group 1 and 2 organometallic compounds, i.e., involving alkali (e.g., lithium, sodium, and potassium) or alkaline earth metals (e.g., magnesium)Metalloids (e.g., boron and silicon) or other metals (e.g., aluminium and tin)In addition, much modern research focuses on organic chemistry involving further organometallics, including the lanthanides, but especially the transition metals; (e.g., zinc, copper, palladium, nickel, cobalt, titanium and chromium)Finally, organic compounds form the basis of all earthly life and constitute a significant part of human endeavors in chemistry. The bonding patterns open to carbon, with its valence of four—formal single, double, and triple bonds, as well as various structures with delocalized electrons—make the array of organic compounds structurally diverse, and their range of applications enormous. They either form the basis of, or are important constituents of, many commercial products including pharmaceuticals; petrochemicals and products made from them (including lubricants, solvents, etc.); plastics; fuels and explosives; etc. As indicated, the study of organic chemistry overlaps with organometallic chemistry and biochemistry, but also with medicinal chemistry, polymer chemistry, as well as many aspects of materials science.